Key Points
-
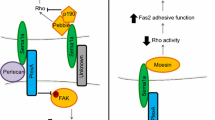
Semaphorins are secreted or transmembrane proteins that mediate repulsive axon guidance, immune cell regulation, and vascular growth and remodelling.
-
Semaphorins signal through plexins. Plexins, in turn, recruit and regulate kinases and Rho-family GTPases to control cell motility.
-
It has recently been shown that plexins also have intrinsic GAP (GTPase-activating protein) activity towards R-Ras on semaphorin binding. Plexin GAP activity decreases the activity of R-Ras.
-
Active R-Ras promotes integrin-mediated attachment to the extracellular matrix.
-
Semaphorins have been shown in many systems to decrease integrin-mediated attachment. It is now possible to attribute these phenomena to the regulation of R-Ras through plexins.
Abstract
Semaphorins are secreted or transmembrane proteins that regulate cell motility and attachment in axon guidance, vascular growth, immune cell regulation and tumour progression. The main receptors for semaphorins are plexins, which have established roles in regulating Rho-family GTPases. Recent work shows that plexins can also influence R-Ras, which, in turn, can regulate integrins. Such regulation is probably a common feature of semaphorin signalling and contributes substantially to our understanding of semaphorin biology.





Similar content being viewed by others
References
Comeau, M. R. et al. A poxvirus-encoded semaphorin induces cytokine production from monocytes and binds to a novel cellular semaphorin receptor, VESPR. Immunity 8, 473–482 (1998).
Winberg, M. L. et al. Plexin A is a neuronal semaphorin receptor that controls axon guidance. Cell 95, 903–916 (1998).
Tamagnone, L. et al. Plexins are a large family of receptors for transmembrane, secreted, and GPI-anchored semaphorins in vertebrates. Cell 99, 71–80 (1999).
Oinuma, I., Ishikawa, Y., Katoh, H. & Negishi, M. The Semaphorin 4D receptor Plexin-B1 is a GTPase activating protein for R-Ras. Science 305, 862–865 (2004). Establishes plexin GAP activity towards R-Ras in vitro and in cultured hippocampal neurons.
Pasterkamp, R. J. & Kolodkin, A. L. Semaphorin junction: making tracks toward neural connectivity. Curr. Opin. Neurobiol. 13, 79–89 (2003).
Semaphorin nomenclature committee. Unified nomenclature for the semaphorins/collapsins. Cell 97, 551–552 (1999).
Love, C. A. et al. The ligand-binding face of the semaphorins revealed by the high-resolution crystal structure of SEMA4D. Nature Struct. Biol. 10, 843–848 (2003).
Antipenko, A. et al. Structure of the semaphorin-3A receptor binding module. Neuron 39, 589–598 (2003).
Ohta, K. et al. Plexin: a novel neuronal cell surface molecule that mediates cell adhesion via a homophilic binding mechanism in the presence of calcium ions. Neuron 14, 1189–1199 (1995).
Tong, Y. & Buck, M. 1H, 15N and 13C resonance assignments and secondary structure determination reveal that the minimal Rac1 GTPase binding domain of plexin-B1 has a ubiquitin fold. J. Biomol. NMR 31, 369–370 (2005).
Takahashi, T. & Strittmatter, S. M. PlexinA1 autoinhibition by the plexin sema domain. Neuron 29, 429–439 (2001).
Artigiani, S. et al. Functional regulation of semaphorin receptors by proprotein convertases. J. Biol. Chem. 278, 10094–10101 (2003).
Kolodkin, A. L. et al. Neuropilin is a semaphorin III receptor. Cell 90, 753–762 (1997).
He, Z. & Tessier-Lavigne, M. Neuropilin is a receptor for the axonal chemorepellent Semaphorin III. Cell 90, 739–751 (1997).
Gu, C. et al. Semaphorin 3E and plexin-D1 control vascular pattern independently of neuropilins. Science 307, 265–268 (2005).
Takagi, S. et al. The A5 antigen, a candidate for the neuronal recognition molecule, has homologies to complement components and coagulation factors. Neuron 7, 295–307 (1991).
Mitsui, N. et al. Involvement of Fes/Fps tyrosine kinase in semaphorin3A signaling. EMBO J. 21, 3274–3285 (2002).
Gu, Y. & Ihara, Y. Evidence that collapsin response mediator protein-2 is involved in the dynamics of microtubules. J. Biol. Chem. 275, 17917–17920 (2000).
