Abstract
It is well documented that hypoxia activates the hypoxia-inducible factor 1-alpha (HIF1α)/vascular endothelial growth factor A (VEGFA) axis to promote angiogenesis in breast cancer. However, it is unclear how this axis is negatively regulated. In this study, we demonstrated that miR-153 directly inhibits expression of HIF1α by binding to the 3′UTR of HIF1A mRNA, as well as suppresses tube formation of primary human umbilical vein endothelial cells (HUVECs) and breast cancer angiogenesis by decreasing the secretion of VEGFA. Importantly, expression of miR-153 was induced by hypoxia-stimulated ER stress, which activates IRE1α and its downstream transcription factor X-box binding protein 1 (XBP1). X-box binding protein 1 directly binds to the promoter of the miR-153 host gene PTPRN and activates transcription. These results indicate that hypoxia induces miR-153 to fine tune the HIF1α/VEGFA axis in breast cancer angiogenesis and miR-153 could be used for breast cancer anti-angiogenesis therapy.
Similar content being viewed by others
Introduction
Angiogenesis is a physiological process that form new vessels from pre-existing ones [1, 2], which is critical for solid tumor growth, invasion, and metastasis [1, 3]. Vascular endothelial growth factor A (VEGFA) is one of the most important proangiogenic factors secreted by tumor cells [4, 5]. The expression of VEGFA is directly induced by hypoxia-inducible factor 1-alpha (HIF1α) at the transcriptional level under hypoxia [6, 7]. Bevacizumab, a monoclonal antibody against VEGFA, became the first commercially available anti-angiogenesis drug in multiple solid tumors, although it was prohibited for use for treating metastatic breast cancer by the US FDA in 2011.
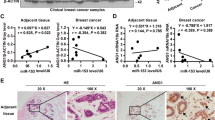
MicroRNAs (miRNAs) are small RNA molecules that silences gene expression through binding to the 3′-UTR of gene’s mRNA [8,9,10]. A number of studies suggest that miR-153 inhibits tumor growth and metastasis by targeting the snail family transcription repressor 1 (SNAI1), the zinc finger E-box binding homeobox 2 (ZEB2) [11], the metadherin (MTDH) [12], the ADAM metallopeptidase domain 19 (ADAM19) [13], the AKT serine/threonine kinase 1 (AKT1) [14], the HECT domain E3 ubiquitin protein ligase 3 (HECTD3) [15], and the BCL2 family apoptosis regulator (MCL-1) [d-luciferin (D12505, Bridgen, Bei**g, China) through intraperitoneal injection. After 5 min, it was anesthetized with ketamine and xylazine. The bioluminescent images were collected by using the system of IVIS Lumina XR (Caliper life Sciences, USA). Each tumor tissue was divided into two parts, one is used to purify the total RNA, the other is used for immunohistochemistry. Some tumors were only used in immunohistochemistry for their too small sizes. For the results of qPCR, the sample will be excluded if its value is higher or lower than the mean ± 3 × s.d.
Immunohistochemistry for CD31
The xenograft tumor tissues were fixed in 3.7% formalin solution. The immunohistochemistry assay was performed on 4-μm-thick paraffin sections after the pressure-cooking for antigen retrieval. The anti-CD31 primary antibody (1:400, Abcam, ab28364) was used. Subsequently, we incubated the slides with the anti-mouse/rabbit ultra sensitive polymer system (PV-8000, ZSGB-BIO, Bei**g, China). Signals for all slides were visualized by DAB staining. The slides were mounted after hematoxylin staining. Ten image fields for each slide, except for those are too small, were randomly collected by the persons who did not participate in this study by using microscopy, and the number of microvessel with positive CD31 expression was counted.
Chromatin immunoprecipitation assays
After hypoxia treatment, the diluted DNA–protein complex derived from MCF10A cells was incubated with the rabbit IgG or the anti-XBP1 antibody (Santa Cruz) in 1.5 ml centrifuge tube for 4 h at 4 °C. Then, each tube of sample was added with herring sperm DNA and protein A/G PLUS-Agarose (sc-2003, Santa Cruz), and continued to incubate overnight at 4 °C. Chromosomal DNA was amplified by PCR after purification. The PCR primers for amplifying the interest region on the PTPRN gene promoter (–2238 to –2225 from ATG) were as follows: 5′-GGACTACAGGTACGTGTTAC-3′ and 5′-GAGCCCAGGAGATTGAGA-3′.
