Abstract
Osteoarthritis (OA) is a chronic degenerative joint disorder that leads to disability and affects more than 500 million population worldwide. OA was believed to be caused by the wearing and tearing of articular cartilage, but it is now more commonly referred to as a chronic whole-joint disorder that is initiated with biochemical and cellular alterations in the synovial joint tissues, which leads to the histological and structural changes of the joint and ends up with the whole tissue dysfunction. Currently, there is no cure for OA, partly due to a lack of comprehensive understanding of the pathological mechanism of the initiation and progression of the disease. Therefore, a better understanding of pathological signaling pathways and key molecules involved in OA pathogenesis is crucial for therapeutic target design and drug development. In this review, we first summarize the epidemiology of OA, including its prevalence, incidence and burdens, and OA risk factors. We then focus on the roles and regulation of the pathological signaling pathways, such as Wnt/β-catenin, NF-κB, focal adhesion, HIFs, TGFβ/ΒΜP and FGF signaling pathways, and key regulators AMPK, mTOR, and RUNX2 in the onset and development of OA. In addition, the roles of factors associated with OA, including MMPs, ADAMTS/ADAMs, and PRG4, are discussed in detail. Finally, we provide updates on the current clinical therapies and clinical trials of biological treatments and drugs for OA. Research advances in basic knowledge of articular cartilage biology and OA pathogenesis will have a significant impact and translational value in develo** OA therapeutic strategies.
Similar content being viewed by others
Introduction
Osteoarthritis (OA) is one of the most common types of arthritis and a chronic degenerative and disabling disease characterized by complex disorders of the whole synovial joint,1 including structural defects of hyaline articular cartilage, loss of intact subchondral bone, tissue hypertrophy and increasing of vascularity in the synovium, and instability of the tendons and ligaments (Fig. 1). In 2021, >22% of adults older than 40 had knee OA, and it is estimated that over 500 million individuals are currently affected by OA worldwide.2 Lacking long-term clinical treatment, OA patients at the end-stage of the disease are ultimately subjected to joint replacement surgery. Joint replacement surgery is growing at a rate of 10% per year globally, and 95% is performed for OA patients.3 However, the lifespan of the artificial joint is limited, and the risk of poor outcomes exists. By 2020, OA is globally estimated to be the fourth leading cause of disability, with a huge amount of medical and healthcare costs and indirect costs caused by loss of jobs and early retirement.
Phenotypes of Osteoarthritis (OA). Clinic evidence shows that the majority of OA patients have a diversity of OA phenotypes, including articular cartilage erosion, synovial hyperplasia, abnormal angiogenesis, synovial inflammation, subchondral bone disturbance, ligaments and tendons instability, and joint stiffness. Left-half side shows the structure of the normal synovial joint. Right-half side showed the possible alterations of synovial joint structure and symptoms in osteoarthritis
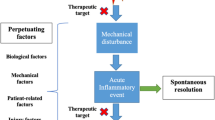
There is currently no cure for OA. It has been a long time since clinical treatments of OA focused on improving joint pain symptoms rather than on the decline of the disease progression. In recent years, strategies for OA have been shifted to its early prevention and halt or delay OA progression before massive destruction occurs. Therefore, understanding and identifying potential biomarkers and therapeutic targets at different stages of OA are urgent. Scientists and clinicians have devoted great efforts to defining major signaling pathways and molecules that play essential roles in the initiation and development of OA and could finally be developed as potential therapeutical targets to slow down or limit the damage to synovial joints.
Besides the updated epidemiology of OA, including its prevalence, incidence, burden, and risk factors, we have reviewed our current understanding of the pathogenesis in terms of synovial tissue interactions and cellular biology in OA, as well as pathological signaling pathways and essential molecules of OA. We have summarized the roles and functions of those pathological molecular signaling pathways and key molecules in different components of the synovial joints at different stages of OA and their related clinical relevance. We have finally reviewed current clinical therapies applied to OA patients and updated clinical trials of new drugs and biological treatments for future OA treatment.
Prevalence
Osteoarthritis (OA) is among the most prevalent diseases globally, which affects multiple joints, including the hip, knee, ankle, hand, and temporal-mandibular joint (TMJ) and other joints.1,4,5 The knee, hand, and hip joints are most susceptible to develo** OA.6 During the past century, the prevalence of OA has grown rapidly in part due to recent increases in lifespan and body weight.7,8 According to a large cohort study in the United States, the prevalence of knee OA has increased by 2.1-fold since the 1950s.9 It is anticipated that, by 2032, the prevalence of OA will rise from 26.6% to 29.5%.10 The prevalence rate of OA can be variable in different studies based on the definitions of OA, e.g., radiographic OA or symptomatic OA. In general, radiographic OA is more prevalent than symptomatic OA.11,43,44,45 In addition, aging-related mitochondrial dysfunction that induces oxidative stress, characterized by excessive accumulation of the reactive oxygen species (ROS) with the imbalance of energy metabolism of articular chondrocytes, is believed to promote articular chondrocyte apoptosis and articular cartilage destruction.46,47,48 Furthermore, age-related inflammation in the synovial joint, which is also associated with SASP, leads to destructive changes in the extracellular matrix (ECM) of the articular cartilage and promotes OA.49
Obesity is another major risk factor that leads to a higher incidence of hip and knee OA. Obesity is one of the most significant risk factors for knee OA partly because the excessive weight of obese patients leads to an abnormal increase in mechanical loading on knee joints, which results in the wearing and tearing of articular cartilage accompanied by ligament destruction and eventually leads to the occurrence of OA. Surprisingly, obese patients also have a higher incidence of OA in the hands that do not usually bear the weight.50,51 This leads to the general belief that it is the systemic factor(s) released by other tissues that induce OA in obese patients. In obese patients, cytokines released by adipocytes, also known as “adipokines”, such as resistin, visfatin, leptin, omentin, adiponectin, retinol-binding protein 4 (RBP4), and other factors, were reported to be associated with promoting the initiation and progression of OA.52,53,54,55,56 Furthermore, cytokines, such as TNF-α, IL-1, IL-6, and IL-8, were shown to trigger joint inflammation, which leads to ECM breakdown and cartilage degeneration.
OA affects more than 500 million populations worldwide, with a higher prevalence in the female gender than in males. Women are known to be more susceptible to OA onset and development than men are.6,57,58 Several studies showed that OA development could be triggered by the plunge in sex hormone levels in menopausal women.59,60 Besides, compared to male OA patients, female patients were reported to have higher levels of joint inflammation and clinical pain, thinner articular cartilage, and severe physical joint mobilities.57,60,61 The potential contributing factors for this gender difference in OA are not fully understood and need further attention in the OA research community.
Knee injury is another major risk factor for knee OA. Post-traumatic OA is one of the OA subtypes that occurs in those joints that have been injured. Current studies have shown that joints that have been traumatized are five times more likely to develop OA than joints that have never been damaged.62 U.S. clinical statistics predict that post-traumatic OA accounts for 12–42% of OA (the proportion varies by age), and the actual proportion could be higher.3 Trauma in the joints has been demonstrated to induce massive gene expression alterations in different compartments of knee joints. Besides injury, sports-related excessive joint loading also increases the chance of OA development. Professional athletes of high-impact sports present a higher prevalence of early knee OA than non-professional athletes and the universal population.63 The new technology of instrument innovations has been developed and utilized for investigating the role of joint mechanical stress in OA pathogenesis, which could study the pattern, force, and duration of mechanical stress on joint loading.64,65 Our advanced knowledge of how mechanical loading contributes to OA onset and progression is just beginning to be used in the applications of biomechanics assessment for guiding the clinical physical therapy of OA patients.66 However, the molecular mechanisms of how mechanical stress contributes to OA onset and development need to be investigated in great detail.
In comparison, few genetic mutations have been confirmed to be linked to human OA before. It is until recently that genetics has been discovered as a risk factor in 11 types of OA, including OAs in the hand, hip, and spine.67 A recently reported genome-wide association study (GWAS) meta-analysis of more than 820,000 East Asian and European individuals from 13 international cohorts of 9 populations, including over 170,000 OA patients, identified around 10,000 significantly associated single-nucleotide variants (SNVs), in which 100 were unique and showed independent genetic correlation with OA phenotypes and symptoms.67 Among these identified 100 SNVs, 60 were genome-wide significantly associated with more than one type of OA, and 77 potential effector genes were identified. Though genetic studies identified risk variants associated with new molecular signals and already reported effector genes contributing to the OA development,68,69,70,71 these genetic risk data need to be further verified and investigated to reveal options for the translational intervention of OA.
Clinical symptoms
Clinical symptoms of OA include joint functional limitations, stiffness, pain, disability of walking or running, and probably other symptoms.1,72,73 Bony enlargement and swollen and inflamed joints could be found in OA patients in physical examination. Clinical radiographic examination, such as MRI (magnetic resonance imaging), is able to visualize marginal osteophytes, joint space narrowing, structural changes of osteochondral tissue, and other OA lesions. Pain is one of the most distinctive symptoms and the main reason OA patients seek medical help,72 but the underlining mechanisms of OA pain are still poorly understood. Pain is a clinical indicator of tissue damage, inflammations, or disorders of the nervous system.74,75 Articular cartilage is an avascular tissue without any nerve invasion, and OA pain can happen both before and after an articular cartilage lesion detected by the imaging system. Therefore, it is unlikely that the destruction of articular cartilage directly causes OA pain. OA pain has been reported to be associated with synovitis and bone marrow lesion,76,77 as well as alterations in subchondral bone, osteophyte formation, abnormalities of infrapatellar fat pads, and lesion of ligaments, in which tissues have highly distributed sensory nerves.78 Molecular mechanisms underlying OA pain have been comprehensively updated and discussed in two recent review articles.79,80
Pathogenesis
During the past decades, the pathogenesis of OA has been extensively studied.81 Although its risk factors were characterized, and the structural changes of the synovial joint in OA are well understood, the complex pathological mechanisms of the onset and development of OA remain elusive. We summarize our current understanding of OA pathogenesis from the perspective of tissue interactions, changes in cellular biology, and pathogenic signaling pathways and molecules.
Tissue interactions in OA
Numerous studies reported that subchondral bone sclerosis could be one of the major reasons that cause aging-related OA and that the abnormal bone remodeling related to dysregulation of osteoblasts and osteoclasts plays key roles in the OA initiation and development.82,83,84,85,86,87 Increased subchondral bone porosity and remodeling, reduced bone density, and bone mineralization with irregular matrix organization, which were believed to be stimulated by bone-cartilage crosstalk through subchondral pores and vascular invasion, were observed in the early stage of OA.88,89,90,91 These changes in subchondral bone were found to be happening at the same time with or earlier than the early destruction of articular cartilage.92,93,94,95 On the other hand, the late stage of OA showed architectural alterations of the subchondral bone characterized by a reduction of bone remodeling and enhanced subchondral bone densification leading to sclerosis.96,97,98 However, the mechanism of how articular cartilage and joint issues crosstalk with subchondral bone leading to the initiation and development of OA is incompletely understood and needs further investigation.
Besides the subchondral bone dysregulation, the synovium is another most-related tissue that showed significant changes at the early stage of OA, even before cartilage degradation occurs.91,99 The contribution of synovium to the initiation and development of OA has been investigated in the past 20 years. At the early stage of OA, histological changes of the synovium include synovial lining hypertrophy and hyperplasia, increased angiogenesis, a low level of synovial inflammation, and synovial fibrosis observed.100,101,102,103 Synovitis with a high level of macrophages could be found at the end stage of the OA.104 Synovitis scores used as one of the OA assessments are based on these histological features.99,105,106 Low-grade synovial inflammation can be detected in >50% of OA patients at the early and late stages of the disease.107,108 Therefore, among the synovial features, synovial inflammation has received the most attention from the OA research community. It is widely believed that pro-inflammatory factors that are released by synovial tissue induce the ECM destruction of the articular cartilage. However, the interactions of different cell types in the synovial joint and features of the synovium at different stages of OA need to be extensively investigated.
Obesity acts as an OA risk factor not solely through loading excessive body weight onto knee joints,109 the pathogenesis involves a complex network of tissue and cellular interactions. As mentioned above, adipokines released by adipose tissue that interact with different tissues are believed to be critically involved in OA pathogenesis.110,111,342 Meanwhile, the signaling crosstalk between the BMP signaling pathway and the Wnt/β-catenin signaling pathway plays an important role in regulating cartilage homeostasis.343 Loss of TGFβR1 in mouse growth plate cartilage increased basal BMP activity, suggesting that TGFβR1inhibits BMP signaling in the development of the growth plate.344 However, the role of TGFβR1 and TGFβR2 in the homeostasis of articular cartilage is still unknown. How the non-canonical BMP pathway regulates the homeostasis of the articular cartilage and contributes to the OA onset and development remains to be determined.