Sasaki, Y. et al. Fyn and Cdk5 mediate semaphorin-3A signaling, which is involved in regulation of dendrite orientation in cerebral cortex. Neuron 35, 907–920 (2002).
Lew, J. & Wang, J. H. Neuronal cdc2-like kinase. Trends Biochem. Sci. 20, 33–37 (1995).
Giordano, S. et al. The semaphorin 4D receptor controls invasive growth by coupling with Met. Nature Cell Biol. 4, 720–724 (2002).
Swiercz, J. M., Kuner, R. & Offermanns, S. Plexin-B1/RhoGEF-mediated RhoA activation involves the receptor tyrosine kinase ErbB-2. J. Cell Biol. 165, 869–880 (2004).
Winberg, M. L. et al. The transmembrane protein Off-track associates with Plexins and functions downstream of Semaphorin signaling during axon guidance. Neuron 32, 53–62 (2001).
Toyofuku, T. et al. Dual roles of Sema6D in cardiac morphogenesis through region-specific association of its receptor, Plexin-A1, with off-track and vascular endothelial growth factor receptor type 2. Genes Dev. 18, 435–447 (2004).
Campbell, D. S. & Holt, C. E. Apoptotic pathway and MAPKs differentially regulate chemotropic responses of retinal growth cones. Neuron 37, 939–952 (2003).
Pasterkamp, R. J., Peschon, J. J., Spriggs, M. K. & Kolodkin, A. L. Semaphorin 7A promotes axon outgrowth through integrins and MAPKs. Nature 424, 398–405 (2003). Shows that integrins mediate Sema7A signalling, and contributes to the growing body of work indicating activation of ERK/MAPK downstream of axon guidance molecules.
Hu, H., Marton, T. F. & Goodman, C. S. Plexin B mediates axon guidance in Drosophila by simultaneously inhibiting active Rac and enhancing RhoA signaling. Neuron 32, 39–51 (2001).
Swiercz, J. M., Kuner, R., Behrens, J. & Offermanns, S. Plexin-B1 directly interacts with PDZ-RhoGEF/LARG to regulate RhoA and growth cone morphology. Neuron 35, 51–63 (2002).
Aurandt, J., Vikis, H. G., Gutkind, J. S., Ahn, N. & Guan, K. L. The semaphorin receptor plexin-B1 signals through a direct interaction with the Rho-specific nucleotide exchange factor, LARG. Proc. Natl Acad. Sci. USA 99, 12085–12090 (2002).
Perrot, V., Vazquez-Prado, J. & Gutkind, J. S. Plexin B regulates Rho through the guanine nucleotide exchange factors leukemia-associated Rho GEF (LARG) and PDZ-RhoGEF. J. Biol. Chem. 277, 43115–43120 (2002).
Turner, L. J., Nicholls, S. & Hall, A. The activity of the plexin-A1 receptor is regulated by Rac. J. Biol. Chem. 279, 33199–33205 (2004).
Vikis, H. G., Li, W., He, Z. & Guan, K. L. The semaphorin receptor plexin-B1 specifically interacts with active Rac in a ligand-dependent manner. Proc. Natl Acad. Sci. USA 97, 12457–12462 (2000).
Vikis, H. G., Li, W. & Guan, K. L. The plexin-B1/Rac interaction inhibits PAK activation and enhances Sema4D ligand binding. Genes Dev. 16, 836–845 (2002).
**, Z. & Strittmatter, S. M. Rac1 mediates collapsin-1-induced growth cone collapse. J. Neurosci. 17, 6256–6263 (1997).
Kuhn, T. B., Brown, M. D., Wilcox, C. L., Raper, J. A. & Bamburg, J. R. Myelin and collapsin-1 induce motor neuron growth cone collapse through different pathways: inhibition of collapse by opposing mutants of rac1. J. Neurosci. 19, 1965–1975 (1999).
Zanata, S. M., Hovatta, I., Rohm, B. & Puschel, A. W. Antagonistic effects of Rnd1 and RhoD GTPases regulate receptor activity in Semaphorin 3A-induced cytoskeletal collapse. J. Neurosci. 22, 471–477 (2002).
Rohm, B., Rahim, B., Kleiber, B., Hovatta, I. & Puschel, A. W. The semaphorin 3A receptor may directly regulate the activity of small GTPases. FEBS Lett. 486, 68–72 (2000).
Wennerberg, K. et al. Rnd proteins function as RhoA antagonists by activating p190 RhoGAP. Curr. Biol. 13, 1106–1115 (2003).