Statistical analysis
All data in this study were analyzed by using the software of SPSS 18.0 (SPSS Inc., USA). Each experiment was performed at least three times, and the results are represented as the means ± s.d. The differences between two groups were analyzed by using the two-sided t-test, and the variance is similar between the groups, which are being statistically compared. The value of P smaller than 0.05 were considered statistically significant.
References
Folkman J. How is blood vessel growth regulated in normal and neoplastic tissue? G.H.A. Clowes memorial Award lecture. Cancer Res. 1986;46:467–73.
Urra H, Hetz C. A novel ER stress-independent function of the UPR in angiogenesis. Mol Cell. 2014;54:542–4.
Albini A, Indraccolo S, Noonan DM, Pfeffer U. Functional genomics of endothelial cells treated with anti-angiogenic or angiopreventive drugs. Clin Exp Metastas-. 2010;27:419–39.
Senger DR, Galli SJ, Dvorak AM, Perruzzi CA, Harvey VS, Dvorak HF. Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Sci (New Y, NY). 1983;219:983–5.
Senger DR, Van de Water L, Brown LF, Nagy JA, Yeo KT, Yeo TK, et al. Vascular permeability factor (VPF, VEGF) in tumor biology. Cancer Metastas- Rev. 1993;12:303–24.
Hirota K, Semenza GL. Regulation of angiogenesis by hypoxia-inducible factor 1. Crit Rev Oncol Hematol. 2006;59:15–26.
Pugh CW, Ratcliffe PJ. Regulation of angiogenesis by hypoxia: role of the HIF system. Nat Med. 2003;9:677–84.
Kim VN, Han J, Siomi MC. Biogenesis of small RNAs in animals. Nat Rev Mol Cell Biol. 2009;10:126–39.
Kota J, Chivukula RR, O’Donnell KA, Wentzel EA, Montgomery CL, Hwang HW, et al. Therapeutic microRNA delivery suppresses tumorigenesis in a murine liver cancer model. Cell. 2009;137:1005–17.
Lee YS, Nakahara K, Pham JW, Kim K, He Z, Sontheimer EJ, et al. Distinct roles for Drosophila Dicer-1 and Dicer-2 in the siRNA/miRNA silencing pathways. Cell. 2004;117:69–81.
Xu Q, Sun Q, Zhang J, Yu J, Chen W, Zhang Z. Downregulation of miR-153 contributes to epithelial-mesenchymal transition and tumor metastasis in human epithelial cancer. Carcinogenesis. 2013;34:539–49.
Li W, Zhai L, Zhao C, Lv S. MiR-153 inhibits epithelial-mesenchymal transition by targeting metadherin in human breast cancer. Breast Cancer Res Treat. 2015;150:501–9.
Shan N, Shen L, Wang J, He D, Duan C. MiR-153 inhibits migration and invasion of human non-small-cell lung cancer by targeting ADAM19. Biochem Biophys Res Commun. 2015;456:385–91.
Yuan Y, Du W, Wang Y, Xu C, Wang J, Zhang Y, et al. Suppression of AKT expression by miR-153 produced anti-tumor activity in lung cancer. Int J Cancer. 2015;136:1333–40.
Wu X, Li L, Li Y, Liu Z. MiR-153 promotes breast cancer cell apoptosis by targeting HECTD3. Am J Cancer Res. 2016;6:1563–71.
Zou Y, Liu W, Zhang J, **ang D. miR-153 regulates apoptosis and autophagy of cardiomyocytes by targeting Mcl-1. Mol Med Rep. 2016;14:1033–9.
Liu R, Shi P, Nie Z, Liang H, Zhou Z, Chen W, et al. Mifepristone suppresses basal triple-negative breast cancer stem cells by down-regulating KLF5 expression. Growth. 2016;17:18.
Wu Z, He B, He J, Mao X. Upregulation of miR-153 promotes cell proliferation via downregulation of the phosphatase and tensin homolog (PTEN) tumor suppressor gene in human prostate cancer. Prostate. 2013;73:596–604.
Zhang L, Pickard K, Jenei V, Bullock MD, Bruce A, Mitter R, et al. miR-153 supports colorectal cancer progression via pleiotropic effects that enhance invasion and chemotherapeutic resistance. Cancer Res. 2013;73:6435–47.
Chen WJ, Zhang EN, Zhong ZK, Jiang MZ, Yang XF, Zhou DM, et al. MicroRNA-153 expression and prognosis in non-small cell lung cancer. Int J Clin Exp Pathol. 2015;8:8671–5.