The BMP signaling pathway is abnormal and unstable in OA patients.345 The expression of BMP-2 and BMP-4 in chondrocytes of articular cartilage of OA patients was significantly upregulated in the early stage and increased with OA degree. BMP-4 promoted the pathological remodeling of the osteochondral junction.346 OA significantly promoted BMP2 and MMP-13 expression in the subchondral bone of the experimental OA rat model. This increase was inhibited by intra-articular injection of noggin protein (a BMP2 inhibitor). Meanwhile, Noggin protein dramatically attenuated OA disease progression in early OA rat model.347 Moderate exercises increased the expression of BMP signaling pathway-associated proteins, such as BMP2, BMP6 and BMP receptor 2, and pSmad-5, thus inhibiting cartilage degeneration and attenuating the OA phenotype.348
More and more studies focus on the potential therapeutic targets of OA. miR-181a is critical for crosstalk between the BMP and Wnt/β-catenin signaling pathways and may become a target for OA therapy.349 BMP7-derived peptides ameliorated OA chondrocyte phenotype in vitro and attenuated cartilage degeneration in vivo.350 The clinical trial phase II of the intra-articular injection of BMP-7 for the treatment of knee OA patients was completed (NCT01111045) in 2011. BMP receptor type I (BMPRI) mimetic peptide CK2.1 promoted articular cartilage repair and inhibited chondrocyte hypertrophy through intra-articular injection.351 Several studies showed that BMP-2 and BMP-4 effectively decreased cartilage degeneration in combination with tissue engineering materials. Recombinant human BMP-2 promoted cartilage regeneration in injured cartilage in vitro and was evaluated in clinical trials (NCT00243295). Tissue engineering materials, such as porous Hydroxyapatite Collagen (HAp/Col), Conically GRADED scaffold of chitosan-hax Hydrogel/Poly (L-Lactide-co-Glycolide) (PLGA), and POLY caprolactone (PCL) scaffolds, combined with BMP-2 treatment greatly promoted cartilage regeneration and repaired the joint cartilage.352,353,354 BMP-3 inhibited cartilage regeneration in both partially and completely defective rabbit joint injury models by inducing ECM degradation, inhibited the expression of BMP-2 and BMP-4, and interfered with chondrocyte survival on the articular surface.355 The intra-articular injection of BMP-4 combined with muscle-derived stem cells (MDSCs) could efficiently repair articular cartilage damage in the experimental OA rat model.356 Regular intra-articular injections of BMP7 alleviated the damage to articular cartilage caused by strenuous running.357 The use of BMP6 combined with BMP2 or TGFB3 induced enhanced chondrogenesis of stem cells in vitro.358,359 The combined use of an osteogenic nanoparticulate mineralized glycosaminoglycan scaffold (MC-GAG) and BMP-9 promoted chondrogenic differentiation of primary human mesenchymal stem cells (hMSCs) by inducing increased expression of collagen II, aggrecan and cartilage oligomeric protein.360
FGF signaling pathways
The fibroblast growth factor (FGF) family is present in a wide range of animal species, including nematodes, zebrafish, mice, and humans.361 FGF signaling is involved in a variety of physiological processes, such as cell proliferation, migration, and differentiation.362 There are 18 FGFs in mammals, and they are FGFs1-10 and FGFs16-23. FGFs19, 21, and 23 are hormone-like FGFs that work in an endocrine fashion, and the other FGF members work in a paracrine fashion. FGFs ligands fulfill their functions through binding and activating FGF receptor (FGFR).362 The mammalian FGFR family consists of four members, FGFR1, FGFR2, FGFR3, and FGFR4. FGF signaling involves multiple downstream signaling pathways, including the RAS/MAPK, PI3K/AKT, and PLCγ pathways. In addition, FGF signaling can activate the STAT1/p21 pathway.363
FGF signaling plays a critical role in bone and cartilage development.363 FGFs/FGFRs function at various stages of bone and cartilage development from limb bud formation to long bone growth and maturation.363 FGFR1 and FGFR3 are the predominantly expressed FGF receptors in cartilage. FGFR2 expression is restricted to the pre-cartilage condensate zone, and FGFR2 can serve as an early marker of chondrocytes.364 During growth plate formation, FGFR1 is expressed in pre-hypertrophic and hypertrophic regions, and FGFR2 is observed in quiescent regions.365 FGFR3 was detected in the center of the mesenchymal condensation and all growth plate chondrocytes.364,365 At the same time, FGFs 1, 2, 5, 8, 9, 16–19, 21, and 23 are expressed in the growth plate chondrocytes, and FGFs 1, 2, 6, 7, 9, 18, 21, and 22 are expressed in the perichondrium.366
Previous studies have shown that FGFs 1, 2, 7, 8, 9,18, and 23 are the major ones associated with the pathogenesis of OA (Table 2), but they have diverse functions in the development of the joint disorder.367 The secretion and expression of FGF1 were significantly increased in the synovium in the late stages of the OA. FGF1 was reported to suppress ECM synthesis of the human articular chondrocytes and inhibit the expression of CCN2 (cellular communication network factor 2), which is an important factor that promotes the regeneration of damaged cartilage.368 FGF2 was reported to bind to perlecan, a heparan sulfate proteoglycan in the ECM, and functioned as a mechanosensor in articular cartilage.369 FGF2 binds to FGFR1 to upregulate the expression of MMP1 and MMP13 promoting matrix degradation through neuro-endocrine pathways in adult articular chondrocytes.370,371 FGF8 expression is upregulated in damaged synovium in a rabbit model of OA, and FGF8 was shown to enhance the production of proteases and prostaglandin E2 in inflamed synovial cells, thereby promoting cartilage degradation.372 The expression of FGF9 was downregulated in the human OA cartilage.373 Treatment of exogenous FGF9 attenuated cartilage degeneration but exacerbated the osteophyte formation in a mouse OA model.373 The function of FGF9 in articular cartilage remains to be defined. FGF18 was highly expressed in the superficial zone of articular cartilage and stimulated the expression and accumulation of type II collagen in articular chondrocytes to protect articular cartilage against degeneration.374,375,376 In addition, the FGF signaling pathway also plays an important role in synovitis. The synthesis and secretion of FGF1 were significantly increased in the synovial fibroblast in OA.377 FGFR2, which is one of the cognate receptors of FGF1, was upregulated in the synovial membrane in OA patients.377 FGF2 is a potent agent to promote the proliferation and chondrogenesis of synovial-derived stem cells.378 The expression of FGF8 was significantly upregulated in hyperplastic synovial cells and fibroblasts in the rabbit OA model.372 Deletion of FGFR1 in adult mouse articular chondrocytes inhibited the progression of articular cartilage degeneration, which was associated with MMP13 downregulation and FGFR3 upregulation.379 Zhou S et al. showed that deletion of FGFR3 in articular chondrocytes in mice resulted in OA-like defects in the temporomandibular joint, which were associated with upregulation of RUNX2 and Indian hedgehog (IHH).380 This suggests that targeting FGF may have a potential strategy for OA treatment. The clinical trials phase II of the only FGF-targeting drug for OA, recombinant human FGF18 (sprifermin), was finished in 2020 (NCT01919164). Intra-articular administration of 100 μg Sprifermin every 6 or 12 months significantly increased the thickness of the femorotibial joint cartilage after two years of treatment with no marked side effects.381
Runx2
Runt-related transcription factor 2 (Runx2) is a runt domain-containing transcription factor that binds to DNA as a monomer or, with higher affinity, as a part of a heterodimeric complex.430,431 Prg4 global knockout mice develop aging-related joint disorders with loss of chondrocytes in the superficial zone of articular cartilage and synovial cell hyperplasia.432 Running and fluid flow shear stress could promote the expression of Prg4 in the superficial zone chondrocytes in vivo and in vitro,433 suggesting the regulation of Prg4 expression can be partially controlled by mechanical forces. As a protective factor for joints, Prg4 is expressed by embryonic joint progenitors. Prg4 positive articular chondrocytes located on the surface of joint cartilage in adult mice have been demonstrated to be the progenitor for deeper layers of the mature articular cartilage.128,434 Exogenous recombinant human (rh) PRG4E was reported to promote ear wound closure and tissue regeneration by increasing VEGF expression and blood flow through a TLR (Toll-like receptor)-dependent mechanism in mice.435 Full-length recombinant human PRG4 (rhPRG4) produced by CHO-M cells and native human PRG4 (nhPRG4) purified from culture supernatants of human fibroblast-like synoviocytes from OA patients were also shown to bind to TLR2 and TLR4, mediating an anti-inflammatory factor.436 However, whether overexpressing Prg4 in the synovial joint can facilitate articular cartilage regeneration or prevent OA development remains to be determined. Lubricin was included as a potential biomarker in human synovial fluid in the diagnosis and progression of OA patients by a clinical observational study in 2022 (NCT02664870).
Other signaling factors
There are several other signaling cascades that have essential roles in the OA onset and development, such as notch signaling. Notch signaling was upregulated in mouse and human OA cartilage.437 Notch2 gain of function mutation in articular cartilage increased the severity of post-trauma OA by crosstalk with NF-κB, Wnt, and TGFβ signaling.438,439 Transient overexpression of NICD (NOTCH1 intracellular domain) led to enhanced synthesis of ECM and promoted the maintenance of articular cartilage, while constitutional overexpression of NICD resulted in early and progressive OA lesions in mice.437 Activation of Hes1, an essential mediator of Notch signaling, suppressed articular cartilage degradation and OA development by decreasing the Adamts5 and Mmp13 expression.440 The endoplasmic reticulum (ER) stress-triggered unfolded protein response (UPR) signaling has been identified as a contributing factor to OA pathology. Older OA patients developed ER stress in the early-stage OA when there was a higher synthesis of ECM proteins.441 How the UPR signaling mediated cell survival and the chronic ER stress-initiated apoptosis in cartilage and synovium contribute to the onset and development of OA needs to be investigated.
Clinical therapy and clinical trials
So far there is no effective cure for OA. The OA treatment approaches are divided into physical modalities, pharmacologic treatments, and surgical treatments.442 Several new therapies have also recently been developed. In the early stages of OA, treatment focuses on reducing pain and joint stiffness. Subsequently, treatment mainly focuses on maintaining joint physical function.443,444 In summary, OA treatment aims to reduce the disease symptoms and slow its progression.
Non-pharmacological treatment
Weight loss
Excessive body weight or obesity is a major risk factor for OA.445 Greater body weight adversely affects joint structure by adding additional load to the joints during daily activities and causing increases in the expression and production of enzymes that degrade the joints or increase joint inflammation.446 Weight loss is recommended for overweight or obese patients with low-extremity osteoarthritis.447
Exercise
According to the recommendations from the International Association for the Study of OA (OARSI), exercise is considered a core approach to the treatment of OA and is recommended for all patients.448 Exercise has been extensively studied as a treatment for OA.448 Uthman and colleagues found that exercise reduced painful movements and improved physical function in OA patients.449 The most common exercises used to treat OA include aquatic exercise,450 aerobic exercise,451 resistance exercise,451 multimodal exercise,452 and combination exercise.452,453
Assistive devices
OA patients often need assistive devices to compensate for decreased strength and impaired pain during exercise.454 Common devices include splints, braces, canes, functional shoes, and other training equipment.455 While there are some positive results from clinical studies, the need for assistive devices and their long-term safety remains in doubt.456
Physical therapy
Physiotherapy has significant therapeutic effects on OA, including therapeutic ultrasound, electrical stimulation, phototherapy, hydrotherapy, magnetotherapy, cryotherapy, and thermotherapy.457,458,459,460,461 Physical therapy provides significant relief of symptoms of OA, including pain, edema, and joint motion disturbances, and is suitable for emergency management in the acute phase.460 Instructing patients to use thermal agents has been recommended as a self-management strategy by the recent American College of Rheumatology Clinical Guidelines.462
Acupuncture
Acupuncture is a non-pharmacological treatment method in Chinese medicine.463,464 Acupuncture has analgesic and functional restorative effects in treating OA.463 The therapeutic effect of acupuncture may come from modulating inflammatory factors.463,465 However, there is evidence of uncertainty in the treatment of OA with acupuncture, particularly a significant difference between electro-acupuncture and hand acupuncture.466 In addition, non-pharmacological strategies, including health education, and lifestyle changes, such as diet, postural correction, and self-management, are important measures to prevent OA.467,468
Pharmacologic treatment
Currently, no drugs can alter the progression of OA and prevent long-term disability.469 Current international guidelines recommend medications for the treatment of OA that revolve solely around reducing the burden of the disease (symptomatic effects) and altering the natural course of the disease by slowing or stop** the biological process of tissue damage.469,470 The following classes of drugs are currently used to treat OA of the knee: non-steroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, opioids, symptomatic, chondroprotective agents, and anti-cytokines.470,471,472
Paracetamol (acetaminophen) is the first-line analgesic for clinical pain control of arthritis.470 The safety profile of paracetamol relative to other analgesics, such as non-steroidal anti-inflammatory drugs (NSAIDs), has led to its increased use, resulting in paracetamol becoming one of the most common drugs used in OA treatment.470 However, there is evidence that paracetamol is associated with an increased risk of gastrointestinal, cardiovascular, and renal disease, as well as mortality.473 NSAIDs are commonly used anti-inflammatory and analgesic drugs in the treatment of OA.529
Artificial joint replacement
Arthroplasty is currently an effective clinical treatment for advanced knee OA. It effectively eliminates pain, corrects joint deformity, and improves knee function.530 However, clinical studies have found that some patients still do not recover satisfactorily after surgery and cannot fully straighten the knee.531 Cartilage injury, ligament injury, postoperative infection, and postoperative deep vein thrombosis in the lower extremity are all factors that influence the outcome of surgical treatment of knee OA, and early postoperative prevention of complications is crucial.531,532
Conclusions and perspectives
OA is a tremendously complex synovial whole-joint disorder, and how OA is initiated and developed remains poorly understood. Research on the cellular and molecular mechanisms of OA is still in the beginning phase. We have summarized from the current knowledge changes of key molecules in the essential signaling pathways in the articular cartilage, synovial membrane, subchondral bone, and synovial fluids of OA patients and animal models (Table 3), as well as the potential therapeutic reagents that have been reported (Table 4). Wnt, TNF, TGFβ/BMP, FGF pathway receptors, FA proteins, and other factors that are located on the chondrocyte membrane sense and transduce biochemical and mechanical signals. Activated signaling pathways and regulators, such as AMPK, mTOR, FGF, BMP, HIFs, and NF-κB, via crosstalk and feedback mechanisms in a complicated network, regulate the expression of key downstream factors, such as Runx2, MMP13, ADAMTS4/5, Prg4, and other factors, in articular chondrocytes and synovium. The initiation factors of OA are various, including excessive mechanical loading, inflammatory factors, aging and etc., which lead to the different primary effects of early-stage OA with unique molecular signaling signatures. The destruction and erosion of the articular cartilage, the synovial hyperplasia and inflammation, the abnormal angiogenesis of the synovial joint, the subchondral bone disturbance, and the instability of the ligaments and tendons could all contribute together or respectfully to the onset and progression of the disease. Scientists and clinic doctors are still debating, and there is still no conclusion on which one happens earlier than the others during the initiation of OA. No matter which pathological factor is the first dominant over others, or they contribute equally to the progression of OA; it has been well accepted that interference at the early stage of OA to prevent its progression will be a more efficient and effective strategy for better outcomes than focusing on the medical treatments and joint replacement surgery at the late and end-stage of the disease. Therefore, targeting critical signaling pathways and key molecules that are significantly changed at the early stage of the disease to control crosstalk between molecular pathways and the interaction of different compartments of synovial joint is critical for future research.
The multiple layers of complexity of the pathogenesis of OA and limited knowledge of the pathogenetic molecular signaling pathways and specific mechanisms have made the therapeutic pharmacological targeting of OA extremely difficult. At this point, in the future, a comprehensive understanding of alterations in distinct signaling pathways and expression of key factors in the superficial, middle and deep zones of articular cartilage and synovial membrane at different stages of OA triggered by different factors should be investigated in great detail. The whole picture of functions and regulations of each pathological signaling and key factors in various early-stage OA conditions could help us to develop more specific resolutions to halt or reverse the OA disease.
References
Hunter, D. J. & Bierma-Zeinstra, S. Osteoarthritis. Lancet 393, 1745–1759 (2019).
Quicke, J. G., Conaghan, P. G., Corp, N. & Peat, G. Osteoarthritis year in review 2021: epidemiology & therapy. Osteoarthr. Cartil. 30, 196–206 (2022).
Little, C. B. & Hunter, D. J. Post-traumatic osteoarthritis: from mouse models to clinical trials. Nat. Rev. Rheumatol. 9, 485–497 (2013).
Leung, Y. Y. & Thumboo, J. An overview of OA research in two urban APLAR populations. Int. J. Rheum. Dis. 14, 130–135 (2011).
Lu, K. et al. Molecular signaling in temporomandibular joint osteoarthritis. J. Orthop. Transl. 32, 21–27 (2022).
Prieto-Alhambra, D. et al. Incidence and risk factors for clinically diagnosed knee, hip and hand osteoarthritis: influences of age, gender and osteoarthritis affecting other joints. Ann. Rheum. Dis. 73, 1659–1664 (2014).
De Luna, A., Otahal, A. & Nehrer, S. Mesenchymal stromal cell-derived extracellular vesicles - silver linings for cartilage regeneration? Front. Cell Dev. Biol. 8, 593386 (2020).
Nedunchezhiyan, U. et al. Obesity, inflammation, and immune system in osteoarthritis. Front. Immunol. 13, 907750 (2022).
Wallace, I. J. et al. Knee osteoarthritis has doubled in prevalence since the mid-20th century. Proc. Natl Acad. Sci. USA 114, 9332–9336 (2017).
Turkiewicz, A. et al. Current and future impact of osteoarthritis on health care: a population-based study with projections to year 2032. Osteoarthr. Cartil. 22, 1826–1832 (2014).
Pereira, D. et al. The effect of osteoarthritis definition on prevalence and incidence estimates: a systematic review. Osteoarthr. Cartil. 19, 1270–1285 (2011).
Li, Y., **e, W., **ao, W. & Dou, D. Progress in osteoarthritis research by the national natural science foundation of China. Bone Res 10, 41 (2022).
Safiri, S. et al. Global, regional and national burden of osteoarthritis 1990–2017: a systematic analysis of the Global Burden of Disease Study 2017. Ann. Rheum. Dis. 79, 819–828 (2020).
Long, H. et al. Prevalence trends of site-specific osteoarthritis from 1990 to 2019: findings from the global burden of disease study 2019. Arthritis Rheumatol. 74, 1172–1183 (2022).
Arslan, I. G. et al. Estimating incidence and prevalence of hip osteoarthritis using electronic health records: a population-based cohort study. Osteoarthr. Cartil. 30, 843–851 (2022).
**, Z. Y. et al. Incidence trend of five common musculoskeletal disorders from 1990 to 2017 at the global, regional and national level: results from the global burden of disease study 2017. Ann. Rheum. Dis. 79, 1014–1022 (2020).
Disease, G. B. D., Injury, I. & Prevalence, C. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392, 1789–1858 (2018).
Abbafati, C. et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1204–1222 (2020).
Swain, S. et al. Trends in incidence and prevalence of osteoarthritis in the United Kingdom: findings from the Clinical Practice Research Datalink (CPRD). Osteoarthr. Cartil. 28, 792–801 (2020).
Tie, X.-J. et al. Prevalence of knee osteoarthritis in the middle-aged and elderly in China: </br> a Meta-analysis. Chin. J. Tissue Eng. Res. 22, 650–656 (2018).
Tang, X. et al. The prevalence of symptomatic knee osteoarthritis in china: results from the China health and retirement longitudinal study. Arthritis Rheumatol. 68, 648–653 (2016).
Kang, X. et al. The high prevalence of knee osteoarthritis in a rural Chinese population: the Wuchuan osteoarthritis study. Arthritis Rheum. 61, 641–647 (2009).
Sun, X. et al. Osteoarthritis in the middle-aged and elderly in china: prevalence and influencing factors. Int. J. Environ. Res. Public Health 16, 4701 (2019).
Zhang, Y. et al. Comparison of the prevalence of knee osteoarthritis between the elderly Chinese population in Bei**g and whites in the United States: The Bei**g Osteoarthritis Study. Arthritis Rheum. 44, 2065–2071 (2001).
Nevitt, M. C. et al. Very low prevalence of hip osteoarthritis among Chinese elderly in Bei**g, China, compared with whites in the United States: the Bei**g osteoarthritis study. Arthritis Rheum. 46, 1773–1779 (2002).
Zhang, Y. et al. Lower prevalence of hand osteoarthritis among Chinese subjects in Bei**g compared with white subjects in the United States: the Bei**g Osteoarthritis Study. Arthritis Rheum. 48, 1034–1040 (2003).
Chen, H. et al. Trends and patterns of knee osteoarthritis in China: a longitudinal study of 17.7 million adults from 2008 to 2017. Int. J. Environ. Res. Public Health 18, 8864 (2021).