Oinuma, I., Katoh, H., Harada, A. & Negishi, M. Direct interaction of Rnd1 with Plexin-B1 regulates PDZ-RhoGEF-mediated Rho activation by Plexin-B1 and induces cell contraction in COS-7 cells. J. Biol. Chem. 278, 25671–25677 (2003).
Driessens, M. H. et al. Plexin-B semaphorin receptors interact directly with active Rac and regulate the actin cytoskeleton by activating Rho. Curr. Biol. 11, 339–344 (2001).
Barberis, D. et al. Plexin signaling hampers integrin-based adhesion, leading to Rho-kinase independent cell rounding, and inhibiting lamellipodia extension and cell motility. FASEB J. 18, 592–594 (2004). Implicates integrin regulation as a key feature of plexin signalling, prefiguring the results of Ref. 4.
Guan, K. L. & Rao, Y. Signalling mechanisms mediating neuronal responses to guidance cues. Nature Rev. Neurosci. 4, 941–956 (2003).
Oinuma, I., Katoh, H. & Negishi, M. Molecular dissection of the semaphorin 4D receptor plexin-B1-stimulated R-Ras GTPase-activating protein activity and neurite remodeling in hippocampal neurons. J. Neurosci. 24, 11473–11480 (2004).
Lowe, D. G. et al. Structure of the human and murine R-ras genes, novel genes closely related to ras proto-oncogenes. Cell 48, 137–146 (1987).
Kinbara, K., Goldfinger, L. E., Hansen, M., Chou, F. L. & Ginsberg, M. H. Ras GTPases: integrins' friends or foes? Nature Rev. Mol. Cell Biol. 4, 767–776 (2003). An extremely useful review on Ras GTPases, including R-Ras, and the regulation of integrins.
Self, A. J., Caron, E., Paterson, H. F. & Hall, A. Analysis of R-Ras signalling pathways. J. Cell Sci. 114, 1357–1366 (2001).
Keely, P. J., Rusyn, E. V., Cox, A. D. & Parise, L. V. R-Ras signals through specific integrin a cytoplasmic domains to promote migration and invasion of breast epithelial cells. J. Cell Biol. 145, 1077–1088 (1999).
Serini, G. et al. Class 3 semaphorins control vascular morphogenesis by inhibiting integrin function. Nature 424, 391–397 (2003). Provides evidence that integrin regulation is downstream of class 3 semaphorins. Also establishes a role for semaphorins in vascular remodelling using retroviruses to overexpress wild-type semaphorins and dominant-negative semaphorin receptors in develo** chicks.
Fiore, R. & Puschel, A. W. The function of semaphorins during nervous system development. Front. Biosci. 8, S484–S499 (2003).
Huber, A. B., Kolodkin, A. L., Ginty, D. D. & Cloutier, J. F. Signaling at the growth cone: ligand–receptor complexes and the control of axon growth and guidance. Annu. Rev. Neurosci. 26, 509–563 (2003). Comprehensive and authoritative resource for semaphorins and other axon guidance molecules in the nervous system.
Goldberg, J. L. et al. An oligodendrocyte lineage-specific semaphorin, Sema5A, inhibits axon growth by retinal ganglion cells. J. Neurosci. 24, 4989–4999 (2004).
Liu, Y. et al. Semaphorin3D guides retinal axons along the dorsoventral axis of the tectum. J. Neurosci. 24, 310–318 (2004).
**ao, T., Shoji, W., Zhou, W., Su, F. & Kuwada, J. Y. Transmembrane sema4E guides branchiomotor axons to their targets in zebrafish. J. Neurosci. 23, 4190–4198 (2003).
Sahay, A., Molliver, M. E., Ginty, D. D. & Kolodkin, A. L. Semaphorin 3F is critical for development of limbic system circuitry and is required in neurons for selective CNS axon guidance events. J. Neurosci. 23, 6671–6680 (2003).
Taniguchi, M. et al. Distorted odor maps in the olfactory bulb of semaphorin 3A-deficient mice. J. Neurosci. 23, 1390–1397 (2003).
Dent, E. W., Barnes, A. M., Tang, F. & Kalil, K. Netrin-1 and semaphorin 3A promote or inhibit cortical axon branching, respectively, by reorganization of the cytoskeleton. J. Neurosci. 24, 3002–3012 (2004).
