Abstract
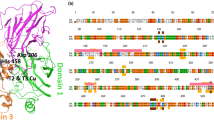
We have crystallized the ascomycete laccase from Melanocarpus albomyces with all four coppers present and determined the crystal structure at 2.4 Å resolution. The enzyme is heavily glycosylated and consists of three cupredoxin-like domains, similar to those found in the Cu-depleted basidiomycete laccase from Coprinus cinereus. However, there are significant differences in the loops forming the substrate-binding pocket. In addition, the crystal structure of the M. albomyces laccase revealed elongated electron density between all three coppers in the trinuclear copper site, suggesting that an oxygen molecule binds with a novel geometry. This oxygen, required in the reaction, may enter the trinuclear site through the tunnel, which is open in the structure of the C. cinereus laccase. In contrast, the C-terminus on the M. albomyces laccase forms a plug that blocks this access.



Similar content being viewed by others
References
Bao, W., O'Malley, D.M., Whetten, R. & Sederoff, R.R. Science 260, 672–674 (1993).
Thurston, C.F. Microbiology 140, 19–26 (1994).
Xu, F. in The Encyclopedia of Bioprocessing Technology: Fermentation, Biocatalysis and Bioseparation (eds Flickinger, M.C. & Drew, S.W.) 1545–1554 (John Wiley & Sons, New York; 1999).
Xu, F. Biochemistry 35, 7608–7614 (1996).
Xu, F. et al. Biochim. Biophys. Acta 1341, 303–311 (1996).
Messerschmidt, A. in Comprehensive Biological Catalysis Vol. III (ed. Sinnott, M.) 401–426 (Academic Press, London; 1997).
Solomon, E.I., Sundaram, U.M. & Machonkin, T.E. Chem. Rev. 96, 2563–2605 (1996).
Ducros, V. et al. Nature Struct. Biol. 5, 310–316 (1998).
Ducros, V. et al. Acta Crystallogr. D 57, 333–336 (2001).
Antorini, M. et al. Biochim. Biophys. Acta 1594, 109–114 (2002).
Bertrand, T. et al. Acta Crystallogr. D 58, 319–321 (2002).
Messerschmidt, A., Ladenstein, R. & Huber, R. J. Mol. Biol. 224, 179–205 (1992).
Messerschmidt, A., Luecke, H. & Huber, R. J. Mol. Biol. 230, 997–1014 (1993).
Zaitseva, I. et al. J. Biol. Inorg. Chem. 1, 15–23 (1996).
Huang, H., Zoppellaro, G. & Sakurai, T. J. Biol. Chem. 274, 32718–32724 (1999).
Sunduram, U.M., Zhang, H.H., Hedman, B., Hodgson, K.O. & Solomon, E.I. J. Am. Chem. Soc. 119, 12525–12540 (1997).
Lee, S.-K. et al. J. Am. Chem. Soc. 124, 6180–l6193 (2002).
Roberts, S.A. et al. Proc. Natl. Acad. Sci. USA 99, 2766–2771 (2002).
Magnus, K.A. et al. Proteins 19, 302–309 (1994).
Blackman, A.G. & Tolman, W.B. Struct. Bond. 97, 179–211 (2000).
Germann, U.A., Muller, G., Hunziker, P.E. & Lerch, K. J. Biol. Chem. 263, 885–896 (1988).
Berka, R.M. et al. Appl. Environ. Microbiol. 63, 3151–3157 (1997).
Gelo-Pujic, M., Kim, H.-H., Butlin, N.G. & Palmore, T.S. Appl. Environ. Microbiol. 65, 5515–5521 (1999).
Kiiskinen, L.-L., Viikari, L. & Kruus, K. Appl. Microbiol. Biotechnol. in the press(2002).
Stura, E.A. in Crystallization of Nucleic Acids and Proteins (eds Ducruix, A. & Giegé, R.) 177–208 (Oxford University Press, Oxford; 1999).
Otwinowski, Z. & Minor, W. Methods Enzymol. 276, 307–326 (1997).
Navaza, J. Acta Crystallogr. A 50, 157–163 (1994).
Beraman, H.M. et al. Nucleic Acids Res. 28, 235–242 (2000).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjeelgard, M. Acta Crystallogr. A 47, 110–119 (1991).
Brünger, A.T. et al. Acta Crystallogr. D 54, 905–921 (1998).
QUANTA 3.0 (Polygen Corporation, Massachusetts; 1990).
Evans, S.V. J. Mol. Graph. 11, 134–138 (1993).
Nicholls, A., Sharp, K.A. & Honig, B. Proteins 11, 281–296 (1991).
Murphy, M.E., Lindley, P.F. & Adman, E.T. Protein Sci. 6, 761–770 (1997).
Acknowledgements
This work was supported by TEKES (Structural Biology Research Program), the Academy of Finland (Finnish Centre of Excellence Programme (V.T.T.)) and the Neste Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Hakulinen, N., Kiiskinen, LL., Kruus, K. et al. Crystal structure of a laccase from Melanocarpus albomyces with an intact trinuclear copper site. Nat Struct Mol Biol 9, 601–605 (2002). https://doi.org/10.1038/nsb823
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb823
- Springer Nature America, Inc.
This article is cited by
-
Genome-wide study of Cerrena unicolor 87613 laccase gene family and their mode prediction in association with substrate oxidation
BMC Genomics (2023)
-
Insight of the optical property of laccase during polymerics formation for application in real-time biosensing
Journal of Materials Science (2021)
-
Crystal structures of multicopper oxidase CueO G304K mutant: structural basis of the increased laccase activity
Scientific Reports (2018)
-
Mechanism of salt-induced activity enhancement of a marine-derived laccase, Lac15
European Biophysics Journal (2018)