Abstract
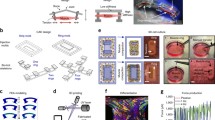
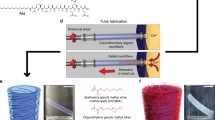
Current procedures for manual extraction of mature muscle tissue in micromechanical structures are time consuming and can damage the living components. To overcome these limitations, we have devised a new system for assembling muscle-powered microdevices based on judicious manipulations of materials phases and interfaces. In this system, individual cells grow and self-assemble into muscle bundles that are integrated with micromechanical structures and can be controllably released to enable free movement. Having realized such an assembly with cardiomyocytes we demonstrate two potential applications: a force transducer able to characterize in situ the mechanical properties of muscle and a self-assembled hybrid (biotic/abiotic) microdevice that moves as a consequence of collective cooperative contraction of muscle bundles. Because the fabrication of silicon microdevices is independent of the subsequent assembly of muscle cells, this system is highly versatile and may lead to the integration of cells and tissues with a variety of other microstructures.





Similar content being viewed by others
References
Soong, R. et al. Powering an inorganic nanodevice with a biomolecular motor. Science 290, 1555–1558 (2000).
Bachand, G. & Montemagno, C. Constructing organic/inorganic NEMS device powered by biomolecular motors. Biomed. Microdevices 2, 179–184 (2000).
Hess, H., Bachand, G. D. & Vogel, V. Powering nanodevices with biomolecular motors. Chem. Eur. J 10, 2110–2116 (2004).
Shu, D., Huang, L. P., Hoeprich, S. & Guo, P. Construction of phi 29 DNA-packing RNA monomers, dimers, and trimers with variables sizes and shapes as potential parts for nanodevices. J. Nanosci. Nanotechnol. 3, 295–302 (2003).
West, J. B. Physiological Basis of Medical Practice 12th edn (Williams & Wilkins, Baltimore, USA, 1990).
Herzog, W. Skeletal Muscle Mechanics (Wiley, New York, USA, 2000).
Lin, G., Pister, K. S. J. & Roos, K. P. Surface micromachined polysilicon heart cell force transducer. J. Microelectromech. Syst. 9, 9–17 (2000).
Lin, G., Palmer, R. E., Pister, K. S. J. & Roos, K. P. Miniature heart cell force transducer system implemented in MEMS technology. IEEE Trans. Biomed. Eng. 48, 996–1006 (2001).
McMahon, D. et al. C2C12 cells: biophysical, biochemical, and immunocytochemical properties. Am. Physiol. Soc. 35, C1795–C1802 (1994).
van der Velden, J. et al. Force production in mechanically isolated cardiac myocyte from human ventricular muscle tissue. Cardiovasc. Res. 38, 414–423 (1998).
Tashe, E., Meyhofer, E. & Brenner, B. A force transducer for measuring mechanical properties of single cardiac myocytes. Am. J. Physiol. 277, H2400 (1999).
Folch, A. & Toner, M. Microengineering of cellular interactions. Annu. Rev. Biomed. Eng. 2, 227–256 (2000).
Kane, R. S., Takayama, S., Ostuni, E., Ingber, D. E. & Whitesides, G. M. Patterning proteins and cells using soft lithography. Biomater. 20, 2363–2376 (1999).
Balaban, N. et al. Force and focal adhesion assembly: a close relationship studied using elastic micropatterned substrates. Nature Cell Biol. 3, 466–473 (2001).
Beningo, K., Dembo, M., Kaverina, I., Small, J. V. & Wang, Y. L. Nascent focal adhesion are responsible for the generation of strong propulsive forces in migrating fibroblasts. J. Cell Biol. 153, 881–887 (2001).
Galbraith, C. G. & Sheetz, M. P. A micromachined device provides a new bend on fibroblast traction forces. Proc. Natl Acad. Sci. USA 94, 9114–9118 (1997).
Tan, J. L. et al. Cells lying on a bed of microneedles: an approach to isolate mechanical force. Proc. Natl Acad. Sci. USA 100, 1484–1489 (2003).