Bagri, A., Cheng, H. J., Yaron, A., Pleasure, S. J. & Tessier-Lavigne, M. Stereotyped pruning of long hippocampal axon branches triggered by retraction inducers of the semaphorin family. Cell 113, 285–299 (2003).
Terman, J. R., Mao, T., Pasterkamp, R. J., Yu, H. H. & Kolodkin, A. L. MICALs, a family of conserved flavoprotein oxidoreductases, function in plexin-mediated axonal repulsion. Cell 109, 887–900 (2002).
Piper, M., Salih, S., Weinl, C., Holt, C. E. & Harris, W. A. Endocytosis-dependent desensitization and protein synthesis-dependent resensitization in retinal growth cone adaptation. Nature Neurosci. 8, 179–186 (2005).
Fournier, A. E. et al. Semaphorin3A enhances endocytosis at sites of receptor-F-actin colocalization during growth cone collapse. J. Cell Biol. 149, 411–422 (2000).
Castellani, V., Falk, J. & Rougon, G. Semaphorin3A-induced receptor endocytosis during axon guidance responses is mediated by L1 CAM. Mol. Cell Neurosci. 26, 89–100 (2004).
Jurney, W. M., Gallo, G., Letourneau, P. C. & McLoon, S. C. Rac1-mediated endocytosis during ephrin-A2- and semaphorin 3A-induced growth cone collapse. J. Neurosci. 22, 6019–6028 (2002).
Milner, R. & Campbell, I. L. The integrin family of cell adhesion molecules has multiple functions within the CNS. J. Neurosci. Res. 69, 286–291 (2002).
Grabham, P. W. & Goldberg, D. J. Nerve growth factor stimulates the accumulation of β1 integrin at the tips of filopodia in the growth cones of sympathetic neurons. J. Neurosci. 17, 5455–5465 (1997).
Ivins, J. K., Yurchenco, P. D. & Lander, A. D. Regulation of neurite outgrowth by integrin activation. J. Neurosci. 20, 6551–6560 (2000).
Ivins, J. K., Parry, M. K. & Long, D. A. A novel cAMP-dependent pathway activates neuronal integrin function in retinal neurons. J. Neurosci. 24, 1212–1216 (2004).
Carmeliet, P. Blood vessels and nerves: common signals, pathways and diseases. Nature Rev. Genet. 4, 710–720 (2003).
Gitler, A. D., Lu, M. M. & Epstein, J. A. PlexinD1 and semaphorin signaling are required in endothelial cells for cardiovascular development. Dev. Cell 7, 107–116 (2004). The study of plexin-D1−/− mice established a role for Plexin-D1 in heart development that is independent of a neural crest migration defect.
Torres-Vazquez, J. et al. Semaphorin–plexin signaling guides patterning of the develo** vasculature. Dev. Cell 7, 117–123 (2004). Establishes an analogous role for plexin signalling in the vasculature to that in the nervous system.
Bates, D. et al. Neurovascular congruence results from a shared patterning mechanism that utilizes Semaphorin3A and Neuropilin-1. Dev. Biol 255, 77–98 (2003).
Yee, C. S. et al. Molecular cloning, expression, and activity of zebrafish semaphorin Z1a. Brain Res. Bull. 48, 581–593 (1999).
Gu, C. et al. Neuropilin-1 conveys semaphorin and VEGF signaling during neural and cardiovascular development. Dev. Cell 5, 45–57 (2003). Convincingly and elegantly establishes binding between a class 3 semaphorin and plexin-D1 that is not mediated by neuropilin. This interaction restricts the intersomitic vasculature from entering the somites.
Feiner, L. et al. Targeted disruption of semaphorin 3C leads to persistent truncus arteriosus and aortic arch interruption. Development 128, 3061–3070 (2001).
Behar, O., Golden, J. A., Mashimo, H., Schoen, F. J. & Fishman, M. C. Semaphorin III is needed for normal patterning and growth of nerves, bones and heart. Nature 383, 525–528 (1996).
Toyofuku, T. et al. Guidance of myocardial patterning in cardiac development by Sema6D reverse signalling. Nature Cell Biol. 6, 1204–1211 (2004).
Francis, S. E. et al. Central roles of α5β1 integrin and fibronectin in vascular development in mouse embryos and embryoid bodies. Arterioscler. Thromb. Vasc. Biol. 22, 927–933 (2002).
Cole, A. L., Subbanagounder, G., Mukhopadhyay, S., Berliner, J. A. & Vora, D. K. Oxidized phospholipid-induced endothelial cell/monocyte interaction is mediated by a cAMP-dependent R-Ras/PI3-kinase pathway. Arterioscler. Thromb. Vasc. Biol. 23, 1384–1390 (2003).