Yu, D., Peat, G., Bedson, J. & Jordan, K. P. Annual consultation incidence of osteoarthritis estimated from population-based health care data in England. Rheumatology 54, 2051–2060 (2015).
Oliveria, S. A., Felson, D. T., Reed, J. I., Cirillo, P. A. & Walker, A. M. Incidence of symptomatic hand, hip, and knee osteoarthritis among patients in a health maintenance organization. Arthritis Rheum. 38, 1134–1141 (1995).
Johnson, V. L. & Hunter, D. J. The epidemiology of osteoarthritis. Best. Pract. Res Clin. Rheumatol. 28, 5–15 (2014).
Liu, Q. et al. Knee symptomatic osteoarthritis, walking disability, nsaids use and all-cause mortality: population-based wuchuan osteoarthritis study. Sci. Rep. 7, 3309 (2017).
Cross, M. et al. The global burden of hip and knee osteoarthritis: estimates from the Global Burden of Disease 2010 study. Ann. Rheum. Dis. 73, 1323–1330 (2014).
Lo, J., Chan, L. & Flynn, S. A systematic review of the incidence, prevalence, costs, and activity and work limitations of amputation, osteoarthritis, rheumatoid arthritis, back pain, multiple sclerosis, spinal cord injury, stroke, and traumatic brain injury in the united states: a 2019 update. Arch. Phys. Med. Rehab. 102, 115–131 (2021).
Wang, S. X. et al. Healthcare resource utilization and costs by age and joint location among osteoarthritis patients in a privately insured population. J. Med Econ. 20, 1299–1306 (2017).
Kiadaliri, A. A., Lohmander, L. S., Moradi-Lakeh, M., Petersson, I. F. & Englund, M. High and rising burden of hip and knee osteoarthritis in the Nordic region, 1990–2015: findings from the Global Burden of Disease Study 2015. Acta Orthop. 89, 177–183 (2018).
Long, H. et al. Burden of osteoarthritis in China, 1990–2017: findings from the Global Burden of Disease Study 2017. Lancet Rheumatol. 2, e164–e172 (2020).
Kassebaum, N. J. et al. Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388, 1603–1658 (2016).
Liu, Q., Wang, S., Lin, J. & Zhang, Y. The burden for knee osteoarthritis among Chinese elderly: estimates from a nationally representative study. Osteoarthr. Cartil. 26, 1636–1642 (2018).
O’Brien, M. S. & McDougall, J. J. Age and frailty as risk factors for the development of osteoarthritis. Mech. Ageing Dev. 180, 21–28 (2019).
Sacitharan, P. K. Ageing and Osteoarthritis. Subcell. Biochem 91, 123–159 (2019).
Loeser, R. F., Collins, J. A. & Diekman, B. O. Ageing and the pathogenesis of osteoarthritis. Nat. Rev. Rheumatol. 12, 412–420 (2016).
Coryell, P. R., Diekman, B. O. & Loeser, R. F. Mechanisms and therapeutic implications of cellular senescence in osteoarthritis. Nat. Rev. Rheumatol. 17, 47–57 (2021).
Coppe, J. P. et al. Tumor suppressor and aging biomarker p16(INK4a) induces cellular senescence without the associated inflammatory secretory phenotype. J. Biol. Chem. 286, 36396–36403 (2011).
Freund, A., Patil, C. K. & Campisi, J. p38MAPK is a novel DNA damage response-independent regulator of the senescence-associated secretory phenotype. EMBO J. 30, 1536–1548 (2011).
Tsuchida, A. I. et al. Cytokine profiles in the joint depend on pathology, but are different between synovial fluid, cartilage tissue and cultured chondrocytes. Arthritis Res. Ther. 16, 441 (2014).
Hui, W. et al. Oxidative changes and signalling pathways are pivotal in initiating age-related changes in articular cartilage. Ann. Rheum. Dis. 75, 449–458 (2016).
Blanco, F. J., Rego, I. & Ruiz-Romero, C. The role of mitochondria in osteoarthritis. Nat. Rev. Rheumatol. 7, 161–169 (2011).
Scott, J. L. et al. Superoxide dismutase downregulation in osteoarthritis progression and end-stage disease. Ann. Rheum. Dis. 69, 1502–1510 (2010).
Greene, M. A. & Loeser, R. F. Aging-related inflammation in osteoarthritis. Osteoarthr. Cartil. 23, 1966–1971 (2015).
Raud, B. et al. Level of obesity is directly associated with the clinical and functional consequences of knee osteoarthritis. Sci. Rep. 10, 3601 (2020).
Reyes, C. et al. Association between overweight and obesity and risk of clinically diagnosed knee, hip, and hand osteoarthritis: a population-based cohort study. Arthritis Rheumatol. 68, 1869–1875 (2016).
Conde, J. et al. Identification of novel adipokines in the joint. Differential expression in healthy and osteoarthritis tissues. PLoS ONE 10, e0123601 (2015).
Jiang, H. et al. Adiponectin, may be a potential protective factor for obesity-related osteoarthritis. Diabetes Metab. Syndr. Obes. 15, 1305–1319 (2022).
Huang, M. J. et al. Enhancement of the synthesis of n-3 PUFAs in fat-1 transgenic mice inhibits mTORC1 signalling and delays surgically induced osteoarthritis in comparison with wild-type mice. Ann. Rheum. Dis. 73, 1719–1727 (2014).
Sekar, S. et al. Saturated fatty acids promote chondrocyte matrix remodeling through reprogramming of autophagy pathways. Nutrition 54, 144–152 (2018).
Dai, Y. et al. A low proportion n-6/n-3 PUFA diet supplemented with Antarctic krill (Euphausia superba) oil protects against osteoarthritis by attenuating inflammation in ovariectomized mice. Food Funct. 12, 6766–6779 (2021).
Peshkova, M. et al. Gender-related aspects in osteoarthritis development and progression: a review. Int. J. Mol. Sci. 23, 2767 (2022).
Price, M. D. & Herndon, J. H. Gender differences in osteoarthritis. Menopause 16, 624–625 (2009).
Yan, Y. S. et al. Sex steroids and osteoarthritis: a mendelian randomization study. Front. Endocrinol. 12, 683226 (2021).
Tschon, M., Contartese, D., Pagani, S., Borsari, V. & Fini, M. Gender and sex are key determinants in osteoarthritis not only confounding variables. a systematic review of clinical data. J. Clin. Med. 10, 3178 (2021).
Laitner, M. H., Erickson, L. C. & Ortman, E. Understanding the impact of sex and gender in osteoarthritis: assessing research gaps and unmet needs. J. Women’s Health (Larchmt.) 30, 634–641 (2021).
Blaker, C. L., Clarke, E. C. & Little, C. B. Using mouse models to investigate the pathophysiology, treatment, and prevention of post-traumatic osteoarthritis. J. Orthop. Res. 35, 424–439 (2017).
Papalia, R. et al. Sport activity as risk factor for early knee osteoarthritis. J. Biol. Regulators Homeost. Agents 33, 29–37 (2019). XIX Congresso Nazionale S I C O O P Societa’ Italiana Chirurghi Ortopedici Dell’ospedalita’ Privata Accreditata.
Astephen Wilson, J. L. & Kobsar, D. Osteoarthritis year in review 2020: mechanics. Osteoarthr. Cartil. 29, 161–169 (2021).
Andersson, J. K., Hagert, E. & Brittberg, M. Cartilage injuries and posttraumatic osteoarthritis in the wrist: a review. Cartilage 13, 156S–168S (2021).
Harlaar, J., Macri, E. M. & Wesseling, M. Osteoarthritis year in review 2021: mechanics. Osteoarthr. Cartil. 30, 663–670 (2022).
Boer, C. G. et al. Deciphering osteoarthritis genetics across 826,690 individuals from 9 populations. Cell 184, 4784–4818 e4717 (2021).
Ratneswaran, A. & Kapoor, M. Osteoarthritis year in review: genetics, genomics, epigenetics. Osteoarthr. Cartil. 29, 151–160 (2021).
Aubourg, G., Rice, S. J., Bruce-Wootton, P. & Loughlin, J. Genetics of osteoarthritis. Osteoarthr. Cartil. 30, 636–649 (2022).
Loughlin, J. Translating osteoarthritis genetics research: challenging times ahead. Trends Mol. Med. 28, 176–182 (2022).
Young, D. A., Barter, M. J. & Soul, J. Osteoarthritis year in review: genetics, genomics, epigenetics. Osteoarthr. Cartil. 30, 216–225 (2022).
Fayet, M. & Hagen, M. Pain characteristics and biomarkers in treatment approaches for osteoarthritis pain. Pain. Manag. 11, 59–73 (2021).
Katz, J. N., Arant, K. R. & Loeser, R. F. Diagnosis and treatment of hip and knee osteoarthritis: a review. JAMA 325, 568–578 (2021).
Seifert, O. & Baerwald, C. Interaction of pain and chronic inflammation. Z. Rheumatol. 80, 205–213 (2021).
Baral, P., Udit, S. & Chiu, I. M. Pain and immunity: implications for host defence. Nat. Rev. Immunol. 19, 433–447 (2019).
Wood, M. J., Miller, R. E. & Malfait, A. M. The genesis of pain in osteoarthritis: inflammation as a mediator of osteoarthritis pain. Clin. Geriatr. Med. 38, 221–238 (2022).
Conaghan, P. G., Cook, A. D., Hamilton, J. A. & Tak, P. P. Therapeutic options for targeting inflammatory osteoarthritis pain. Nat. Rev. Rheumatol. 15, 355–363 (2019).
Vincent, T. L. Peripheral pain mechanisms in osteoarthritis. Pain 161, S138–S146 (2020).
Yu, H., Huang, T., Lu, W. W., Tong, L. & Chen, D. Osteoarthritis pain. Int. J. Mol. Sci. 23, 4642 (2022).
Malfait, A. M., Miller, R. E. & Miller, R. J. Basic mechanisms of pain in osteoarthritis: experimental observations and new perspectives. Rheum. Dis. Clin. North Am. 47, 165–180 (2021).
Tong, L. et al. Current understanding of osteoarthritis pathogenesis and relevant new approaches. Bone Res 10, 60 (2022).
Burr, D. B. & Gallant, M. A. Bone remodelling in osteoarthritis. Nat. Rev. Rheumatol. 8, 665–673 (2012).
Funck-Brentano, T. & Cohen-Solal, M. Subchondral bone and osteoarthritis. Curr. Opin. Rheumatol. 27, 420–426 (2015).
Maruotti, N., Corrado, A. & Cantatore, F. P. Osteoblast role in osteoarthritis pathogenesis. J. Cell. Physiol. 232, 2957–2963 (2017).
Hu, W., Chen, Y., Dou, C. & Dong, S. Microenvironment in subchondral bone: predominant regulator for the treatment of osteoarthritis. Ann. Rheum. Dis. 80, 413–422 (2021).
Han, X. et al. Abnormal subchondral trabecular bone remodeling in knee osteoarthritis under the influence of knee alignment. Osteoarthr. Cartil. 30, 100–109 (2022).
Zhang, L. et al. Expansion of myeloid-derived suppressor cells contributes to metabolic osteoarthritis through subchondral bone remodeling. Arthritis Res. Ther. 23, 287 (2021).
Amir, G., Pirie, C. J., Rashad, S. & Revell, P. A. Remodelling of subchondral bone in osteoarthritis: a histomorphometric study. J. Clin. Pathol. 45, 990–992 (1992).
Benske, J., Schunke, M. & Tillmann, B. Subchondral bone formation in arthrosis. Polychrome labeling studies in mice. Acta Orthop. Scand. 59, 536–541 (1988).
Hu, Y., Chen, X., Wang, S., **g, Y. & Su, J. Subchondral bone microenvironment in osteoarthritis and pain. Bone Res. 9, 20 (2021).
Hugle, T. & Geurts, J. What drives osteoarthritis?-synovial versus subchondral bone pathology. Rheumatology 56, 1461–1471 (2017).
Dieppe, P., Cushnaghan, J., Young, P. & Kirwan, J. Prediction of the progression of joint space narrowing in osteoarthritis of the knee by bone scintigraphy. Ann. Rheum. Dis. 52, 557–563 (1993).
Mansell, J. P., Tarlton, J. F. & Bailey, A. J. Biochemical evidence for altered subchondral bone collagen metabolism in osteoarthritis of the hip. Br. J. Rheumatol. 36, 16–19 (1997).
Bolbos, R. I. et al. Relationship between trabecular bone structure and articular cartilage morphology and relaxation times in early OA of the knee joint using parallel MRI at 3 T. Osteoarthr. Cartil. 16, 1150–1159 (2008).
Ding, R., Zhang, N., Wang, Q. & Wang, W. Alterations of the subchondral bone in osteoarthritis: complying with Wolff’s law. Curr. Rheumatol. Rev. 18, 178–185 (2022).
Fazzalari, N. L. & Parkinson, I. H. Fractal properties of subchondral cancellous bone in severe osteoarthritis of the hip. J. Bone Miner. Res. 12, 632–640 (1997).
Arden, N. K., Griffiths, G. O., Hart, D. J., Doyle, D. V. & Spector, T. D. The association between osteoarthritis and osteoporotic fracture: the Chingford Study. Br. J. Rheumatol. 35, 1299–1304 (1996).
Hannan, M. T., Anderson, J. J., Zhang, Y., Levy, D. & Felson, D. T. Bone mineral density and knee osteoarthritis in elderly men and women. The Framingham Study. Arthritis Rheum. 36, 1671–1680 (1993).
Mathiessen, A. & Conaghan, P. G. Synovitis in osteoarthritis: current understanding with therapeutic implications. Arthritis Res. Ther. 19, 18 (2017).
Smith, M. D., Triantafillou, S., Parker, A., Youssef, P. P. & Coleman, M. Synovial membrane inflammation and cytokine production in patients with early osteoarthritis. J. Rheumatol. 24, 365–371 (1997).
Goldring, M. B. & Otero, M. Inflammation in osteoarthritis. Curr. Opin. Rheumatol. 23, 471–478 (2011).
Pelletier, J. P., Martel-Pelletier, J. & Abramson, S. B. Osteoarthritis, an inflammatory disease: potential implication for the selection of new therapeutic targets. Arthritis Rheum. 44, 1237–1247 (2001).
Maglaviceanu, A., Wu, B. & Kapoor, M. Fibroblast-like synoviocytes: Role in synovial fibrosis associated with osteoarthritis. Wound Repair Regen. 29, 642–649 (2021).
Thomson, A. & Hilkens, C. M. U. Synovial macrophages in osteoarthritis: the key to understanding pathogenesis? Front. Immunol. 12, 678757 (2021).
Krenn, V. et al. Grading of chronic synovitis-a histopathological grading system for molecular and diagnostic pathology. Pathol. Res Pract. 198, 317–325 (2002).
Krenn, V. et al. 15 years of the histopathological synovitis score, further development and review: a diagnostic score for rheumatology and orthopaedics. Pathol. Res. Pract. 213, 874–881 (2017).
Bonnet, C. S. & Walsh, D. A. Osteoarthritis, angiogenesis and inflammation. Rheumatology 44, 7–16 (2005).
Benito, M. J., Veale, D. J., FitzGerald, O., van den Berg, W. B. & Bresnihan, B. Synovial tissue inflammation in early and late osteoarthritis. Ann. Rheum. Dis. 64, 1263–1267 (2005).
Fantuzzi, G. Adipose tissue, adipokines, and inflammation. J. Allergy Clin. Immunol. 115, 911–919 (2005).
Zapata-Linares, N., Eymard, F., Berenbaum, F. & Houard, X. Role of adipose tissues in osteoarthritis. Curr. Opin. Rheumatol. 33, 84–93 (2021).
Belluzzi, E. et al. Systemic and local adipose tissue in knee osteoarthritis. J. Cell. Physiol. 232, 1971–1978 (2017).
**e, C. & Chen, Q. Adipokines: new therapeutic target for osteoarthritis? Curr. Rheumatol. Rep. 21, 71 (2019).
Chang, J. et al. Systemic and local adipose tissue in knee osteoarthritis. Osteoarthr. Cartil. 26, 864–871 (2018).
Collins, K. H. et al. Adipose tissue is a critical regulator of osteoarthritis. Proc. Natl Acad. Sci. USA 118, e2021096118 (2021).
Frazier, T. et al. Non-homologous use of adipose-derived cell and tissue therapies: osteoarthritis as a case study. Bone Rep. 17, 101601 (2022).
Sandell, L. J. & Aigner, T. Articular cartilage and changes in arthritis. An introduction: cell biology of osteoarthritis. Arthritis Res. 3, 107–113 (2001).
Hwang, H. S. & Kim, H. A. Chondrocyte apoptosis in the pathogenesis of osteoarthritis. Int. J. Mol. Sci. 16, 26035–26054 (2015).
Janczi, T. et al. ADAM15 in apoptosis resistance of synovial fibroblasts: converting Fas/CD95 death signals into the activation of prosurvival pathways by calmodulin recruitment. Arthritis Rheumatol. 71, 63–72 (2019).