MacDonald, N. C. SCREAM Microelectromechanical system. Microelectronic Eng. 32, 49–73 (1996).
Yamato, M., Konno, C., Utsumi, M., Kikuchi, A. & Okano, T. Thermally responsive polymer-grafted surfaces facilitate patterned cell seeding and co-culture. Biomater. 23, 561–567 (2002).
Okano, T., Yamada, N., Sakai, H. & Sakurai, Y. A novel recovery system for cultured cells using plasma-treated polystyrene dishes grafted with poly(N-isopropylacrylamide). J. Biomed. Mater. Res. 27, 1243–1251 (1993).
Yamato, M. et al. Thermo-responsive culture dishes allow the intact harvest of multilayered keratinocyte sheets without dispase by reducing temperature. Tissue Eng. 7, 473–480 (2001).
Chen, G., Ito, Y. & Imanishi, Y. Regulation of growth and adhesion of cultured cells by insulin conjugated with thermoresponsive polymers. Biotechnol. Bioeng. 53, 339–344 (1997).
Shimizu, T., Yamato, M., Kikuchi, A. & Okano, T. Two-dimensional manipulation of cardiac myocytes sheets utilizing temperature-responsive culture dishes augments the pulsatile amplitude. Tissue Eng. 7, 141–151 (2001).
Hu, Z., Chen, Y., Wang, C., Zheng, Y. & Li, Y. Polymer gels with engineered environmentally responsive surface patterns. Nature 393, 149–152 (1998).
Hollar, S., Flynn, A., Bergbreiter, S. & Pister, K. S. J. in Hilton Head 2002 Workshop (Transducer Research Foundation, Hilton Head Island, South Carolina, 2002).
Fearing, R. S. in IEEE Int. Conf. Robotics and Automation-Tutorial on Micro Mechanics and Micro Robotics (Leuven, Belgium,1998).
McMahon, A. T. & Bonner, J. T. On Size and Life (Scientific American, New York, USA, 1983).
**, J., Dy, E., Hung, M.-T. & Montemagno, C. in Nanotech 2004: NSTI Nanotechnology Conference and Trade Show 3–6 (Boston, USA, 2004).
Haddad, S. A. P., Gieltjes, S., Houben, R. P. M. & Serdijn, W. A. in Proc. IEEE International Symposium on Circuits and Systems (Bangkok, Thailand, 2003).
Hibbeler, R. C. Mechanics of Materials (Macmillan, London, 1991).
Ross, R. S. et al. ß1 integrins participate in the hypertrophic response of rat ventricular myocytes. Circulation Res. 82, 1160–1172 (1998).
Sen, A., Dunnmon, P., Henderson, S. A., Gerard, R. D. & Chien, K. R. Terminally differentiated neonatal rat myocardial cells proliferate and maintain specific differentiated functions following expression of SV40 large T antigen. J. Biol. Chem. 263, 19132–19136 (1988).
Acknowledgements
We thank Robert S. Ross, Jane Chen and Shane Antrobus at UCLA for the cells and Cassanna Ouellette for the graphic images. We thank Neves Hercules, Benjamin M. Wu, Chinh-Ming Ho, Toshikazu Hamasaki and Earl Homsher for suggestions of revising the paper. Supporter by Center for Cell Mimetic Space Exploration (CMISE), a NASA University Research, Engineering and Technology Institute (URETI), under award number NCC 2-1364.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
**, J., Schmidt, J. & Montemagno, C. Self-assembled microdevices driven by muscle. Nature Mater 4, 180–184 (2005). https://doi.org/10.1038/nmat1308
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat1308
- Springer Nature Limited
This article is cited by
-
Micro(bio)robotics: design and applications
Journal of Micro and Bio Robotics (2023)
-
The emerging technology of biohybrid micro-robots: a review
Bio-Design and Manufacturing (2022)
-
Biomimetic cell-actuated artificial muscle with nanofibrous bundles
Microsystems & Nanoengineering (2021)
-
Development of micro- and nanorobotics: A review
Science China Technological Sciences (2019)
-
Bio-inspired Hybrid Carbon Nanotube Muscles
Scientific Reports (2016)