Yamada, A. et al. Molecular cloning of a glycosylphosphat-idylinositol-anchored molecule CDw108. J. Immunol. 162, 4094–4100 (1999).
Walzer, T., Galibert, L., Comeau, M. R. & De Smedt, T. Plexin C1 engagement on mouse dendritic cells by viral semaphorin A39R induces actin cytoskeleton rearrangement and inhibits integrin-mediated adhesion and chemokine-induced migration. J. Immunol. 174, 51–59 (2005). Shows that a viral semaphorin can decrease integrin adhesion through a plexin receptor in the immune system.
Walzer, T., Galibert, L. & Smedt, T. D. Poxvirus semaphorin A39R inhibits phagocytosis by dendritic cells and neutrophils. Eur. J. Immunol. 35, 391–398 (2005).
Kikutani, H. & Kumanogoh, A. Semaphorins in interactions between T cells and antigen-presenting cells. Nature Rev. Immunol. 3, 159–167 (2003).
Kumanogoh, A. et al. Class IV semaphorin Sema4A enhances T-cell activation and interacts with Tim-2. Nature 419, 629–633 (2002).
Kumanogoh, A. et al. Identification of CD72 as a lymphocyte receptor for the class IV semaphorin CD100: a novel mechanism for regulating B cell signaling. Immunity 13, 621–631 (2000).
Kumanogoh, A. et al. Requirement for the lymphocyte semaphorin, CD100, in the induction of antigen-specific T cells and the maturation of dendritic cells. J. Immunol. 169, 1175–1181 (2002).
Wang, X. et al. Functional soluble CD100/Sema4D released from activated lymphocytes: possible role in normal and pathologic immune responses. Blood 97, 3498–3504 (2001).
Delaire, S. et al. Biological activity of soluble CD100. II. Soluble CD100, similarly to H-SemaIII, inhibits immune cell migration. J. Immunol. 166, 4348–4354 (2001).
Chabbert-de Ponnat, I. et al. Soluble CD100 functions on human monocytes and immature dendritic cells require plexin C1 and plexin B1, respectively. Int. Immunol. 17, 439–447 (2005).
Wong, A. W. et al. CIITA-regulated plexin-A1 affects T-cell–dendritic cell interactions. Nature Immunol. 4, 891–898 (2003). Indicates that plexins, in addition to other semaphorin receptors in the immune system, can regulate T-cell interactions with APCs.
Pribila, J. T., Quale, A. C., Mueller, K. L. & Shimizu, Y. Integrins and T cell-mediated immunity. Annu. Rev. Immunol. 22, 157–180 (2004).
Cannon, J. L. & Burkhardt, J. K. The regulation of actin remodeling during T-cell–APC conjugate formation. Immunol. Rev. 186, 90–99 (2002).
Caron, E., Self, A. J. & Hall, A. The GTPase Rap1 controls functional activation of macrophage integrin αMβ2 by LPS and other inflammatory mediators. Curr. Biol. 10, 974–978 (2000).
Sekido, Y. et al. Human semaphorins A(V) and IV reside in the 3p21.3 small cell lung cancer deletion region and demonstrate distinct expression patterns. Proc. Natl Acad. Sci. USA 93, 4120–4125 (1996).
Zabarovsky, E. R., Lerman, M. I. & Minna, J. D. Tumor suppressor genes on chromosome 3p involved in the pathogenesis of lung and other cancers. Oncogene 21, 6915–6935 (2002).
Tse, C., **ang, R. H., Bracht, T. & Naylor, S. L. Human Semaphorin 3B (SEMA3B) located at chromosome 3p21.3 suppresses tumor formation in an adenocarcinoma cell line. Cancer Res. 62, 542–546 (2002).
**ang, R. et al. Semaphorin 3F gene from human 3p21.3 suppresses tumor formation in nude mice. Cancer Res. 62, 2637–2643 (2002).
Tomizawa, Y. et al. Inhibition of lung cancer cell growth and induction of apoptosis after reexpression of 3p21.3 candidate tumor suppressor gene SEMA3B. Proc. Natl Acad. Sci. USA 98, 13954–13959 (2001).
Castro-Rivera, E., Ran, S., Thorpe, P. & Minna, J. D. Semaphorin 3B (SEMA3B) induces apoptosis in lung and breast cancer, whereas VEGF165 antagonizes this effect. Proc. Natl Acad. Sci. USA 101, 11432–11437 (2004).