Ghasemi A, Fallah S, Ansari M. MiR-153 as a tumor suppressor in glioblastoma multiforme is downregulated by DNA methylation. Clin Lab. 2016;62:573–80.
Xu J, Liao X, Wong C. Downregulations of B-cell lymphoma 2 and myeloid cell leukemia sequence 1 by microRNA 153 induce apoptosis in a glioblastoma cell line DBTRG-05MG. Int J Cancer. 2010;126:1029–35.
Bao B, Rodriguez-Melendez R, Zempleni J. Cytosine methylation in miR-153 gene promoters increases the expression of holocarboxylase synthetase, thereby increasing the abundance of histone H4 biotinylation marks in HEK-293 human kidney cells. J Nutr Biochem. 2012;23:635–9.
Mandemakers W, Abuhatzira L, Xu H, Caromile LA, Hebert SS, Snellinx A, et al. Co-regulation of intragenic microRNA miR-153 and its host gene Ia-2 beta: identification of miR-153 target genes with functions related to IA-2beta in pancreas and brain. Diabetologia. 2013;56:1547–56.
Dioufa N, Kassi E, Papavassiliou AG, Kiaris H. Atypical induction of the unfolded protein response by mifepristone. Endocrine. 2010;38:167–73.
Wang Y, Gao L, Li Y, Chen H, Sun Z. Nifedipine protects INS-1 beta-cell from high glucose-induced ER stress and apoptosis. Int J Mol Sci. 2011;12:7569–80.
He X, Bi XY, Lu XZ, Zhao M, Yu XJ, Sun L, et al. Reduction of mitochondria-endoplasmic reticulum interactions by acetylcholine protects human umbilical vein endothelial cells from hypoxia/reoxygenation injury. Arter Throm Vas. 2015;35:1623–34.
Yang B, Xu Y, Hu Y, Luo Y, Lu X, Tsui CK, et al. Madecassic acid protects against hypoxia-induced oxidative stress in retinal microvascular endothelial cells via ROS-mediated endoplasmic reticulum stress. Biomed Pharmacother. 2016;84:845–52.
Boelens J, Lust S, Offner F, Bracke ME, Vanhoecke BW. Review. The endoplasmic reticulum: a target for new anticancer drugs. Vivo. 2007;21:215–26.
Boyce M, Yuan J. Cellular response to endoplasmic reticulum stress: a matter of life or death. Cell Death Differ. 2006;13:363–73.
Healy SJM, Gorman AM, Mousavi-Shafaei P, Gupta S, Samali A. Targeting the endoplasmic reticulum-stress response as an anticancer strategy. Eur J Pharmacol. 2009;625:234–46.
Chitnis N, Pytel D, Diehl JA. UPR-inducible miRNAs contribute to stressful situations. Trends Biochem Sci. 2013;38:447–52.
Chitnis NS, Pytel D, Bobrovnikova-Marjon E, Pant D, Zheng H, Maas NL, et al. miR-211 is a prosurvival microRNA that regulates chop expression in a PERK-dependent manner. Mol Cell. 2012;48:353–64.
Liu F, Liu B, Qian J, Wu G, Li J, Ma Z. miR-153 enhances the therapeutic effect of gemcitabine by targeting Snail in pancreatic cancer. Acta Biochim Biophys Sin (Shanghai). 2017;49:520–9.
Liu R, Shi P, Nie Z, Liang H, Zhou Z, Chen W, et al. Mifepristone suppresses basal triple-negative breast cancer stem cells by down-regulating KLF5 expression. Theranostics. 2016;6:533–44.
Wang Z, Liu C, MiR-153 regulates metastases of gastric cancer through Snail. Tumour Biol. 2015;37:15509–15.
Zeng HF, Yan S, Wu SF. MicroRNA-153-3p suppress cell proliferation and invasion by targeting SNAI1 in melanoma. Biochem Biophys Res Commun. 2017;487:140–5.
Li Q, Song W, Wang W, Yao S, Tian C, Cai X, et al. Suppression of epithelial-mesenchymal transition in hepatocellular carcinoma cells by Kruppel-like factor 4. Oncotarget. 2016;7:29749–60.
Zuo J, Wang D, Shen H, Liu F, Han J, Zhang X, MicroRNA-153 inhibits tumor progression in esophageal squamous cell carcinoma by targeting SNAI1. Tumour Biol. 2016;37:16135–40.
Yang W, Shen Y, Wei J, Liu F. MicroRNA-153/Nrf-2/GPx1 pathway regulates radiosensitivity and stemness of glioma stem cells via reactive oxygen species. Oncotarget. 2015;6:22006–27.