Li, Q. et al. Effects of TNFR1 gene silencing on early apoptosis of marbofloxacin-treated chondrocytes from juvenile dogs. Toxicology 422, 53–59 (2019).
Del Carlo, M. Jr. & Loeser, R. F. Nitric oxide-mediated chondrocyte cell death requires the generation of additional reactive oxygen species. Arthritis Rheum. 46, 394–403 (2002).
Wang, G. et al. Cathelicidin antimicrobial peptide (CAMP) gene promoter methylation induces chondrocyte apoptosis. Hum. Genomics 15, 24 (2021).
Zhang, J., Hao, X., Chi, R., Qi, J. & Xu, T. Moderate mechanical stress suppresses the IL-1beta-induced chondrocyte apoptosis by regulating mitochondrial dynamics. J. Cell Physiol. 236, 7504–7515 (2021).
Sun, Y. et al. G protein coupled estrogen receptor attenuates mechanical stress-mediated apoptosis of chondrocyte in osteoarthritis via suppression of Piezo1. Mol. Med. 27, 96 (2021).
Castrogiovanni, P., Ravalli, S. & Musumeci, G. Apoptosis and autophagy in the pathogenesis of osteoarthritis. J. Invest. Surg. 33, 874–875 (2020).
Yang, J. et al. Targeting cell death: pyroptosis, ferroptosis, apoptosis and necroptosis in osteoarthritis. Front Cell Dev. Biol. 9, 789948 (2021).
Hu, J. et al. Loganin ameliorates cartilage degeneration and osteoarthritis development in an osteoarthritis mouse model through inhibition of NF-kappaB activity and pyroptosis in chondrocytes. J. Ethnopharmacol. 247, 112261 (2020).
Ulrich-Vinther, M., Maloney, M. D., Schwarz, E. M., Rosier, R. & O’Keefe, R. J. Articular cartilage biology. J. Am. Acad. Orthop. Surg. 11, 421–430 (2003).
Kozhemyakina, E. et al. Identification of a Prg4-expressing articular cartilage progenitor cell population in mice. Arthritis Rheumatol. 67, 1261–1273 (2015).
Sun, M. M. & Beier, F. Chondrocyte hypertrophy in skeletal development, growth, and disease. Birth Defects Res. C. Embryo Today 102, 74–82 (2014).
Chen, D., Kim, D. J., Shen, J., Zou, Z. & O’Keefe, R. J. Runx2 plays a central role in osteoarthritis development. J. Orthop. Transl. 23, 132–139 (2020).
Huang, J. et al. The microRNAs miR-204 and miR-211 maintain joint homeostasis and protect against osteoarthritis progression. Nat. Commun. 10, 2876 (2019).
Mobasheri, A. et al. The role of metabolism in the pathogenesis of osteoarthritis. Nat. Rev. Rheumatol. 13, 302–311 (2017).
Zheng, L., Zhang, Z., Sheng, P. & Mobasheri, A. The role of metabolism in chondrocyte dysfunction and the progression of osteoarthritis. Ageing Res. Rev. 66, 101249 (2021).
Nusse, R. & Clevers, H. Wnt/beta-catenin signaling, disease, and emerging therapeutic modalities. Cell 169, 985–999 (2017).
Kadowaki, T., Wilder, E., Klingensmith, J., Zachary, K. & Perrimon, N. The segment polarity gene porcupine encodes a putative multitransmembrane protein involved in Wingless processing. Genes Dev. 10, 3116–3128 (1996).
Reya, T. & Clevers, H. Wnt signalling in stem cells and cancer. Nature 434, 843–850 (2005).
Valenta, T., Hausmann, G. & Basler, K. The many faces and functions of β-catenin. EMBO J. 31, 2714–2736 (2012).
Schunk, S. J., Floege, J., Fliser, D. & Speer, T. WNT-β-catenin signalling - a versatile player in kidney injury and repair. Nat. Rev. Nephrol. 17, 172–184 (2021).
Alok, A. et al. Wnt proteins synergize to activate β-catenin signaling. J. Cell Sci. 130, 1532–1544 (2017).
MacDonald, B. T., Tamai, K. & He, X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev. Cell 17, 9–26 (2009).
Nusse, R. & Clevers, H. Wnt/β-catenin signaling, disease, and emerging therapeutic modalities. Cell 169, 985–999 (2017).
Zhu, M. et al. Inhibition of beta-catenin signaling in articular chondrocytes results in articular cartilage destruction. Arthritis Rheum. 58, 2053–2064 (2008).
Nalesso, G. et al. WNT16 antagonises excessive canonical WNT activation and protects cartilage in osteoarthritis. Ann. Rheum. Dis. 76, 218–226 (2017).
Tong, W. et al. Wnt16 attenuates osteoarthritis progression through a PCP/JNK-mTORC1-PTHrP cascade. Ann. Rheum. Dis. 78, 551–561 (2019).
Guo, X. et al. Wnt/beta-catenin signaling is sufficient and necessary for synovial joint formation. Genes Dev. 18, 2404–2417 (2004).
Loughlin, J. et al. Functional variants within the secreted frizzled-related protein 3 gene are associated with hip osteoarthritis in females. Proc. Natl Acad. Sci. USA 101, 9757–9762 (2004).
Lories, R. J. et al. Articular cartilage and biomechanical properties of the long bones in Frzb-knockout mice. Arthritis Rheum. 56, 4095–4103 (2007).
Akiyama, H. et al. Interactions between Sox9 and beta-catenin control chondrocyte differentiation. Genes Dev. 18, 1072–1087 (2004).
Harada, N. et al. Intestinal polyposis in mice with a dominant stable mutation of the beta-catenin gene. EMBO J. 18, 5931–5942 (1999).
Wu, Q. et al. Beta-catenin, cartilage, and osteoarthritis. Ann. N. Y. Acad. Sci. 1192, 344–350 (2010).
Zhu, M. et al. Activation of beta-catenin signaling in articular chondrocytes leads to osteoarthritis-like phenotype in adult beta-catenin conditional activation mice. J. Bone Miner. Res. 24, 12–21 (2009).
Wang, M. et al. Activation of beta-catenin signalling leads to temporomandibular joint defects. Eur. Cells Mater. 28, 223–235 (2014).
Zhou, Y., Wang, T., Hamilton, J. L. & Chen, D. Wnt/beta-catenin signaling in osteoarthritis and in other forms of arthritis. Curr. Rheumatol. Rep. 19, 53 (2017).
**a, C. et al. Activation of beta-catenin in Col2-expressing chondrocytes leads to osteoarthritis-like defects in hip joint. J. Cell. Physiol. 234, 18535–18543 (2019).
Li, W., **ong, Y., Chen, W. & Wu, L. Wnt/beta-catenin signaling may induce senescence of chondrocytes in osteoarthritis. Exp. Ther. Med 20, 2631–2638 (2020).
Yuasa, T. et al. Transient activation of Wnt/{beta}-catenin signaling induces abnormal growth plate closure and articular cartilage thickening in postnatal mice. Am. J. Pathol. 175, 1993–2003 (2009).
Yasuhara, R. et al. Roles of beta-catenin signaling in phenotypic expression and proliferation of articular cartilage superficial zone cells. Lab. Investig. 91, 1739–1752 (2011).
Blom, A. B. et al. Involvement of the Wnt signaling pathway in experimental and human osteoarthritis: prominent role of Wnt-induced signaling protein 1. Arthritis Rheum. 60, 501–512 (2009).
Honsawek, S. et al. Dickkopf-1 (Dkk-1) in plasma and synovial fluid is inversely correlated with radiographic severity of knee osteoarthritis patients. BMC Musculoskelet. Disord. 11, 257 (2010).
van den Bosch, M. H. et al. Brief report: induction of matrix metalloproteinase expression by synovial Wnt signaling and association with disease progression in early symptomatic osteoarthritis. Arthritis Rheumatol. 69, 1978–1983 (2017).
Oh, H., Chun, C. H. & Chun, J. S. Dkk-1 expression in chondrocytes inhibits experimental osteoarthritic cartilage destruction in mice. Arthritis Rheum. 64, 2568–2578 (2012).
Funck-Brentano, T. et al. Dkk-1-mediated inhibition of Wnt signaling in bone ameliorates osteoarthritis in mice. Arthritis Rheumatol. 66, 3028–3039 (2014).
Snelling, S. J. et al. Dickkopf-3 is upregulated in osteoarthritis and has a chondroprotective role. Osteoarthr. Cartil. 24, 883–891 (2016).
Conde, J. et al. Dickkopf-3 (DKK3) signaling in IL-1alpha-challenged chondrocytes: involvement of the NF-kappaB pathway. Cartilage 13, 925S–934S (2021).
van den Bosch, M. H. et al. Induction of canonical Wnt signaling by synovial overexpression of selected wnts leads to protease activity and early osteoarthritis-like cartilage damage. Am. J. Pathol. 185, 1970–1980 (2015).
Clevers, H. & Nusse, R. Wnt/β-catenin signaling and disease. Cell 149, 1192–1205 (2012).
Li, Y. et al. The expression of osteopontin and Wnt5a in articular cartilage of patients with knee osteoarthritis and its correlation with disease severity. Biomed. Res. Int 2016, 9561058 (2016).
Onuora, S. Osteoarthritis: Wnt inhibitor shows potential as a DMOAD. Nat. Rev. Rheumatol. 13, 634 (2017).
Deshmukh, V. et al. A small-molecule inhibitor of the Wnt pathway (SM04690) as a potential disease modifying agent for the treatment of osteoarthritis of the knee. Osteoarthr. Cartil. 26, 18–27 (2018).
De Palma, A. & Nalesso, G. WNT signalling in osteoarthritis and its pharmacological targeting. Handb. Exp. Pharmacol. 269, 337–356 (2021).
Deshmukh, V. et al. Modulation of the Wnt pathway through inhibition of CLK2 and DYRK1A by lorecivivint as a novel, potentially disease-modifying approach for knee osteoarthritis treatment. Osteoarthr. Cartil. 27, 1347–1360 (2019).
Lietman, C. et al. Inhibition of Wnt/beta-catenin signaling ameliorates osteoarthritis in a murine model of experimental osteoarthritis. JCI Insight 3, e96308 (2018).
Held, A. et al. Targeting beta-catenin dependent Wnt signaling via peptidomimetic inhibitors in murine chondrocytes and OA cartilage. Osteoarthr. Cartil. 26, 818–823 (2018).
Takamatsu, A. et al. Verapamil protects against cartilage degradation in osteoarthritis by inhibiting Wnt/beta-catenin signaling. PLoS One 9, e92699 (2014).
**a, H. et al. Jiawei Yanghe decoction ameliorates cartilage degradation in vitro and vivo via Wnt/beta-catenin signaling pathway. Biomed. Pharmacother. 122, 109708 (2020).
Li, K. et al. Tyrosine kinase Fyn promotes osteoarthritis by activating the beta-catenin pathway. Ann. Rheum. Dis. 77, 935–943 (2018).
Chen, M. et al. Inhibition of beta-catenin signaling causes defects in postnatal cartilage development. J. Cell Sci. 121, 1455–1465 (2008).
Jimi, E. & Ghosh, S. Role of nuclear factor-kappaB in the immune system and bone. Immunol. Rev. 208, 80–87 (2005).
Chen, S. et al. Kindlin-2 inhibits Nlrp3 inflammasome activation in nucleus pulposus to maintain homeostasis of the intervertebral disc. Bone Res. 10, 5 (2022).
Marcu, K. B., Otero, M., Olivotto, E., Borzi, R. M. & Goldring, M. B. NF-kappaB signaling: multiple angles to target OA. Curr. Drug Targets 11, 599–613 (2010).
Hoffmann, A. & Baltimore, D. Circuitry of nuclear factor κB signaling. Immunol. Rev. 210, 171–186 (2006).
Oeckinghaus, A. & Ghosh, S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb. Perspect. Biol. 1, a000034 (2009).
Yasuda, T. Activation of Akt leading to NF-κB up-regulation in chondrocytes stimulated with fibronectin fragment. Biomed. Res. 32, 209–215 (2011).
Arra, M., Swarnkar, G., Alippe, Y., Mbalaviele, G. & Abu-Amer, Y. IκB-ζ signaling promotes chondrocyte inflammatory phenotype, senescence, and erosive joint pathology. Bone Res. 10, 12 (2022).
Huang, B., Yang, X. D., Lamb, A. & Chen, L. F. Posttranslational modifications of NF-kappaB: another layer of regulation for NF-kappaB signaling pathway. Cell Signal. 22, 1282–1290 (2010).
Iotsova, V. et al. Osteopetrosis in mice lacking NF-kappaB1 and NF-kappaB2. Nat. Med. 3, 1285–1289 (1997).
Caron, M. M. et al. Activation of NF-κB/p65 facilitates early chondrogenic differentiation during endochondral ossification. PLoS One 7, e33467 (2012).
Kanegae, Y., Tavares, A. T., Izpisúa Belmonte, J. C. & Verma, I. M. Role of Rel/NF-kappaB transcription factors during the outgrowth of the vertebrate limb. Nature 392, 611–614 (1998).
Wu, S., Fadoju, D., Rezvani, G. & De Luca, F. Stimulatory effects of insulin-like growth factor-I on growth plate chondrogenesis are mediated by nuclear factor-kappaB p65. J. Biol. Chem. 283, 34037–34044 (2008).
Nakatomi, C. et al. Constitutive activation of the alternative NF-κB pathway disturbs endochondral ossification. Bone 121, 29–41 (2019).
Kobayashi, H. et al. Biphasic regulation of chondrocytes by Rela through induction of anti-apoptotic and catabolic target genes. Nat. Commun. 7, 13336 (2016).
Ulivi, V., Giannoni, P., Gentili, C., Cancedda, R. & Descalzi, F. p38/NF-kB-dependent expression of COX-2 during differentiation and inflammatory response of chondrocytes. J. Cell Biochem. 104, 1393–1406 (2008).
Yoon, D. S. et al. TLR4 downregulation by the RNA-binding protein PUM1 alleviates cellular aging and osteoarthritis. Cell Death Differ. 29, 1364–1378 (2022).
Kapoor, M., Martel-Pelletier, J., Lajeunesse, D., Pelletier, J. P. & Fahmi, H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat. Rev. Rheumatol. 7, 33–42 (2011).
Goldring, M. B. et al. Roles of inflammatory and anabolic cytokines in cartilage metabolism: signals and multiple effectors converge upon MMP-13 regulation in osteoarthritis. Eur. Cell Mater. 21, 202–220 (2011).
Li, W. et al. Role of HIF-2α/NF-κB pathway in mechanical stress-induced temporomandibular joint osteoarthritis. Oral Dis. 28, 2239–2247 (2021).
Zheng, T., Li, Y., Zhang, X., Xu, J. & Luo, M. Exosomes derived from miR-212-5p overexpressed human synovial mesenchymal stem cells suppress chondrocyte degeneration and inflammation by targeting ELF3. Front. Bioeng. Biotechnol. 10, 816209 (2022).
Lai, Y. et al. Kindlin-2 loss in condylar chondrocytes causes spontaneous osteoarthritic lesions in the temporomandibular joint in mice. Int. J. Oral. Sci. 14, 33 (2022).
Goldring, M. B. & Marcu, K. B. Cartilage homeostasis in health and rheumatic diseases. Arthritis Res. Ther. 11, 224 (2009).
Deng, Y. et al. Reciprocal inhibition of YAP/TAZ and NF-κB regulates osteoarthritic cartilage degradation. Nat. Commun. 9, 4564 (2018).
Yin, M. J., Yamamoto, Y. & Gaynor, R. B. The anti-inflammatory agents aspirin and salicylate inhibit the activity of I(kappa)B kinase-beta. Nature 396, 77–80 (1998).
Chen, S. et al. Moderate fluid shear stress regulates heme oxygenase-1 expression to promote autophagy and ECM homeostasis in the nucleus pulposus cells. Front Cell Dev. Biol. 8, 127 (2020).
Scheinman, R. I., Gualberto, A., Jewell, C. M., Cidlowski, J. A. & Baldwin, A. S. Jr. Characterization of mechanisms involved in transrepression of NF-kappa B by activated glucocorticoid receptors. Mol. Cell Biol. 15, 943–953 (1995).
Aitken, D. et al. A randomised double-blind placebo-controlled crossover trial of HUMira (adalimumab) for erosive hand OsteoaRthritis - the HUMOR trial. Osteoarthr. Cartil. 26, 880–887 (2018).
Kloppenburg, M. et al. Etanercept in patients with inflammatory hand osteoarthritis (EHOA): a multicentre, randomised, double-blind, placebo-controlled trial. Ann. Rheum. Dis. 77, 1757–1764 (2018).
Wang, S. X. et al. Safety, tolerability, and pharmacodynamics of an anti-interleukin-1alpha/beta dual variable domain immunoglobulin in patients with osteoarthritis of the knee: a randomized phase 1 study. Osteoarthr. Cartil. 25, 1952–1961 (2017).