Brambilla, E., Constantin, B., Drabkin, H. & Roche, J. Semaphorin SEMA3F localization in malignant human lung and cell lines: a suggested role in cell adhesion and cell migration. Am. J. Pathol. 156, 939–950 (2000).
Bielenberg, D. R. et al. Semaphorin 3F, a chemorepulsant for endothelial cells, induces a poorly vascularized, encapsulated, nonmetastatic tumor phenotype. J. Clin. Invest. 114, 1260–1271 (2004). The strongest of the investigations into the role of semaphorins in cancer, implying that semaphorins can decrease β 1 -integrin expression to block metastasis.
Rincon-Arano, H., Rosales, R., Mora, N., Rodriguez-Castaneda, A. & Rosales, C. R-Ras promotes tumor growth of cervical epithelial cells. Cancer 97, 575–585 (2003).
Yu, Y. & Feig, L. A. Involvement of R-Ras and Ral GTPases in estrogen-independent proliferation of breast cancer cells. Oncogene 21, 7557–7568 (2002).
Cox, A. D., Brtva, T. R., Lowe, D. G. & Der, C. J. R-Ras induces malignant, but not morphologic, transformation of NIH3T3 cells. Oncogene 9, 3281–3288 (1994).
Martin-Satue, M. & Blanco, J. Identification of semaphorin E gene expression in metastatic human lung adenocarcinoma cells by mRNA differential display. J. Surg. Oncol. 72, 18–23 (1999).
Christensen, C. R. et al. Transcription of a novel mouse semaphorin gene, M-semaH, correlates with the metastatic ability of mouse tumor cell lines. Cancer Res. 58, 1238–1244 (1998).
Guo, W. & Giancotti, F. G. Integrin signalling during tumour progression. Nature Rev. Mol. Cell Biol. 5, 816–826 (2004).
Hinck, L. The versatile roles of 'axon guidance' cues in tissue morphogenesis. Dev. Cell 7, 783–793 (2004).
Zou, J. X. et al. An Eph receptor regulates integrin activity through R-Ras. Proc. Natl Acad. Sci. USA 96, 13813–13818 (1999).
Kagoshima, M. & Ito, T. Diverse gene expression and function of semaphorins in develo** lung: positive and negative regulatory roles of semaphorins in lung branching morphogenesis. Genes Cells 6, 559–571 (2001).
Brown, C. B. et al. PlexinA2 and semaphorin signaling during cardiac neural crest development. Development 128, 3071–3080 (2001).
Giraudon, P. et al. Semaphorin CD100 from activated T lymphocytes induces process extension collapse in oligodendrocytes and death of immature neural cells. J. Immunol. 172, 1246–1255 (2004). An interesting paper indicating that crosstalk between semaphorin expressing T cells could infiltrate the nervous system to cause neuronal demyelination and death in neuroinflammatory diseases.
Bagnard, D., Lohrum, M., Uziel, D., Puschel, A. W. & Bolz, J. Semaphorins act as attractive and repulsive guidance signals during the development of cortical projections. Development 125, 5043–5053 (1998).
Fenstermaker, V., Chen, Y., Ghosh, A. & Yuste, R. Regulation of dendritic length and branching by semaphorin 3A. J. Neurobiol. 58, 403–412 (2004).
Polleux, F., Morrow, T. & Ghosh, A. Semaphorin 3A is a chemoattractant for cortical apical dendrites. Nature 404, 567–573 (2000).
Song, H. et al. Conversion of neuronal growth cone responses from repulsion to attraction by cyclic nucleotides. Science 281, 1515–1518 (1998).
Terman, J. R. & Kolodkin, A. L. Nervy links protein kinase A to plexin-mediated semaphorin repulsion. Science 303, 1204–1207 (2004).
Wolman, M. A., Liu, Y., Tawarayama, H., Shoji, W. & Halloran, M. C. Repulsion and attraction of axons by Semaphorin3D are mediated by different neuropilins in vivo. J. Neurosci. 24, 8428–8435 (2004).
Massey, V. Activation of molecular oxygen by flavins and flavoproteins. J. Biol. Chem. 269, 22459–22462 (1994).
Suzuki, T. et al. MICAL, a novel CasL interacting molecule, associates with vimentin. J. Biol. Chem. 277, 14933–14941 (2002).