Bartoszewska S, Kochan K, Piotrowski A, Kamysz W, Ochocka RJ, Collawn JF, et al. The hypoxia-inducible miR-429 regulates hypoxia-inducible factor-1alpha expression in human endothelial cells through a negative feedback loop. FASEB J. 2015;29:1467–79.
Cha ST, Chen PS, Johansson G, Chu CY, Wang MY, Jeng YM, et al. MicroRNA-519c suppresses hypoxia-inducible factor-1alpha expression and tumor angiogenesis. Cancer Res. 2010;70:2675–85.
Dai L, Lou W, Zhu J, Zhou X, Di W. MiR-199a inhibits the angiogenic potential of endometrial stromal cells under hypoxia by targeting HIF-1alpha/VEGF pathway. Int J Clin Exp Pathol. 2015;8:4735–44.
Janaszak-Jasiecka A, Bartoszewska S, Kochan K, Piotrowski A, Kalinowski L, Kamysz W, et al. miR-429 regulates the transition between Hypoxia-Inducible Factor (HIF)1A and HIF3A expression in human endothelial cells. Sci Rep. 2016;6:22775.
Kang SG, Lee WH, Lee YH, Lee YS, Kim SG. Hypoxia-inducible factor-1alpha inhibition by a pyrrolopyrazine metabolite of oltipraz as a consequence of microRNAs 199a-5p and 20a induction. Carcinogenesis. 2012;33:661–9.
Wu Z, Cai X, Huang C, Xu J, Liu A. miR-497 suppresses angiogenesis in breast carcinoma by targeting HIF-1alpha. Oncol Rep. 2016;35:1696–702.
Chen X, Iliopoulos D, Zhang Q, Tang Q, Greenblatt MB, Hatziapostolou M, et al. XBP1 promotes triple-negative breast cancer by controlling the HIF1alpha pathway. Nature. 2014;508:103–7.
Paridaens A, Laukens D, Vandewynckel YP, Coulon S, Van Vlierberghe H, Geerts A, et al. Endoplasmic reticulum stress and angiogenesis: is there an interaction between them? Liver Int. 2014;34:e10–18.
Chen C, Sun X, Ran Q, Wilkinson KD, Murphy TJ, Simons JW, et al. Ubiquitin-proteasome degradation of KLF5 transcription factor in cancer and untransformed epithelial cells. Oncogene. 2005;24:3319–27.
Wu J, Ding Y, Chen CH, Zhou Z, Ding C, Chen H, et al. A new oridonin analog suppresses triple-negative breast cancer cells and tumor growth via the induction of death receptor 5. Cancer Lett. 2016;380:393–402.
Acknowledgements
We sincerely thank Professor Lingqiang Zhang from the Academy of Chinese Military Medical Sciences and Professor Wen Liu from **amen University for providing reagents. This study was supported by a Strategic Priority Research Program of the Chinese Academy of Sciences (XDA12010303), National Natural Science Foundation of China (31771516, U1502222, U1602221, 81772847, and 81660438), and the Yunnan Applied Basic Research Key Projects (2015FA027).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. If you remix, transform, or build upon this article or a part thereof, you must distribute your contributions under the same license as the original. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/.
About this article
Cite this article
Liang, H., **ao, J., Zhou, Z. et al. Hypoxia induces miR-153 through the IRE1α-XBP1 pathway to fine tune the HIF1α/VEGFA axis in breast cancer angiogenesis. Oncogene 37, 1961–1975 (2018). https://doi.org/10.1038/s41388-017-0089-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-017-0089-8
- Springer Nature Limited
This article is cited by
-
JAM3 promotes cervical cancer metastasis by activating the HIF-1α/VEGFA pathway
BMC Women's Health (2024)
-
Decoding contextual crosstalk: revealing distinct interactions between non-coding RNAs and unfolded protein response in breast cancer
Cancer Cell International (2024)
-
Endoplasmic reticulum stress: a novel targeted approach to repair bone defects by regulating osteogenesis and angiogenesis
Journal of Translational Medicine (2023)
-
Protein disulfide-isomerase A4 confers glioblastoma angiogenesis promotion capacity and resistance to anti-angiogenic therapy
Journal of Experimental & Clinical Cancer Research (2023)
-
Sevoflurane Confers Protection Against the Malignant Phenotypes of Lung Cancer Cells via the microRNA-153-3p/HIF1α/KDM2B Axis
Biochemical Genetics (2023)