Yan, Y., Zhou, X. E., Xu, H. E. & Melcher, K. Structure and physiological regulation of AMPK. Int J. Mol. Sci. 19, 3534 (2018).
Petursson, F. et al. Linked decreases in liver kinase B1 and AMP-activated protein kinase activity modulate matrix catabolic responses to biomechanical injury in chondrocytes. Arthritis Res. Ther. 15, R77 (2013).
Terkeltaub, R., Yang, B., Lotz, M. & Liu-Bryan, R. Chondrocyte AMP-activated protein kinase activity suppresses matrix degradation responses to proinflammatory cytokines interleukin-1beta and tumor necrosis factor alpha. Arthritis Rheum. 63, 1928–1937 (2011).
Ge, Y. et al. Estrogen prevents articular cartilage destruction in a mouse model of AMPK deficiency via ERK-mTOR pathway. Ann. Transl. Med. 7, 336 (2019).
Qiu, L. et al. Effect of Electroacupuncture on Synovial M 1/M 2 Macrophage Polarization in Rats with Acute Gouty Arthritis. Zhen Ci Yan Jiu 43, 767–772 (2018).
Zhou, S. et al. AMPK deficiency in chondrocytes accelerated the progression of instability-induced and ageing-associated osteoarthritis in adult mice. Sci. Rep. 7, 43245 (2017).
Mevel, E. et al. Systemic inhibition or global deletion of CaMKK2 protects against post-traumatic osteoarthritis. Osteoarthr. Cartil. 30, 124–136 (2022).
Momcilovic, M., Hong, S. P. & Carlson, M. Mammalian TAK1 activates Snf1 protein kinase in yeast and phosphorylates AMP-activated protein kinase in vitro. J. Biol. Chem. 281, 25336–25343 (2006).
Neumann, D. Is TAK1 a direct upstream kinase of AMPK? Int. J. Mol. Sci. 19, 2412 (2018).
Cheng, J. et al. Inhibition of transforming growth factor beta-activated kinase 1 prevents inflammation-related cartilage degradation in osteoarthritis. Sci. Rep. 6, 34497 (2016).
Wang, Y., Zhao, X., Lotz, M., Terkeltaub, R. & Liu-Bryan, R. Mitochondrial biogenesis is impaired in osteoarthritis chondrocytes but reversible via peroxisome proliferator-activated receptor gamma coactivator 1alpha. Arthritis Rheumatol. 67, 2141–2153 (2015).
Wang, J. et al. AMPK: implications in osteoarthritis and therapeutic targets. Am. J. Transl. Res. 12, 7670–7681 (2020).
Zhu, H. et al. CCAL1 enhances osteoarthritis through the NF-kappaB/AMPK signaling pathway. FEBS Open Bio 10, 2553–2563 (2020).
Wang, Y. et al. Inhibition of glycolysis ameliorate arthritis in adjuvant arthritis rats by inhibiting synoviocyte activation through AMPK/NF-small ka, CyrillicB pathway. Inflamm. Res. 69, 569–578 (2020).
Shang, L. & Wang, X. AMPK and mTOR coordinate the regulation of Ulk1 and mammalian autophagy initiation. Autophagy 7, 924–926 (2011).
Husa, M., Petursson, F., Lotz, M., Terkeltaub, R. & Liu-Bryan, R. C/EBP homologous protein drives pro-catabolic responses in chondrocytes. Arthritis Res. Ther. 15, R218 (2013).
Feng, K., Chen, Z., Pengcheng, L., Zhang, S. & Wang, X. Quercetin attenuates oxidative stress-induced apoptosis via SIRT1/AMPK-mediated inhibition of ER stress in rat chondrocytes and prevents the progression of osteoarthritis in a rat model. J. Cell Physiol. 234, 18192–18205 (2019).
Zhao, X. et al. Peroxisome proliferator-activated receptor gamma coactivator 1alpha and FoxO3A mediate chondroprotection by AMP-activated protein kinase. Arthritis Rheumatol. 66, 3073–3082 (2014).
Li, J. et al. Metformin limits osteoarthritis development and progression through activation of AMPK signalling. Ann. Rheum. Dis. 79, 635–645 (2020).
Feng, X. et al. Metformin attenuates cartilage degeneration in an experimental osteoarthritis model by regulating AMPK/mTOR. Aging (Albany NY) 12, 1087–1103 (2020).
Wang, C. et al. Metformin mitigates cartilage degradation by activating AMPK/SIRT1-mediated autophagy in a mouse osteoarthritis model. Front. Pharm. 11, 1114 (2020).
Story, G. M. et al. ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell 112, 819–829 (2003).
Wang, S. et al. Negative regulation of TRPA1 by AMPK in primary sensory neurons as a potential mechanism of painful diabetic neuropathy. Diabetes 67, 98–109 (2018).
Kong, C. et al. Active vitamin D activates chondrocyte autophagy to reduce osteoarthritis via mediating the AMPK-mTOR signaling pathway. Biochem. Cell Biol. 98, 434–442 (2020).
Kunanusornchai, W. et al. Chitosan oligosaccharide suppresses synovial inflammation via AMPK activation: an in vitro and in vivo study. Pharm. Res. 113, 458–467 (2016).
Carames, B. et al. Autophagy activation by rapamycin reduces severity of experimental osteoarthritis. Ann. Rheum. Dis. 71, 575–581 (2012).
Wang, C. et al. Safflower yellow alleviates osteoarthritis and prevents inflammation by inhibiting PGE2 release and regulating NF-kappaB/SIRT1/AMPK signaling pathways. Phytomedicine 78, 153305 (2020).
Wang, L. et al. Puerarin attenuates osteoarthritis via upregulating AMP-activated protein kinase/proliferator-activated receptor-gamma coactivator-1 signaling pathway in osteoarthritis rats. Pharmacology 102, 117–125 (2018).
Ma, T. et al. Bilobalide exerts anti-inflammatory effects on chondrocytes through the AMPK/SIRT1/mTOR pathway to attenuate aclt-induced post-traumatic osteoarthritis in rats. Front. Pharm. 13, 783506 (2022).
He, M. et al. Metformin prevents or delays the development and progression of osteoarthritis: new insight and mechanism of action. Cells 11, 3012 (2022).
Nageeb, M. M., Saadawy, S. F. & Attia, S. H. Breast milk mesenchymal stem cells abate cisplatin-induced cardiotoxicity in adult male albino rats via modulating the AMPK pathway. Sci. Rep. 12, 17554 (2022).
Yerevanian, A. & Soukas, A. A. Metformin: mechanisms in human obesity and weight loss. Curr. Obes. Rep. 8, 156–164 (2019).
Wang, Y. et al. Association between metformin use and disease progression in obese people with knee osteoarthritis: data from the Osteoarthritis Initiative-a prospective cohort study. Arthritis Res. Ther. 21, 127 (2019).
Ameen, O., Samaka, R. M. & Abo-Elsoud, R. A. A. Metformin alleviates neurocognitive impairment in aging via activation of AMPK/BDNF/PI3K pathway. Sci. Rep. 12, 17084 (2022).
Zhang, Y. et al. Neuromedin B receptor stimulation of Cav3.2 T-type Ca(2+) channels in primary sensory neurons mediates peripheral pain hypersensitivity. Theranostics 11, 9342–9357 (2021).
**ng, H. et al. Metformin mitigates cholesterol accumulation via the AMPK/SIRT1 pathway to protect osteoarthritis chondrocytes. Biochem. Biophys. Res. Commun. 632, 113–121 (2022).
Na, H. S. et al. Metformin attenuates monosodium-iodoacetate-induced osteoarthritis via regulation of pain mediators and the autophagy-lysosomal pathway. Cells 10, 681 (2021).
Song, Y., Wu, Z. & Zhao, P. The effects of metformin in the treatment of osteoarthritis: current perspectives. Front. Pharm. 13, 952560 (2022).
Nagata, K. et al. Runx2 and Runx3 differentially regulate articular chondrocytes during surgically induced osteoarthritis development. Nat. Commun. 13, 6187 (2022).
Lin, C. C. et al. Metformin enhances cisplatin cytotoxicity by suppressing signal transducer and activator of transcription-3 activity independently of the liver kinase B1-AMP-activated protein kinase pathway. Am. J. Respir. Cell Mol. Biol. 49, 241–250 (2013).
Zheng, Z., Bian, Y., Zhang, Y., Ren, G. & Li, G. Metformin activates AMPK/SIRT1/NF-kappaB pathway and induces mitochondrial dysfunction to drive caspase3/GSDME-mediated cancer cell pyroptosis. Cell Cycle 19, 1089–1104 (2020).
Luo, X., Hu, R., Zheng, Y., Liu, S. & Zhou, Z. Metformin shows anti-inflammatory effects in murine macrophages through Dicer/microribonucleic acid-34a-5p and microribonucleic acid-125b-5p. J. Diabetes Investig. 11, 101–109 (2020).
Tian, R. et al. Metformin ameliorates endotoxemia-induced endothelial pro-inflammatory responses via AMPK-dependent mediation of HDAC5 and KLF2. Biochim. Biophys. Acta Mol. Basis Dis. 1865, 1701–1712 (2019).
Lin, Z. et al. JUNB-FBXO21-ERK axis promotes cartilage degeneration in osteoarthritis by inhibiting autophagy. Aging Cell 20, e13306 (2021).
Hwang, H. Y., Shim, J. S., Kim, D. & Kwon, H. J. Antidepressant drug sertraline modulates AMPK-MTOR signaling-mediated autophagy via targeting mitochondrial VDAC1 protein. Autophagy 17, 2783–2799 (2021).
Guo, H. et al. Induction of autophagy via the ROS-dependent AMPK-mTOR pathway protects copper-induced spermatogenesis disorder. Redox Biol. 49, 102227 (2022).
Wang, C. et al. Protective effects of metformin against osteoarthritis through upregulation of SIRT3-mediated PINK1/Parkin-dependent mitophagy in primary chondrocytes. Biosci. Trends 12, 605–612 (2019).
Klionsky, D. J. The autophagy connection. Dev. Cell 19, 11–12 (2010).
Saxton, R. A. & Sabatini, D. M. mTOR signaling in growth, metabolism, and disease. Cell 168, 960–976 (2017).
Mossmann, D., Park, S. & Hall, M. N. mTOR signalling and cellular metabolism are mutual determinants in cancer. Nat. Rev. Cancer 18, 744–757 (2018).
Holz, M. K., Ballif, B. A., Gygi, S. P. & Blenis, J. mTOR and S6K1 mediate assembly of the translation preinitiation complex through dynamic protein interchange and ordered phosphorylation events. Cell 123, 569–580 (2005).
Sarbassov, D. D., Guertin, D. A., Ali, S. M. & Sabatini, D. M. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307, 1098–1101 (2005).
Liu, P. D. et al. PtdIns(3,4,5)P-3-dependent activation of the mTORC2 kinase complex. Cancer Discov. 5, 1194–1209 (2015).
Gangloff, Y. G. et al. Disruption of the mouse mTOR gene leads to early postimplantation lethality and prohibits embryonic stem cell development. Mol. Cell. Biol. 24, 9508–9516 (2004).
Pal, B., Endisha, H., Zhang, Y. & Kapoor, M. mTOR: a potential therapeutic target in osteoarthritis? Drugs R. D. 15, 27–36 (2015).
Zhang, Y. et al. Cartilage-specific deletion of mTOR upregulates autophagy and protects mice from osteoarthritis. Ann. Rheum. Dis. 74, 1432–1440 (2015).
Zhang, H. et al. mTORC1 activation downregulates FGFR3 and PTH/PTHrP receptor in articular chondrocytes to initiate osteoarthritis. Osteoarthr. Cartil. 25, 952–963 (2017).
Vasheghani, F. et al. PPAR gamma deficiency results in severe, accelerated osteoarthritis associated with aberrant mTOR signalling in the articular cartilage. Ann. Rheum. Dis. 74, 569–578 (2015).
Kim, J., Kundu, M., Viollet, B. & Guan, K. L. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat. Cell Biol. 13, 132–U171 (2011).
Bijlsma, J. W. J., Berenbaum, F. & Lafeber, F. P. J. G. Osteoarthritis: an update with relevance for clinical practice. Lancet 377, 2115–2126 (2011).
Yang, H. X. et al. MTORC1 coordinates the autophagy and apoptosis signaling in articular chondrocytes in osteoarthritic temporomandibular joint. Autophagy 16, 271–288 (2020).
Lin, C. X. et al. Activation of mTORC1 in subchondral bone preosteoblasts promotes osteoarthritis by stimulating bone sclerosis and secretion of CXCL12. Bone Res. 7, 5 (2019).
Karonitsch, T. et al. mTOR senses environmental cues to shape the fibroblast-like synoviocyte response to inflammation. Cell Rep. 23, 2157–2167 (2018).
Moutinho, C. & Esteller, M. MicroRNAs and epigenetics. Adv. Cancer Res. 135, 189–220 (2017).
Jiang, H. T., Dai, J., Zhang, C., Sun, H. L. & Tang, X. M. Circ_0045714 alleviates TNF-alpha-induced chondrocyte injury and extracellular matrix degradation through miR-218-5p/HRAS axis. J. Bioenerg. Biomembr. 53, 97–107 (2021).
Zhou, X. D. et al. Down-regulated ciRS-7/up-regulated miR-7 axis aggravated cartilage degradation and autophagy defection by PI3K/AKT/mTOR activation mediated by IL-17A in osteoarthritis. Aging Us 12, 20163–20183 (2020).
Cai, C. et al. MiR-27a promotes the autophagy and apoptosis of IL-1 beta treated-articular chonarocytes in osteoarthritis through PI3K/AKT/mTOR signaling. Aging Us 11, 6371–6384 (2019).
Wu, J. Y. et al. miR-100-5p-abundant exosomes derived from infrapatellar fat pad MSCs protect articular cartilage and ameliorate gait abnormalities via inhibition of mTOR in osteoarthritis. Biomaterials 206, 87–100 (2019).
Ansari, M. Y., Khan, N. M. & Haqqi, T. M. A standardized extract of Butea monosperma (Lam.) flowers suppresses the IL-1 beta-induced expression of IL-6 and matrix-metalloproteases by activating autophagy in human osteoarthritis chondrocytes. Biomed. Pharmacother. 96, 198–207 (2017).
Dunwoodie, S. L. The role of hypoxia in development of the Mammalian embryo. Dev. Cell 17, 755–773 (2009).
Semenza, G. L. Hypoxia-inducible factors in physiology and medicine. Cell 148, 399–408 (2012).
Schipani, E. et al. Hypoxia in cartilage: HIF-1alpha is essential for chondrocyte growth arrest and survival. Genes Dev. 15, 2865–2876 (2001).
Yao, Q. et al. Suppressing mitochondrial respiration is critical for hypoxia tolerance in the fetal growth plate. Dev. Cell 49, 748–763 e747 (2019).
Long, F. Less is more: ditching mitochondria saves hypoxic cartilage. Dev. Cell 49, 656–658 (2019).
Yao, Q., Parvez-Khan, M. & Schipani, E. In vivo survival strategies for cellular adaptation to hypoxia: HIF1alpha-dependent suppression of mitochondrial oxygen consumption and decrease of intracellular hypoxia are critical for survival of hypoxic chondrocytes. Bone 140, 115572 (2020).
Saito, T. et al. Transcriptional regulation of endochondral ossification by HIF-2alpha during skeletal growth and osteoarthritis development. Nat. Med. 16, 678–686 (2010).
Araldi, E., Khatri, R., Giaccia, A. J., Simon, M. C. & Schipani, E. Lack of HIF-2alpha in limb bud mesenchyme causes a modest and transient delay of endochondral bone development. Nat. Med. 17, 25–26 (2011).
Pfander, D. & Gelse, K. Hypoxia and osteoarthritis: how chondrocytes survive hypoxic environments. Curr. Opin. Rheumatol. 19, 457–462 (2007).
Ummarino, D. Osteoarthritis: hypoxia protects against cartilage loss by regulating Wnt signalling. Nat. Rev. Rheumatol. 12, 315 (2016).
Pfander, D., Cramer, T. & Swoboda, B. Hypoxia and HIF-1alpha in osteoarthritis. Int. Orthop. 29, 6–9 (2005).
Yang, S. et al. Hypoxia-inducible factor-2alpha is a catabolic regulator of osteoarthritic cartilage destruction. Nat. Med. 16, 687–693 (2010).
Bouaziz, W. et al. Interaction of HIF1alpha and beta-catenin inhibits matrix metalloproteinase 13 expression and prevents cartilage damage in mice. Proc. Natl Acad. Sci. USA 113, 5453–5458 (2016).
Qing, L. et al. Expression of hypoxia-inducible factor-1alpha in synovial fluid and articular cartilage is associated with disease severity in knee osteoarthritis. Exp. Ther. Med. 13, 63–68 (2017).