Inagaki, S. et al. Sema4C, a transmembrane semaphorin, interacts with a post-synaptic density protein, PSD-95. J. Biol. Chem. 276, 9174–9181 (2001).
Schultze, W. et al. Semaphorin4F interacts with the synapse-associated protein SAP90/PSD-95. J. Neurochem. 78, 482–489 (2001).
Herold, C., Elhabazi, A., Bismuth, G., Bensussan, A. & Boumsell, L. CD100 is associated with CD45 at the surface of human T lymphocytes. Role in T cell homotypic adhesion. J. Immunol. 157, 5262–5268 (1996).
Elhabazi, A. et al. The human semaphorin-like leukocyte cell surface molecule CD100 associates with a serine kinase activity. J. Biol. Chem. 272, 23515–23520 (1997).
Klostermann, A., Lutz, B., Gertler, F. & Behl, C. The orthologous human and murine semaphorin 6A-1 proteins (SEMA6A-1/Sema6A-1) bind to the enabled/vasodilator-stimulated phosphoprotein-like protein (EVL) via a novel carboxyl-terminal zyxin-like domain. J. Biol. Chem. 275, 39647–39653 (2000).
Eckhardt, F. et al. A novel transmembrane semaphorin can bind c-src. Mol. Cell Neurosci. 9, 409–419 (1997).
Kikuchi, K., Ishida, H. & Kimura, T. Molecular cloning of a novel member of semaphorin family genes, semaphorin Z. Brain Res. Mol. Brain Res. 51, 229–237 (1997).
Godenschwege, T. A., Hu, H., Shan-Crofts, X., Goodman, C. S. & Murphey, R. K. Bi-directional signaling by Semaphorin 1a during central synapse formation in Drosophila. Nature Neurosci. 5, 1294–1301 (2002).
Yaron, A., Huang, P. H., Cheng, H. J. & Tessier-Lavigne, M. Differential requirement for plexin-A3 and-A4 in mediating responses of sensory and sympathetic neurons to distinct class 3 semaphorins. Neuron 45, 513–523 (2005).
Masuda, K. et al. Sema4D stimulates axonal outgrowth of embryonic DRG sensory neurones. Genes Cells 9, 821–829 (2004).
Artigiani, S. et al. Plexin-B3 is a functional receptor for semaphorin 5A. EMBO Rep. 5, 710–714 (2004).
Ice, R. J. et al. Comment on “Nervy links protein kinase A to plexin-mediated semaphorin repulsion” Science 309, 558 (2005).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- RHO-FAMILY GTPASES
-
Ras-related GTPases that are involved in controlling the dynamics of the actin cytoskeleton.
- IMMUNOGLOBULIN-LIKE DOMAIN
-
A protein domain composed of two β pleated sheets held together by a disulphide bond.
- THROMBOSPONDIN DOMAIN
-
A small three-stranded domain of ∼60 amino acids that has homology to thrombospondin and that is characterized by three conserved disulphide bonds.
- PDZ DOMAIN
-
(Postsynaptic-density protein of 95 kDa, Discs large, Zona occludens-1). A protein-interaction domain that often occurs in scaffolding proteins and is named after the founding members of this protein family.
- CONVERTASE-CLEAVAGE SITES
-
Sites of post-translational proteolytic cleavage by subtilisin or proteases that regulate the activity of secreted proteins.
- GROWTH CONE
-
The motile tip of growing axons that senses gradients of attractive and repulsive cues to direct motility.
- GAPS
-
Proteins that inactivate small GTP-binding proteins, such as Ras family members, by increasing their rate of GTP hydrolysis.
- ACTOMYOSIN CONTRACTILITY
-
The process underlying growth cone retraction whereby actin filaments interact with myosin to generate contractile force.
- STRESS FIBRES
-
Also termed 'actin-microfilament bundles', these are bundles of parallel filaments that contain F-actin and other contractile molecules, and often stretch between cell attachments as if under stress.
- BILATERIA
-
A subgroup of animals with bilateral symmetry during development, including worms, flies and humans. These animals have more complex nervous systems than the more distantly related (and radially symmetrical) sponges and jellyfish.
- EXTRACELLULAR MATRIX
-
(ECM). The complex, multi-molecular material that surrounds cells. The ECM comprises a scaffold on which tissues are organized, it provides cellular microenvironments and it regulates various cellular functions.
- DOMINANT NEGATIVE
-
A defective protein that retains interaction capabilities and therefore competes with normal proteins, thereby impairing normal protein function.
