Abstract
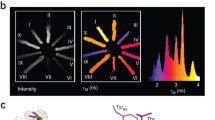
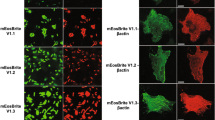
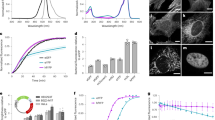
Monomeric (m)Eos2 is an engineered photoactivatable fluorescent protein widely used for super-resolution microscopy. We show that mEos2 forms oligomers at high concentrations and forms aggregates when labeling membrane proteins, limiting its application as a fusion partner. We solved the crystal structure of tetrameric mEos2 and rationally designed improved versions, mEos3.1 and mEos3.2, that are truly monomeric, are brighter, mature faster and exhibit higher photon budget and label density.


Similar content being viewed by others
Accession codes
References
Betzig, E. et al. Science 313, 1642–1645 (2006).
Hess, S.T., Girirajan, T.P. & Mason, M.D. Biophys. J. 91, 4258–4272 (2006).
Rust, M.J., Bates, M. & Zhuang, X. Nat. Methods 3, 793–795 (2006).
Huang, B., Bates, M. & Zhuang, X. Annu. Rev. Biochem. 78, 993–1016 (2009).
Wiedenmann, J. et al. Proc. Natl. Acad. Sci. USA 101, 15905–15910 (2004).
Lippincott-Schwartz, J. & Patterson, G.H. Trends Cell Biol. 19, 555–565 (2009).
McKinney, S.A., Murphy, C.S., Hazelwood, K.L., Davidson, M.W. & Looger, L.L. Nat. Methods 6, 131–133 (2009).
Nienhaus, G.U. et al. Photochem. Photobiol. 82, 351–358 (2006).
Hoi, H. et al. J. Mol. Biol. 401, 776–791 (2010).
Zacharias, D.A., Violin, J.D., Newton, A.C. & Tsien, R.Y. Science 296, 913–916 (2002).
Ando, R., Mizuno, H. & Miyawaki, A. Science 306, 1370–1373 (2004).
Fuchs, J. et al. Nat. Methods 7, 627–630 (2010).
Shroff, H., Galbraith, C.G., Galbraith, J.A. & Betzig, E. Nat. Methods 5, 417–423 (2008).
Jones, S.A., Shim, S.H., He, J. & Zhuang, X. Nat. Methods 8, 499–508 (2011).
Riedl, J. et al. Nat. Methods 5, 605–607 (2008).
Wiedenmann, J. et al. J. Biophotonics 4, 377–390 (2011).
Bai, L. et al. Cell Metab. 5, 47–57 (2007).
Chudakov, D.M. et al. Nat. Biotechnol. 22, 1435–1439 (2004).
Subach, O.M. et al. Nat. Methods 8, 771–777 (2011).
Shaner, N.C. et al. Nat. Methods 5, 545–551 (2008).
Ji, W. et al. Proc. Natl. Acad. Sci. USA 105, 13668–13673 (2008).
Li, Z. et al. J. Biol. Chem. 282, 29448–29456 (2007).
Xu, P. et al. Biochem. Biophys. Res. Commun. 350, 969–976 (2006).
Shroff, H. et al. Proc. Natl. Acad. Sci. USA 104, 20308–20313 (2007).
Olivo-Marin, J.C. Pattern Recognit. 35, 1989–1996 (2002).
Smith, C.S., Joseph, N., Rieger, B. & Lidke, K.A. Nat. Methods 7, 373–375 (2010).
Thompson, R.E., Larson, D.R. & Webb, W.W. Biophys. J. 82, 2775–2783 (2002).
Grotjohann, T. et al. Nature 478, 204–208 (2011).
Annibale, P., Scarselli, M., Kodiyan, A. & Radenovic, A. J. Phys. Chem. Lett. 1, 1506–1510 (2010).
Annibale, P., Vanni, S., Scarselli, M., Rothlisberger, U. & Radenovic, A. Nat. Methods 8, 527–528 (2011).
Acknowledgements
We thank L.L. Looger (Janelia Farm Research Campus) and Addgene for providing mEos2 cDNA, and X. Yu for providing technical support of analytical ultracentrifugation. This work was supported by grants from the Major State Basic Research Program of the People's Republic of China (2010CB833701 and 2010CB912303), the National Science Foundation of China (31130065, 31170818, 90913022 and 31127901), projects from Chinese Academy of Sciences (YZ200838, KSCX1-1W-J-3 and KSCX2-EW-Q-11), and the talent introduction program to Universities (B08029).
Author information
Authors and Affiliations
Contributions
M.Z., H.C., Y.Z., J.Y., W.J., J.C., B.L. and J.L. performed the research; L.W. and Y.L. assisted with data collection and solved the mEos2 structure; J.Z. assisted with spectrum measurement; M.Z., H.C., Y.Z., P.X. and T.X. analyzed data; T.X. and P.X. designed the research and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–17 and Supplementary Tables 1–7 (PDF 2305 kb)
Rights and permissions
About this article
Cite this article
Zhang, M., Chang, H., Zhang, Y. et al. Rational design of true monomeric and bright photoactivatable fluorescent proteins. Nat Methods 9, 727–729 (2012). https://doi.org/10.1038/nmeth.2021
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.2021
- Springer Nature America, Inc.
This article is cited by
-
Near-infrared PAINT localization microscopy via chromophore replenishment of phytochrome-derived fluorescent tag
Communications Biology (2024)
-
Bio-friendly long-term subcellular dynamic recording by self-supervised image enhancement microscopy
Nature Methods (2023)
-
Blue-shift photoconversion of near-infrared fluorescent proteins for labeling and tracking in living cells and organisms
Nature Communications (2023)
-
Spatially map** the diffusivity of proteins in live cells based on cumulative area analysis
Science China Chemistry (2023)
-
Optical control of ultrafast structural dynamics in a fluorescent protein
Nature Chemistry (2023)