Lu, J. et al. Hypoxia inducible factor-1alpha is a regulator of autophagy in osteoarthritic chondrocytes. Cartilage 13, 1030S–1040S (2021).
Yang, F., Huang, R., Ma, H., Zhao, X. & Wang, G. miRNA-411 regulates chondrocyte autophagy in osteoarthritis by targeting hypoxia-inducible factor 1 alpha (HIF-1alpha). Med. Sci. Monit. 26, e921155 (2020).
Hu, S. et al. Stabilization of HIF-1alpha alleviates osteoarthritis via enhancing mitophagy. Cell Death Dis. 11, 481 (2020).
Weng, T. et al. Loss of Vhl in cartilage accelerated the progression of age-associated and surgically induced murine osteoarthritis. Osteoarthr. Cartil. 22, 1197–1205 (2014).
Rius, J. et al. NF-kappaB links innate immunity to the hypoxic response through transcriptional regulation of HIF-1alpha. Nature 453, 807–811 (2008).
Chen, J., Crawford, R. & **ao, Y. Vertical inhibition of the PI3K/Akt/mTOR pathway for the treatment of osteoarthritis. J. Cell. Biochem. 114, 245–249 (2013).
Fernandez-Torres, J., Zamudio-Cuevas, Y., Martinez-Nava, G. A. & Lopez-Reyes, A. G. Hypoxia-Inducible Factors (HIFs) in the articular cartilage: a systematic review. Eur. Rev. Med. Pharmacol. Sci. 21, 2800–2810 (2017).
Li, W., Cai, L., Zhang, Y., Cui, L. & Shen, G. Intra-articular resveratrol injection prevents osteoarthritis progression in a mouse model by activating SIRT1 and thereby silencing HIF-2alpha. J. Orthop. Res. 33, 1061–1070 (2015).
**a, Y., Choi, H. K. & Lee, K. Recent advances in hypoxia-inducible factor (HIF)-1 inhibitors. Eur. J. Med. Chem. 49, 24–40 (2012).
Burridge, K. Focal adhesions: a personal perspective on a half century of progress. FEBS J. 284, 3355–3361 (2017).
Cao, H. et al. Focal adhesion protein Kindlin-2 regulates bone homeostasis in mice. Bone Res. 8, 2 (2020).
Wang, Y. et al. Focal adhesion proteins Pinch1 and Pinch2 regulate bone homeostasis in mice. JCI Insight 4, e131692 (2019).
Fu, X. et al. Kindlin-2 regulates skeletal homeostasis by modulating PTH1R in mice. Signal Transduct. Target Ther. 5, 297 (2020).
Wu, C. et al. Kindlin-2 controls TGF-beta signalling and Sox9 expression to regulate chondrogenesis. Nat. Commun. 6, 7531 (2015).
Gao, H. et al. Lipoatrophy and metabolic disturbance in mice with adipose-specific deletion of kindlin-2. JCI Insight. 4, e128405 (2019).
Gao, H. et al. Pinch loss ameliorates obesity, glucose intolerance, and fatty liver by modulating adipocyte apoptosis in mice. Diabetes 70, 2492–2505 (2021).
Gao, H. et al. Kindlin-2 haploinsufficiency protects against fatty liver by targeting Foxo1 in mice. Nat. Commun. 13, 1025 (2022).
Guo, L. et al. Kindlin-2 regulates mesenchymal stem cell differentiation through control of YAP1/TAZ. J. Cell Biol. 217, 1431–1451 (2018).
Wu, X. et al. Kindlin-2 preserves integrity of the articular cartilage to protect against osteoarthritis. Nat. Aging 2, 332–347 (2022).
Wu, X. et al. Kindlin-2 deletion in osteoprogenitors causes severe chondrodysplasia and low-turnover osteopenia in mice. J. Orthop. Transl. 32, 41–48 (2022).
Zhu, K. et al. Kindlin-2 modulates MafA and beta-catenin expression to regulate beta-cell function and mass in mice. Nat. Commun. 11, 484 (2020).
Lei, Y. et al. LIM domain proteins Pinch1/2 regulate chondrogenesis and bone mass in mice. Bone Res. 8, 37 (2020).
Raducanu, A., Hunziker, E. B., Drosse, I. & Aszodi, A. Beta1 integrin deficiency results in multiple abnormalities of the knee joint. J. Biol. Chem. 284, 23780–23792 (2009).
**, H. et al. Mechanistic insight into the roles of integrins in osteoarthritis. Front. Cell Dev. Biol. 9, 693484 (2021).
Qin, L., Liu, W., Cao, H. & **ao, G. Molecular mechanosensors in osteocytes. Bone Res. 8, 23 (2020).
Qin, L. et al. Roles of mechanosensitive channel Piezo1/2 proteins in skeleton and other tissues. Bone Res. 9, 44 (2021).
Lian, C. et al. Collagen type II suppresses articular chondrocyte hypertrophy and osteoarthritis progression by promoting integrin beta1-SMAD1 interaction. Bone Res. 7, 8 (2019).
Zhen, G. et al. Mechanical stress determines the configuration of TGFbeta activation in articular cartilage. Nat. Commun. 12, 1706 (2021).
Zhen, G. et al. Inhibition of TGF-beta signaling in mesenchymal stem cells of subchondral bone attenuates osteoarthritis. Nat. Med. 19, 704–712 (2013).
Ciregia, F. et al. Modulation of alphaVbeta6 integrin in osteoarthritis-related synovitis and the interaction with VTN(381-397 a.a.) competing for TGF-beta1 activation. Exp. Mol. Med. 53, 210–222 (2021).
Wang, Q. et al. Dysregulated integrin alphaVbeta3 and CD47 signaling promotes joint inflammation, cartilage breakdown, and progression of osteoarthritis. JCI Insight. 4, e128616 (2019).
Wu, H. et al. Specific inhibition of FAK signaling attenuates subchondral bone deterioration and articular cartilage degeneration during osteoarthritis pathogenesis. J. Cell Physiol. 235, 8653–8666 (2020).
Yanoshita, M. et al. FAK inhibition protects condylar cartilage under excessive mechanical stress. Oral. Dis. 26, 1736–1746 (2020).
Miyauchi, A. et al. Alleviation of murine osteoarthritis by deletion of the focal adhesion mechanosensitive adapter, Hic-5. Sci. Rep. 9, 15770 (2019).
Salazar, V. S., Gamer, L. W. & Rosen, V. BMP signalling in skeletal development, disease and repair. Nat. Rev. Endocrinol. 12, 203–221 (2016).
Zhang, Y. & Que, J. BMP signaling in development, stem cells, and diseases of the gastrointestinal tract. Annu Rev. Physiol. 82, 251–273 (2020).
Heldin, C. H., Miyazono, K. & ten Dijke, P. TGF-beta signalling from cell membrane to nucleus through SMAD proteins. Nature 390, 465–471 (1997).
Wang, R. N. et al. Bone Morphogenetic Protein (BMP) signaling in development and human diseases. Genes Dis. 1, 87–105 (2014).
Lowery, J. W. & Rosen, V. The BMP pathway and its inhibitors in the skeleton. Physiol. Rev. 98, 2431–2452 (2018).
Ashraf, S. et al. Regulation of senescence associated signaling mechanisms in chondrocytes for cartilage tissue regeneration. Osteoarthr. Cartil. 24, 196–205 (2016).
Deng, Z. H., Li, Y. S., Gao, X., Lei, G. H. & Huard, J. Bone morphogenetic proteins for articular cartilage regeneration. Osteoarthr. Cartil. 26, 1153–1161 (2018).
Thielen, N. G. M., van der Kraan, P. M. & van Caam, A. P. M. TGFbeta/BMP signaling pathway in cartilage homeostasis. Cells 8, 969 (2019).
Zhong, L., Huang, X., Karperien, M. & Post, J. N. The regulatory role of signaling crosstalk in hypertrophy of MSCs and human articular chondrocytes. Int. J. Mol. Sci. 16, 19225–19247 (2015).
Blaney Davidson, E. N. et al. Elevated extracellular matrix production and degradation upon bone morphogenetic protein-2 (BMP-2) stimulation point toward a role for BMP-2 in cartilage repair and remodeling. Arthritis Res. Ther. 9, R102 (2007).
van Beuningen, H. M., Glansbeek, H. L., van der Kraan, P. M. & van den Berg, W. B. Differential effects of local application of BMP-2 or TGF-beta 1 on both articular cartilage composition and osteophyte formation. Osteoarthr. Cartil. 6, 306–317 (1998).
Zhou, N. et al. BMP2 induces chondrogenic differentiation, osteogenic differentiation and endochondral ossification in stem cells. Cell Tissue Res. 366, 101–111 (2016).
Flechtenmacher, J. et al. Recombinant human osteogenic protein 1 is a potent stimulator of the synthesis of cartilage proteoglycans and collagens by human articular chondrocytes. Arthritis Rheum. 39, 1896–1904 (1996).
Luyten, F. P., Chen, P., Paralkar, V. & Reddi, A. H. Recombinant bone morphogenetic protein-4, transforming growth factor-beta 1, and activin A enhance the cartilage phenotype of articular chondrocytes in vitro. Exp. Cell Res 210, 224–229 (1994).
Bobacz, K. et al. Expression of bone morphogenetic protein 6 in healthy and osteoarthritic human articular chondrocytes and stimulation of matrix synthesis in vitro. Arthritis Rheum. 48, 2501–2508 (2003).
Gamer, L. W. et al. The Role of Bmp2 in the maturation and maintenance of the murine knee joint. J. Bone Min. Res. 33, 1708–1717 (2018).
Caron, M. M. et al. Hypertrophic differentiation during chondrogenic differentiation of progenitor cells is stimulated by BMP-2 but suppressed by BMP-7. Osteoarthr. Cartil. 21, 604–613 (2013).
Caron, M. M. et al. BAPX-1/NKX-3.2 acts as a chondrocyte hypertrophy molecular switch in osteoarthritis. Arthritis Rheumatol. 67, 2944–2956 (2015).
Abula, K. et al. Elimination of BMP7 from the develo** limb mesenchyme leads to articular cartilage degeneration and synovial inflammation with increased age. FEBS Lett. 589, 1240–1248 (2015).
Papathanasiou, I., Malizos, K. N. & Tsezou, A. Bone morphogenetic protein-2-induced Wnt/beta-catenin signaling pathway activation through enhanced low-density-lipoprotein receptor-related protein 5 catabolic activity contributes to hypertrophy in osteoarthritic chondrocytes. Arthritis Res. Ther. 14, R82 (2012).
Wang, W. et al. The TGFbeta type I receptor TGFbetaRI functions as an inhibitor of BMP signaling in cartilage. Proc. Natl. Acad. Sci. USA 116, 15570–15579 (2019).
Zhai, G. Clinical relevance of biochemical and metabolic changes in osteoarthritis. Adv. Clin. Chem. 101, 95–120 (2021).
Perez-Lozano, M. L. et al. Gremlin-1 and BMP-4 overexpressed in osteoarthritis drive an osteochondral-remodeling program in osteoblasts and hypertrophic chondrocytes. Int. J. Mol. Sci. 23, 2084 (2022).
Chien, S. Y. et al. Noggin Inhibits IL-1beta and BMP-2 expression, and attenuates cartilage degeneration and subchondral bone destruction in experimental osteoarthritis. Cells 9, 927 (2020).
Iijima, H. et al. Physiological exercise loading suppresses post-traumatic osteoarthritis progression via an increase in bone morphogenetic proteins expression in an experimental rat knee model. Osteoarthr. Cartil. 25, 964–975 (2017).
Melnik, S., Hofmann, N., Gabler, J., Hecht, N. & Richter, W. MiR-181a targets RSPO2 and regulates bone morphogenetic protein - WNT signaling crosstalk during chondrogenic differentiation of mesenchymal stromal cells. Front. Cell Dev. Biol. 9, 747057 (2021).
Caron, M. M. J. et al. Discovery of bone morphogenetic protein 7-derived peptide sequences that attenuate the human osteoarthritic chondrocyte phenotype. Mol. Ther. Methods Clin. Dev. 21, 247–261 (2021).
Akkiraju, H. et al. CK2.1, a bone morphogenetic protein receptor type Ia mimetic peptide, repairs cartilage in mice with destabilized medial meniscus. Stem Cell Res. Ther. 8, 82 (2017).
Taniyama, T. et al. Repair of osteochondral defects in a rabbit model using a porous hydroxyapatite collagen composite impregnated with bone morphogenetic protein-2. Artif. Organs 39, 529–535 (2015).
Han, F. X. et al. A pilot study of conically graded chitosan-gelatin hydrogel/PLGA scaffold with dual-delivery of TGF-beta 1 and BMP-2 for regeneration of cartilage-bone interface. J. Biomed. Mater. Res. Part B-Appl. Biomater. 103, 1344–1353 (2015).
Jeong, C. G., Zhang, H. N. & Hollister, S. J. Three-dimensional polycaprolactone scaffold-conjugated bone morphogenetic protein-2 promotes cartilage regeneration from primary chondrocytes in vitro and in vivo without accelerated endochondral ossification. J. Biomed. Mater. Res. Part A 100a, 2088–2096 (2012).
Zhang, Z. et al. The functions of BMP3 in rabbit articular cartilage repair. Int. J. Mol. Sci. 16, 25934–25946 (2015).
Li, H. S. et al. The superior regenerative potential of muscle-derived stem cells for articular cartilage repair is attributed to high cell survival and chondrogenic potential. Mol. Ther.-Methods Clin. Dev. 3, 16065 (2016).
Sekiya, I. et al. Periodic knee injections of BMP-7 delay cartilage degeneration induced by excessive running in rats. J. Orthop. Res. 27, 1088–1092 (2009).
Mendes, L. F. et al. Combinatorial analysis of growth factors reveals the contribution of bone morphogenetic proteins to chondrogenic differentiation of human periosteal cells. Tissue Eng. Part C. 22, 473–486 (2016).
Choi, S., Cho, T. J., Kwon, S. K., Lee, G. & Cho, J. Chondrogenesis of periodontal ligament stem cells by transforming growth factor-beta 3 and bone morphogenetic protein-6 in a normal healthy impacted third molar. Int. J. Oral. Sci. 5, 7–13 (2013).
Ren, X. Y. et al. Nanoparticulate mineralized collagen scaffolds and BMP-9 induce a long-term bone cartilage construct in human mesenchymal stem cells. Adv. Healthc. Mater. 5, 1821–1830 (2016).
Thisse, B. & Thisse, C. Functions and regulations of fibroblast growth factor signaling during embryonic development. Dev. Biol. 287, 390–402 (2005).
Beenken, A. & Mohammadi, M. The FGF family: biology, pathophysiology and therapy. Nat. Rev. Drug Discov. 8, 235–253 (2009).
Ornitz, D. M. & Itoh, N. New developments in the biology of fibroblast growth factors. WIREs Mech. Dis. 14, e1549 (2022).
Im, H. J. et al. Basic fibroblast growth factor stimulates matrix metalloproteinase-13 via the molecular cross-talk between the mitogen-activated protein kinases and protein kinase Cdelta pathways in human adult articular chondrocytes. J. Biol. Chem. 282, 11110–11121 (2007).
Orr-Urtreger, A., Givol, D., Yayon, A., Yarden, Y. & Lonai, P. Developmental expression of two murine fibroblast growth factor receptors, flg and bek. Development 113, 1419–1434 (1991).
Mohammadi, M., Olsen, S. K. & Ibrahimi, O. A. Structural basis for fibroblast growth factor receptor activation. Cytokine Growth Factor Rev. 16, 107–137 (2005).
Kisand, K., Tamm, A. E., Lintrop, M. & Tamm, A. O. New insights into the natural course of knee osteoarthritis: early regulation of cytokines and growth factors, with emphasis on sex-dependent angiogenesis and tissue remodeling. A pilot study. Osteoarthr. Cartil. 26, 1045–1054 (2018).
El-Seoudi, A. et al. Catabolic effects of FGF-1 on chondrocytes and its possible role in osteoarthritis. J. Cell Commun. Signal. 11, 255–263 (2017).
Vincent, T. L., McLean, C. J., Full, L. E., Peston, D. & Saklatvala, J. FGF-2 is bound to perlecan in the pericellular matrix of articular cartilage, where it acts as a chondrocyte mechanotransducer. Osteoarthr. Cartil. 15, 752–763 (2007).
Im, H.-J. et al. Basic fibroblast growth factor accelerates matrix degradation via a neuro-endocrine pathway in human adult articular chondrocytes. J. Cell. Physiol. 215, 452–463 (2008).
Nummenmaa, E., Hämäläinen, M., Moilanen, T., Vuolteenaho, K. & Moilanen, E. Effects of FGF-2 and FGF receptor antagonists on MMP enzymes, aggrecan, and type II collagen in primary human OA chondrocytes. Scand. J. Rheumatol. 44, 321–330 (2015).