- MONOOXYGENASE DOMAIN
-
A protein domain that catalyses oxidoreduction reactions using a flavin cofactor.
- FOCAL ADHESIONS
-
Integrin-mediated cell–substrate adhesion structures that anchor the ends of actin filaments (stress fibres) and mediate strong attachments to substrates. They also function as an integrin signalling platform.
- LAMELLIPODIUM
-
A fan-shaped membrane extension in growth cones, which is more frequently observed at the edge facing the direction of growth.
- RGD SEQUENCE
-
The primary adhesive motif in many extracellular-matrix molecules, which contains the amino-acid triplet Arg–Gly–Asp.
- PLEXUS
-
A primitive vascular network characterized by a honeycomb appearance and connectivity.
- AUTOCRINE
-
Describing, or relating to, a cell that produces the ligands by which it is activated.
- PARACRINE
-
Describing, or relating to, a regulatory cell that secretes an agonist into intercellular spaces from which it diffuses to a target cell other than the one that produces it.
- ANGIOGENESIS
-
The growth and proliferation of new blood vessels from existing vasculature.
- NEURAL CREST CELLS
-
Cells that originate from the neural crest region of a vertebrate embryo. They migrate outward and are progenitors of mature cell types, including peripheral neurons.
- MYOCARDIUM
-
The muscle wall of the heart.
- COMPACT LAYER
-
The outer layer of the myocardium.
- BIDIRECTIONAL SIGNALLING
-
Occurs when a transmembrane protein stimulates a neighbouring cell as a ligand, and its own cell as a receptor.
- POSTSYNAPTIC DENSITY
-
Higher-order protein structure present in postsynaptic membranes that functions to concentrate neurotransmitter receptors.
- SRC-HOMOLOGY-3 (SH3) DOMAIN
-
A protein module of 80 amino acids that is present in a range of proteins and was first identified in the protein kinase Src. SH3 domains interact with proline-rich sequences that usually contain a PXXXPXR motif.
- TRABECULATION
-
The process of making ridges in the myocardium, caused by the perpendicular migration of myocardial cells.
- POXVIRUSES
-
A group of enveloped, DNA viruses that can cause pox diseases in vertebrates.
- MONOCYTES
-
Large leukocytes with a horseshoe-shaped nucleus. They derive from pluripotent stem cells and become phagocytic macrophages when they enter the tissues.
- DENDRITIC CELLS
-
Sentries of the immune system named for their extensive membrane extensions. They ingest antigens by phagocytosis, degrade them and present them to T cells as part of the adaptive immune response.
- PHAGOCYTOSIS
-
An actin-dependent process by which cells engulf external particulate material by extension and fusion of pseudopods.
- ANTIGEN-PRESENTING CELL
-
A cell, most often a macrophage or dendritic cell, that presents an antigen to activate a T cell.
- MAJOR HISTOCOMPATIBILITY COMPLEX
-
A complex of genetic loci in higher vertebrates that encodes a family of cellular antigens that allow the immune system to recognize self from non-self.
- LOSS OF HETEROZYGOSITY
-
Deletion or disruption of one of two copies of a gene.
- METASTASIS
-
The movement of cancerous cells from one area of the body to another.
- FASCICULATION
-
The bundling of axons.
- OLIGODENDROCYTE
-
A supporting cell in the nervous system that forms a myelin sheath around axons.
- EXTRAVASATION
-
The process of cancer- and immune-cell migration across the blood vessel endothelial layer into surrounding tissues.
Rights and permissions
About this article
Cite this article
Kruger, R., Aurandt, J. & Guan, KL. Semaphorins command cells to move. Nat Rev Mol Cell Biol 6, 789–800 (2005). https://doi.org/10.1038/nrm1740
Issue Date:
DOI: https://doi.org/10.1038/nrm1740
- Springer Nature Limited
This article is cited by
-
Regulation of Semaphorin3A in the process of cutaneous wound healing
Cell Death & Differentiation (2022)
-
MicroRNAs and Synaptic Plasticity: From Their Molecular Roles to Response to Therapy
Molecular Neurobiology (2022)
-
Quantitative modeling of regular retinal microglia distribution
Scientific Reports (2021)
-
MAOA promotes prostate cancer cell perineural invasion through SEMA3C/PlexinA2/NRP1–cMET signaling
Oncogene (2021)
-
Semaphorin 7A promotes endothelial to mesenchymal transition through ATF3 mediated TGF-β2/Smad signaling
Cell Death & Disease (2020)