Uchii, M. et al. Role of fibroblast growth factor 8 (FGF8) in animal models of osteoarthritis. Arthritis Res. Ther. 10, R90 (2008).
Zhou, S. et al. Exogenous fibroblast growth factor 9 attenuates cartilage degradation and aggravates osteophyte formation in post-traumatic osteoarthritis. Osteoarthr. Cartil. 24, 2181–2192 (2016).
Rockel, J. S. et al. Hedgehog inhibits β-catenin activity in synovial joint development and osteoarthritis. J. Clin. Investig. 126, 1649–1663 (2016).
Mori, Y. et al. Identification of fibroblast growth factor-18 as a molecule to protect adult articular cartilage by gene expression profiling. J. Biol. Chem. 289, 10192–10200 (2014).
Meloni, G. R. et al. Recombinant human FGF18 preserves depth-dependent mechanical inhomogeneity in articular cartilage. Eur. Cells Mater. 38, 23–34 (2019).
Li, R. et al. Upregulation of fibroblast growth factor 1 in the synovial membranes of patients with late stage osteoarthritis. Genet. Mol. Res. 14, 11191–11199 (2015).
Kim, J. H., Lee, M. C., Seong, S. C., Park, K. H. & Lee, S. Enhanced proliferation and chondrogenic differentiation of human synovium-derived stem cells expanded with basic fibroblast growth factor. Tissue Eng. Part A 17, 991–1002 (2011).
Weng, T. et al. Genetic inhibition of fibroblast growth factor receptor 1 in knee cartilage attenuates the degeneration of articular cartilage in adult mice. Arthritis Rheum. 64, 3982–3992 (2012).
Deng, C., Wynshaw-Boris, A., Zhou, F., Kuo, A. & Leder, P. Fibroblast growth factor receptor 3 is a negative regulator of bone growth. Cell 84, 911–921 (1996).
Hochberg, M. C. et al. Effect of intra-articular sprifermin vs placebo on femorotibial joint cartilage thickness in patients with osteoarthritis: the FORWARD randomized clinical trial. Jama 322, 1360–1370 (2019).
**ao, G., Jiang, D., Gopalakrishnan, R. & Franceschi, R. T. Fibroblast growth factor 2 induction of the osteocalcin gene requires MAPK activity and phosphorylation of the osteoblast transcription factor, Cbfa1/Runx2. J. Biol. Chem. 277, 36181–36187 (2002).
Komori, T. Runx2, an inducer of osteoblast and chondrocyte differentiation. Histochem. Cell Biol. 149, 313–323 (2018).
Renny, T. F. et al. Multiple signaling pathways converge on the Cbfa1/Runx2 transcription factor to regulate osteoblast differentiation. Connect Tissue Res. 44, 109–116 (2003).
**ao, G. et al. Cooperative interactions between activating transcription factor 4 and Runx2/Cbfa1 stimulate osteoblast-specific osteocalcin gene expression. J. Biol. Chem. 280, 30689–30696 (2005).
Zhao, Z., Zhao, M., **ao, G. & Franceschi, R. T. Gene transfer of the Runx2 transcription factor enhances osteogenic activity of bone marrow stromal cells in vitro and in vivo. Mol. Ther. 12, 247–253 (2005).
Yu, S. et al. General transcription factor IIA-gamma increases osteoblast-specific osteocalcin gene expression via activating transcription factor 4 and runt-related transcription factor 2. J. Biol. Chem. 283, 5542–5553 (2008).
Zheng, Q. et al. Type X collagen gene regulation by Runx2 contributes directly to its hypertrophic chondrocyte-specific expression in vivo. J. Cell Biol. 162, 833–842 (2003).
Enomoto, H. et al. Cbfa1 is a positive regulatory factor in chondrocyte maturation. J. Biol. Chem. 275, 8695–8702 (2000).
Otto, F. et al. Cbfa1, a candidate gene for cleidocranial dysplasia syndrome, is essential for osteoblast differentiation and bone development. Cell 89, 765–771 (1997).
Tsuji, K., Komori, T. & Noda, M. Aged mice require full transcription factor, Runx2/Cbfa1, gene dosage for cancellous bone regeneration after bone marrow ablation. J. Bone Min. Res. 19, 1481–1489 (2004).
Catheline, S. E. et al. Chondrocyte-specific RUNX2 overexpression accelerates post-traumatic osteoarthritis progression in adult mice. J. Bone Min. Res. 34, 1676–1689 (2019).
Liao, L. et al. Deletion of Runx2 in articular chondrocytes decelerates the progression of DMM-induced osteoarthritis in adult mice. Sci. Rep. 7, 2371 (2017).
Li, F. et al. Runx2 contributes to murine Col10a1 gene regulation through direct interaction with its cis-enhancer. J. Bone Min. Res. 26, 2899–2910 (2011).
Zhao, W. et al. Runx2 and microRNA regulation in bone and cartilage diseases. Ann. N. Y. Acad. Sci. 1383, 80–87 (2016).
Tosa, I. et al. Postnatal Runx2 deletion leads to low bone mass and adipocyte accumulation in mice bone tissues. Biochem. Biophys. Res. Commun. 516, 1229–1233 (2019).
Adhami, M., Ghori-Javed, F. Y., Chen, H., Gutierrez, S. E. & Javed, A. Runx2 regulates the gene network associated with insulin signaling and energy homeostasis. Cells Tissues Organs 194, 232–237 (2011).
Chopin, M. et al. RUNX2 mediates plasmacytoid dendritic cell egress from the bone marrow and controls viral immunity. Cell Rep. 15, 866–878 (2016).
Kim, M. S., Gernapudi, R., Choi, E. Y., Lapidus, R. G. & Passaniti, A. Characterization of CADD522, a small molecule that inhibits RUNX2-DNA binding and exhibits antitumor activity. Oncotarget 8, 70916–70940 (2017).
Ji, M. L. et al. Precise targeting of miR-141/200c cluster in chondrocytes attenuates osteoarthritis development. Ann. Rheum. Dis. 80, 356–366 (2021).
Wei, Y. et al. Targeting cartilage EGFR pathway for osteoarthritis treatment. Sci. Transl. Med. 13, eabb3946 (2021).
Wei, Y. et al. Phospholipase A(2) inhibitor-loaded micellar nanoparticles attenuate inflammation and mitigate osteoarthritis progression. Sci. Adv. 7, eabe6374 (2021).
Zhao, L. et al. Exploration of CRISPR/Cas9-based gene editing as therapy for osteoarthritis. Ann. Rheum. Dis. 78, 676–682 (2019).
Coutinho de Almeida, R. et al. RNA sequencing data integration reveals an miRNA interactome of osteoarthritis cartilage. Ann. Rheum. Dis. 78, 270–277 (2019).
Ntoumou, E. et al. Serum microRNA array analysis identifies miR-140-3p, miR-33b-3p and miR-671-3p as potential osteoarthritis biomarkers involved in metabolic processes. Clin. Epigenet. 9, 127 (2017).
Zhang, H., Song, B. & Pan, Z. Downregulation of microRNA-9 increases matrix metalloproteinase-13 expression levels and facilitates osteoarthritis onset. Mol. Med. Rep. 17, 3708–3714 (2018).
Mao, G. et al. Exosomal miR-95-5p regulates chondrogenesis and cartilage degradation via histone deacetylase 2/8. J. Cell Mol. Med. 22, 5354–5366 (2018).
Zhao, X., Li, H. & Wang, L. MicroRNA-107 regulates autophagy and apoptosis of osteoarthritis chondrocytes by targeting TRAF3. Int. Immunopharmacol. 71, 181–187 (2019).
Song, J. et al. MicroRNA-181b regulates articular chondrocytes differentiation and cartilage integrity. Biochem. Biophys. Res. Commun. 431, 210–214 (2013).
Lian, W. S. et al. MicroRNA-128a represses chondrocyte autophagy and exacerbates knee osteoarthritis by disrupting Atg12. Cell Death Dis. 9, 919 (2018).
Meng, F. et al. MicroRNA-320 regulates matrix metalloproteinase-13 expression in chondrogenesis and interleukin-1beta-induced chondrocyte responses. Osteoarthr. Cartil. 24, 932–941 (2016).
Liu, J. et al. Exosomal transfer of osteoclast-derived miRNAs to chondrocytes contributes to osteoarthritis progression. Nat. Aging 1, 368–384 (2021).
Little, C. B. et al. Matrix metalloproteinase 13-deficient mice are resistant to osteoarthritic cartilage erosion but not chondrocyte hypertrophy or osteophyte development. Arthritis Rheum. 60, 3723–3733 (2009).
Wang, M. et al. MMP13 is a critical target gene during the progression of osteoarthritis. Arthritis Res. Ther. 15, R5 (2013).
Salazar-Noratto, G. E., De Nijs, N., Stevens, H. Y., Gibson, G. & Guldberg, R. E. Regional gene expression analysis of multiple tissues in an experimental animal model of post-traumatic osteoarthritis. Osteoarthr. Cartil. 27, 294–303 (2019).
Chang, J. C. et al. Global molecular changes in a tibial compression induced ACL rupture model of post-traumatic osteoarthritis. J. Orthop. Res. 35, 474–485 (2017).
Nuti, E. et al. N-O-isopropyl sulfonamido-based hydroxamates: design, synthesis and biological evaluation of selective matrix metalloproteinase-13 inhibitors as potential therapeutic agents for osteoarthritis. J. Med; Chem. 52, 4757–4773 (2009).
De Savi, C. et al. Selective non zinc binding inhibitors of MMP13. Bioorg. Med. Chem. Lett. 21, 4215–4219 (2011).
Gege, C. et al. Discovery and evaluation of a non-Zn chelating, selective matrix metalloproteinase 13 (MMP-13) inhibitor for potential intra-articular treatment of osteoarthritis. J. Med. Chem. 55, 709–716 (2012).
Settle, S. et al. Cartilage degradation biomarkers predict efficacy of a novel, highly selective matrix metalloproteinase 13 inhibitor in a dog model of osteoarthritis: confirmation by multivariate analysis that modulation of type II collagen and aggrecan degradation peptides parallels pathologic changes. Arthritis Rheum. 62, 3006–3015 (2010).
Krzeski, P. et al. Development of musculoskeletal toxicity without clear benefit after administration of PG-116800, a matrix metalloproteinase inhibitor, to patients with knee osteoarthritis: a randomized, 12-month, double-blind, placebo-controlled study. Arthritis Res. Ther. 9, R109 (2007).
Glasson, S. S. et al. Deletion of active ADAMTS5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 434, 644–648 (2005).
Stanton, H. et al. ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 434, 648–652 (2005).
Gardiner, M. D. et al. Transcriptional analysis of micro-dissected articular cartilage in post-traumatic murine osteoarthritis. Osteoarthr. Cartil. 23, 616–628 (2015).
Yang, C. Y., Chanalaris, A. & Troeberg, L. ADAMTS and ADAM metalloproteinases in osteoarthritis - looking beyond the ‘usual suspects’. Osteoarthr. Cartil. 25, 1000–1009 (2017).
Dunn, S. L. et al. Gene expression changes in damaged osteoarthritic cartilage identify a signature of non-chondrogenic and mechanical responses. Osteoarthr. Cartil. 24, 1431–1440 (2016).
Djouad, F. et al. Microenvironmental changes during differentiation of mesenchymal stem cells towards chondrocytes. Arthritis Res. Ther. 9, R33 (2007).
Jay, G. D. & Waller, K. A. The biology of lubricin: near frictionless joint motion. Matrix Biol. 39, 17–24 (2014).
Marcelino, J. et al. CACP, encoding a secreted proteoglycan, is mutated in camptodactyly-arthropathy-coxa vara-pericarditis syndrome. Nat. Genet. 23, 319–322 (1999).
Bahabri, S. A. et al. The camptodactyly-arthropathy-coxa vara-pericarditis syndrome: clinical features and genetic map** to human chromosome 1. Arthritis Rheum. 41, 730–735 (1998).
Ciullini Mannurita, S. et al. CACP syndrome: identification of five novel mutations and of the first case of UPD in the largest European cohort. Eur. J. Hum. Genet. 22, 197–201 (2014).
Rhee, D. K. et al. The secreted glycoprotein lubricin protects cartilage surfaces and inhibits synovial cell overgrowth. J. Clin. Investig. 115, 622–631 (2005).
Ogawa, H., Kozhemyakina, E., Hung, H. H., Grodzinsky, A. J. & Lassar, A. B. Mechanical motion promotes expression of Prg4 in articular cartilage via multiple CREB-dependent, fluid flow shear stress-induced signaling pathways. Genes Dev. 28, 127–139 (2014).
Lefebvre, V. & Bhattaram, P. Prg4-expressing cells: articular stem cells or differentiated progeny in the articular chondrocyte lineage? Arthritis Rheumatol. 67, 1151–1154 (2015).
Krawetz, R. J. et al. Proteoglycan 4 (PRG4) treatment enhances wound closure and tissue regeneration. NPJ Regen. Med. 7, 32 (2022).
Alquraini, A. et al. The interaction of lubricin/proteoglycan 4 (PRG4) with toll-like receptors 2 and 4: an anti-inflammatory role of PRG4 in synovial fluid. Arthritis Res. Ther. 17, 353 (2015).
Liu, Z. et al. A dual role for NOTCH signaling in joint cartilage maintenance and osteoarthritis. Sci. Signal. 8, ra71 (2015).
Zanotti, S., Yu, J., Bridgewater, D., Wolf, J. M. & Canalis, E. Mice harboring a Hajdu Cheney Syndrome mutation are sensitized to osteoarthritis. Bone 114, 198–205 (2018).
Monteagudo, S. & Lories, R. J. A Notch in the joint that exacerbates osteoarthritis. Nat. Rev. Rheumatol. 14, 563–564 (2018).
Sugita, S. et al. Transcription factor Hes1 modulates osteoarthritis development in cooperation with calcium/calmodulin-dependent protein kinase 2. Proc. Natl Acad. Sci. USA 112, 3080–3085 (2015).
Venn, G., Billingham, M. E. & Hardingham, T. E. Increased proteoglycan synthesis in cartilage in experimental canine osteoarthritis does not reflect a permanent change in chondrocyte phenotype. Arthritis Rheum. 38, 525–532 (1995).
Abramoff, B. & Caldera, F. E. Osteoarthritis: pathology, diagnosis, and treatment options. Med Clin. North Am. 104, 293–311 (2020).
Krishnan, Y. & Grodzinsky, A. J. Cartilage diseases. Matrix Biol. 71–72, 51–69 (2018).
Kon, E. et al. Bone marrow lesions and subchondral bone pathology of the knee. Knee Surg. Sports Traumatol. Arthrosc. 24, 1797–1814 (2016).
Arden, N. K. et al. Non-surgical management of knee osteoarthritis: comparison of ESCEO and OARSI 2019 guidelines. Nat. Rev. Rheumatol. 17, 59–66 (2021).
Messier, S. P. et al. Intentional weight loss in overweight and obese patients with knee osteoarthritis: is more better? Arthritis Care Res. 70, 1569–1575 (2018).
Skou, S. T. & Roos, E. M. Physical therapy for patients with knee and hip osteoarthritis: supervised, active treatment is current best practice. Clin. Exp. Rheumatol. 37, 112–117 (2019).
Gay, C., Chabaud, A., Guilley, E. & Coudeyre, E. Educating patients about the benefits of physical activity and exercise for their hip and knee osteoarthritis. Systematic literature review. Ann. Phys. Rehabil. Med. 59, 174–183 (2016).
Uthman, O. A. et al. Exercise for lower limb osteoarthritis: systematic review incorporating trial sequential analysis and network meta-analysis. BMJ 347, f5555 (2013).
Bartels, E. M. et al. Aquatic exercise for the treatment of knee and hip osteoarthritis. Cochrane Database Syst. Rev. 3, Cd005523 (2016).
Vincent, K. R., Vasilopoulos, T., Montero, C. & Vincent, H. K. Eccentric and concentric resistance exercise comparison for knee osteoarthritis. Med Sci. Sports Exerc. 51, 1977–1986 (2019).
Nguyen, C., Lefèvre-Colau, M. M., Poiraudeau, S. & Rannou, F. Rehabilitation (exercise and strength training) and osteoarthritis: a critical narrative review. Ann. Phys. Rehabil. Med. 59, 190–195 (2016).
Goh, S. L. et al. Efficacy and potential determinants of exercise therapy in knee and hip osteoarthritis: a systematic review and meta-analysis. Ann. Phys. Rehabil. Med. 62, 356–365 (2019).
Smith, T. O. et al. Assistive devices, hip precautions, environmental modifications and training to prevent dislocation and improve function after hip arthroplasty. Cochrane Database Syst. Rev. 7, Cd010815 (2016).
Fernandes, L. et al. EULAR recommendations for the non-pharmacological core management of hip and knee osteoarthritis. Ann. Rheum. Dis. 72, 1125–1135 (2013).
Healy, A., Farmer, S., Pandyan, A. & Chockalingam, N. A systematic review of randomised controlled trials assessing effectiveness of prosthetic and orthotic interventions. PLoS One 13, e0192094 (2018).
Dantas, L. O., Osani, M. C. & Bannuru, R. R. Therapeutic ultrasound for knee osteoarthritis: a systematic review and meta-analysis with grade quality assessment. Braz. J. Phys. Ther. 25, 688–697 (2021).
Huang, Z. et al. Effectiveness of low-level laser therapy in patients with knee osteoarthritis: a systematic review and meta-analysis. Osteoarthr. Cartil. 23, 1437–1444 (2015).
Dias, J. M. et al. Hydrotherapy improves pain and function in older women with knee osteoarthritis: a randomized controlled trial. Braz. J. Phys. Ther. 21, 449–456 (2017).
Flynn, D. M. Chronic musculoskeletal pain: nonpharmacologic, noninvasive treatments. Am. Fam. Physician 102, 465–477 (2020).
Brosseau, L. et al. Thermotherapy for treatment of osteoarthritis. Cochrane Database Syst. Rev. 2003, Cd004522 (2003).
Hochberg, M. C. Osteoarthritis year 2012 in review: clinical. Osteoarthr. Cartil. 20, 1465–1469 (2012).
Corbett, M. S. et al. Acupuncture and other physical treatments for the relief of pain due to osteoarthritis of the knee: network meta-analysis. Osteoarthr. Cartil. 21, 1290–1298 (2013).
Kelly, R. B. & Willis, J. Acupuncture for pain. Am. Fam. Physician 100, 89–96 (2019).
Tu, J. F. et al. Efficacy of intensive acupuncture versus sham acupuncture in knee osteoarthritis: a randomized controlled trial. Arthritis Rheumatol. 73, 448–458 (2021).
Shi, G. X. et al. Effect of electro-acupuncture (EA) and manual acupuncture (MA) on markers of inflammation in knee osteoarthritis. J. Pain. Res. 13, 2171–2179 (2020).
Kolasinski, S. L. et al. 2019 American College of Rheumatology/arthritis foundation guideline for the management of osteoarthritis of the hand, hip, and knee. Arthritis Rheumatol. 72, 220–233 (2020).
Zhang, W. et al. OARSI recommendations for the management of hip and knee osteoarthritis, part II: OARSI evidence-based, expert consensus guidelines. Osteoarthr. Cartil. 16, 137–162 (2008).
Mao, L. et al. Targeted treatment for osteoarthritis: drugs and delivery system. Drug Deliv. 28, 1861–1876 (2021).
Hochberg, M. C. et al. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res. 64, 465–474 (2012).
Rannou, F., Pelletier, J. P. & Martel-Pelletier, J. Efficacy and safety of topical NSAIDs in the management of osteoarthritis: Evidence from real-life setting trials and surveys. Semin. Arthritis Rheum. 45, S18–S21 (2016).
Scanzello, C. R., Moskowitz, N. K. & Gibofsky, A. The post-NSAID era: what to use now for the pharmacologic treatment of pain and inflammation in osteoarthritis. Curr. Rheumatol. Rep. 10, 49–56 (2008).
Roberts, E. et al. Paracetamol: not as safe as we thought? A systematic literature review of observational studies. Ann. Rheum. Dis. 75, 552–559 (2016).
**e, P. et al. Safety, tolerability, and pharmacokinetics of ibuprofenamine hydrochloride spray (NSAIDs), a new drug for rheumatoid arthritis and osteoarthritis, in healthy chinese subjects. Drug Des. Dev. Ther. 15, 629–638 (2021).
Kloppenburg, M. Hand osteoarthritis-nonpharmacological and pharmacological treatments. Nat. Rev. Rheumatol. 10, 242–251 (2014).
Dougados, M. Why and how to use NSAIDs in osteoarthritis. J. Cardiovasc. Pharm. 47, S49–S54 (2006).
Bannwarth, B. Acetaminophen or NSAIDs for the treatment of osteoarthritis. Best. Pract. Res. Clin. Rheumatol. 20, 117–129 (2006).
D’Arcy, Y. et al. Treating osteoarthritis pain: mechanisms of action of acetaminophen, nonsteroidal anti-inflammatory drugs, opioids, and nerve growth factor antibodies. Postgrad. Med. 133, 879–894 (2021).
Zhu, X. et al. Comparative effectiveness of glucosamine, chondroitin, acetaminophen or celecoxib for the treatment of knee and/or hip osteoarthritis: a network meta-analysis. Clin. Exp. Rheumatol. 36, 595–602 (2018).
Deyle, G. D. et al. Physical therapy versus glucocorticoid injection for osteoarthritis of the knee. N. Engl. J. Med. 382, 1420–1429 (2020).
Block, J. A. Are intraarticular glucocorticoids safe in osteoarthritis? Arthritis Rheumatol. 74, 181–183 (2022).
Savvidou, O. et al. Glucocorticoid signaling and osteoarthritis. Mol. Cell Endocrinol. 480, 153–166 (2019).
Black, R. & Grodzinsky, A. J. Dexamethasone: chondroprotective corticosteroid or catabolic killer? Eur. Cell Mater. 38, 246–263 (2019).
Chen, L. et al. Long term usage of dexamethasone accelerating accelerates the initiation of osteoarthritis via enhancing chondrocyte apoptosis and the extracellular matrix calcification and apoptosis of chondrocytes. Int. J. Biol. Sci. 17, 4140–4153 (2021).
Zhou, J. et al. Treatment of hip osteoarthritis with glucocorticoids. Ann. Rheum. Dis. 78, e100 (2019).
Riis, R. G. C. et al. The effects of intra-articular glucocorticoids and exercise on pain and synovitis assessed on static and dynamic magnetic resonance imaging in knee osteoarthritis: exploratory outcomes from a randomized controlled trial. Osteoarthr. Cartil. 25, 481–491 (2017).
McAlindon, T. E. et al. Effect of intra-articular triamcinolone vs saline on knee cartilage volume and pain in patients with knee osteoarthritis: a randomized clinical trial. JAMA 317, 1967–1975 (2017).
Li, B. et al. Baicalein alleviates osteoarthritis by protecting subchondral bone, inhibiting angiogenesis and synovial proliferation. J. Cell Mol. Med. 25, 5283–5294 (2021).
Fransen, M. et al. Glucosamine and chondroitin for knee osteoarthritis: a double-blind randomised placebo-controlled clinical trial evaluating single and combination regimens. Ann. Rheum. Dis. 74, 851–858 (2015).
Clegg, D. O. et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N. Engl. J. Med. 354, 795–808 (2006).
**ong, F. et al. pH-responsive and hyaluronic acid-functionalized metal-organic frameworks for therapy of osteoarthritis. J. Nanobiotechnol. 18, 139 (2020).
Bruyère, O., Altman, R. D. & Reginster, J. Y. Efficacy and safety of glucosamine sulfate in the management of osteoarthritis: Evidence from real-life setting trials and surveys. Semin. Arthritis Rheum. 45, S12–S17 (2016).
Su, K. et al. Comparison of hyaluronic acid and PRP intra-articular injection with combined intra-articular and intraosseous PRP injections to treat patients with knee osteoarthritis. Clin. Rheumatol. 37, 1341–1350 (2018).
Tang, J. Z. et al. Platelet-rich plasma versus hyaluronic acid in the treatment of knee osteoarthritis: a meta-analysis. J. Orthop. Surg. Res. 15, 403 (2020).
Malemud, C. J. Cytokines as therapeutic targets for osteoarthritis. BioDrugs 18, 23–35 (2004).
Urech, D. M. et al. Anti-inflammatory and cartilage-protecting effects of an intra-articularly injected anti-TNF{alpha} single-chain Fv antibody (ESBA105) designed for local therapeutic use. Ann. Rheum. Dis. 69, 443–449 (2010).
Millward-Sadler, S. J. et al. Integrin-regulated secretion of interleukin 4: a novel pathway of mechanotransduction in human articular chondrocytes. J. Cell Biol. 145, 183–189 (1999).
Georgescu, L., Vakkalanka, R. K., Elkon, K. B. & Crow, M. K. Interleukin-10 promotes activation-induced cell death of SLE lymphocytes mediated by Fas ligand. J. Clin. Invest. 100, 2622–2633 (1997).
Nees, T. A. et al. Synovial cytokines significantly correlate with osteoarthritis-related knee pain and disability: inflammatory mediators of potential clinical relevance. J. Clin. Med. 8, 1343 (2019).
Han, W., Fan, S., Bai, X. & Ding, C. Strontium ranelate, a promising disease modifying osteoarthritis drug. Expert Opin. Investig. Drugs 26, 375–380 (2017).
Yu, H. et al. Strontium ranelate promotes chondrogenesis through inhibition of the Wnt/β-catenin pathway. Stem Cell Res. Ther. 12, 296 (2021).
Apostu, D. et al. Systemic drugs with impact on osteoarthritis. Drug Metab. Rev. 51, 498–523 (2019).
Sun, Q. et al. Parathyroid hormone attenuates osteoarthritis pain by remodeling subchondral bone in mice. Elife 10, e66532 (2021).
Neogi, T. et al. Observed efficacy and clinically important improvements in participants with osteoarthritis treated with subcutaneous tanezumab: results from a 56-week randomized NSAID-controlled study. Arthritis Res. Ther. 24, 78 (2022).
Wise, B. L., Seidel, M. F. & Lane, N. E. The evolution of nerve growth factor inhibition in clinical medicine. Nat. Rev. Rheumatol. 17, 34–46 (2021).
Schnitzer, T. J. & Marks, J. A. A systematic review of the efficacy and general safety of antibodies to NGF in the treatment of OA of the hip or knee. Osteoarthr. Cartil. 23, S8–S17 (2015).
Karsdal, M. A. et al. Serological biomarker profiles of rapidly progressive osteoarthritis in tanezumab-treated patients. Osteoarthr. Cartil. 27, 484–492 (2019).
Jayabalan, P. & Schnitzer, T. J. Tanezumab in the treatment of chronic musculoskeletal conditions. Expert Opin. Biol. Ther. 17, 245–254 (2017).
Oo, W. M. & Hunter, D. J. Nerve growth factor (NGF) inhibitors and related agents for chronic musculoskeletal pain: a comprehensive review. BioDrugs 35, 611–641 (2021).
Gondal, F. R., Bilal, J. & Kent Kwoh, C. Tanezumab for the treatment of osteoarthritis pain. Drugs Today 58, 187–200 (2022).
Paget, L. D. A. et al. Effect of platelet-rich plasma injections vs placebo on ankle symptoms and function in patients with ankle osteoarthritis: a randomized clinical trial. JAMA 326, 1595–1605 (2021).
Katz, J. N. Platelet-rich plasma for osteoarthritis and achilles tendinitis. JAMA 326, 2012–2014 (2021).
Belk, J. W. et al. Platelet-rich plasma versus hyaluronic acid for knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Am. J. Sports Med. 49, 249–260 (2021).
Pas, H. I. et al. Stem cell injections in knee osteoarthritis: a systematic review of the literature. Br. J. Sports Med. 51, 1125–1133 (2017).
Harrell, C. R., Markovic, B. S., Fellabaum, C., Arsenijevic, A. & Volarevic, V. Mesenchymal stem cell-based therapy of osteoarthritis: current knowledge and future perspectives. Biomed. Pharmacother. 109, 2318–2326 (2019).
Kim, Y. G., Choi, J. & Kim, K. Mesenchymal stem cell-derived exosomes for effective cartilage tissue repair and treatment of osteoarthritis. Biotechnol. J. 15, e2000082 (2020).
De Bari, C. & Roelofs, A. J. Stem cell-based therapeutic strategies for cartilage defects and osteoarthritis. Curr. Opin. Pharm. 40, 74–80 (2018).
Ng, J. et al. Stem cell-directed therapies for osteoarthritis: the promise and the practice. Stem Cells 38, 477–486 (2020).
Shapiro, S. A., Kazmerchak, S. E., Heckman, M. G., Zubair, A. C. & O’Connor, M. I. A prospective, single-blind, placebo-controlled trial of bone marrow aspirate concentrate for knee osteoarthritis. Am. J. Sports Med. 45, 82–90 (2017).
Pers, Y. M. et al. Adipose mesenchymal stromal cell-based therapy for severe osteoarthritis of the knee: a phase I dose-escalation trial. Stem Cells Transl. Med. 5, 847–856 (2016).
Kim, Y. S., Choi, Y. J. & Koh, Y. G. Mesenchymal stem cell implantation in knee osteoarthritis: an assessment of the factors influencing clinical outcomes. Am. J. Sports Med. 43, 2293–2301 (2015).
Chahal, J. et al. Bone marrow mesenchymal stromal cell treatment in patients with osteoarthritis results in overall improvement in pain and symptoms and reduces synovial inflammation. Stem Cells Transl. Med. 8, 746–757 (2019).
Zhu, Y. et al. Crosstalk between Smad2/3 and specific isoforms of ERK in TGF-β1-induced TIMP-3 expression in rat chondrocytes. J. Cell Mol. Med. 21, 1781–1790 (2017).
Kim, M. K. et al. A multicenter, double-blind, phase iii clinical trial to evaluate the efficacy and safety of a cell and gene therapy in knee osteoarthritis patients. Hum. Gene Ther. Clin. Dev. 29, 48–59 (2018).
Liddle, A. D., Pegg, E. C. & Pandit, H. Knee replacement for osteoarthritis. Maturitas 75, 131–136 (2013).
Dieppe, P. Management of osteoarthritis of the hip and knee joints. Curr. Opin. Rheumatol. 5, 487–493 (1993).
Sheikh, A. & Schweitzer, M. Pre- and postoperative assessment in joint preserving and replacing surgery. Rheum. Dis. Clin. North Am. 35, 651–673 (2009).
Katz, J. N., Brownlee, S. A. & Jones, M. H. The role of arthroscopy in the management of knee osteoarthritis. Best. Pract. Res. Clin. Rheumatol. 28, 143–156 (2014).
Li, M. H., **ao, R., Li, J. B. & Zhu, Q. Regenerative approaches for cartilage repair in the treatment of osteoarthritis. Osteoarthr. Cartil. 25, 1577–1587 (2017).
Lo, C. W. T. et al. Risk factors for falls in patients with total hip arthroplasty and total knee arthroplasty: a systematic review and meta-analysis. Osteoarthr. Cartil. 27, 979–993 (2019).
Jayakumar, P. et al. Comparison of an artificial intelligence-enabled patient decision aid vs educational material on decision quality, shared decision-making, patient experience, and functional outcomes in adults with knee osteoarthritis: a randomized clinical trial. JAMA Netw. Open 4, e2037107 (2021).
Nishiwaki, T. et al. Pelvic tilt displacement before and after artificial hip joint replacement surgery. J. Arthroplast. 33, 925–930 (2018).
Acknowledgements
We apologize to the authors whose relevant studies have not been included in this review article. This work was supported, in part, by the National Key Research and Development Program of China Grants (2019YFA0906004), the National Natural Science Foundation of China Grants (82230081, 82250710175, 82172375, 81991513, and 82261160395), the Shenzhen Stable Support Plan Program for Higher Education Institutions (20200925150409001), the Shenzhen Fundamental Research Program (JCYJ20220818100617036), the Shenzhen Key Laboratory of Cell Microenvironment Grant (ZDSYS20140509142721429) and the Guangdong Provincial Science and Technology Innovation Council Grant (2017B030301018).
Author information
Authors and Affiliations
Contributions
Q.Y. and G.X. conceptualized and wrote the outline of the manuscript. Q.Y., X.W., C.T., W.G., M.C., M.Q., Y.Z., T.H., and S.C. did the literature search and wrote the manuscript draft. G.X. reviewed and edited the manuscript. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yao, Q., Wu, X., Tao, C. et al. Osteoarthritis: pathogenic signaling pathways and therapeutic targets. Sig Transduct Target Ther 8, 56 (2023). https://doi.org/10.1038/s41392-023-01330-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41392-023-01330-w
- Springer Nature Limited
This article is cited by
-
M2 macrophage-derived exosomal miR-26b-5p regulates macrophage polarization and chondrocyte hypertrophy by targeting TLR3 and COL10A1 to alleviate osteoarthritis
Journal of Nanobiotechnology (2024)
-
Emerging technology has a brilliant future: the CRISPR-Cas system for senescence, inflammation, and cartilage repair in osteoarthritis
Cellular & Molecular Biology Letters (2024)
-
Identification of therapeutic targets in osteoarthritis by combining heterogeneous transcriptional datasets, drug-induced expression profiles, and known drug-target interactions
Journal of Translational Medicine (2024)
-
Zhuifeng Tougu capsules in the treatment of knee osteoarthritis (cold dampness obstruction syndrome): a randomized, double blind, multicenter clinical study
Chinese Medicine (2024)
-
Gentiopicroside ameliorates the lipopolysaccharide-induced inflammatory response and hypertrophy in chondrocytes
Journal of Orthopaedic Surgery and Research (2024)