Abstract
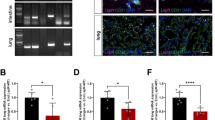
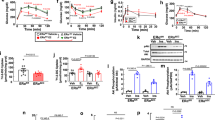
Low-density lipoprotein receptor-related protein 1 (LRP1) regulates lipid and glucose metabolism in liver and adipose tissue. It is also involved in central nervous system regulation of food intake and leptin signalling. Here we demonstrate that endothelial Lrp1 regulates systemic energy homeostasis. Mice with endothelial-specific Lrp1 deletion display improved glucose sensitivity and lipid profiles combined with increased oxygen consumption during high-fat-diet-induced obesity. We show that the intracellular domain of Lrp1 interacts with the nuclear receptor Pparγ, a central regulator of lipid and glucose metabolism, acting as its transcriptional co-activator in endothelial cells. Therefore, Lrp1 not only acts as an endocytic receptor but also directly participates in gene transcription. Our findings indicate an underappreciated functional role of endothelium in maintaining systemic energy homeostasis.
Similar content being viewed by others
Introduction
Low-density lipoprotein (LDL)-related protein 1 (LRP1), a multifunctional member of LDL receptor family, is involved in a variety of biological processes, such as lipid metabolism, endocytosis and signal transduction1,2,3. Global deletion of Lrp1 gene in mice leads to embryonic lethality4. Depletion of Lrp1 in the livers of LDL receptor-deficient mice results in the accumulation of cholesterol-rich remnant lipoproteins in the circulation, suggesting the critical role of Lrp1 in the clearance of cholesterol-rich remnant lipoproteins5. Mice with hepatocyte-specific deletion of Lrp1 also display defects in cholesterol efflux and high-density lipoprotein (HDL) secretion6. On the other hand, inactivation of adipocyte Lrp1 results in delayed postprandial lipid clearance, reduced body weight, alterations in adipocyte tissue metabolism and resistance of high-fat-induced glucose tolerance and obesity7. The studies of Lrp1 forebrain knockout mice suggest that neuronal Lrp1 regulates food intake and energy homeostasis by directly affecting leptin signalling8. Although these tissue-specific knockout studies have provided us enormous information of Lrp1 functions and linked it to lipid metabolism, glucose homeostasis and obesity, the underlying mechanisms remain elusive.
Recently, we and others have demonstrated that Lrp1 is also expressed in endothelial cells (ECs) and can be induced by hypoxia and statins9,10,siRNA design and transient transfection The stealth siRNA duplexes were obtained from Life Technologies. The siRNA against mouse Pparγ is a duplex of 5′-ucaagggugccaguuucgauccgua-3′. The control siRNA is the Stealth RNAi negative control duplex (Cat. No. 12935-300) and was purchased from Life Technologies. The siRNAs were transfected into isolated Wt ECs according to our previous published protocol11. Briefly, for each sample, 2 × 105 ECs were transfected with 100 pmol siRNA. The experiments with siRNA-transfected ECs were performed 2 days later. The retrieved sequence of human LRP1β was used for the prediction of its secondary structure by SABLE server ( http://sable.cchmc.org/)38. No statistical methods were used to predetermine the sample size. No randomization was used as all mice used were genetically defined, inbred mice. Data analysis for metabolic phenotype was performed in a blinded fashion. Statistical data were drawn from normally distributed group with similar variance between groups. All data presented in this study are representative results of at least three independent experiments. Data are shown as the mean±s.e.m. ‘n’ represents the number of biological replicates. Differences were analysed with two-way analysis of variance and followed by a Fisher’s least significant difference test unless otherwise specifically stated. Values of P≤0.05 were considered statistically significant. The data that support the findings of this study are available within the article, its Supplementary Information files and from the corresponding author upon reasonable request.Structure prediction
Statistical analysis
Data availability
Additional information
How to cite this article: Mao, H. et al. Endothelial LRP1 regulates metabolic responses by acting as a co-activator of PPARγ. Nat. Commun. 8, 14960 doi: 10.1038/ncomms14960 (2017).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Boucher, P. & Herz, J. Signaling through LRP1: protection from atherosclerosis and beyond. Biochem. Pharmacol. 81, 1–5 (2011).
Lillis, A. P., Van Duyn, L. B., Murphy-Ullrich, J. E. & Strickland, D. K. LDL receptor-related protein 1: unique tissue-specific functions revealed by selective gene knockout studies. Physiol. Rev. 88, 887–918 (2008).
van der Geer, P. Phosphorylation of LRP1: regulation of transport and signal transduction. Trends Cardiovasc. Med. 12, 160–165 (2002).
Herz, J., Clouthier, D. E. & Hammer, R. E. LDL receptor-related protein internalizes and degrades uPA-PAI-1 complexes and is essential for embryo implantation. Cell 71, 411–421 (1992).
Rohlmann, A., Gotthardt, M., Hammer, R. E. & Herz, J. Inducible inactivation of hepatic LRP gene by cre-mediated recombination confirms role of LRP in clearance of chylomicron remnants. J. Clin. Invest. 101, 689–695 (1998).
Basford, J. E. et al. Hepatic deficiency of low density lipoprotein receptor-related protein-1 reduces high density lipoprotein secretion and plasma levels in mice. J. Biol. Chem. 286, 13079–13087 (2011).
Hofmann, S. M. et al. Adipocyte LDL receptor-related protein-1 expression modulates postprandial lipid transport and glucose homeostasis in mice. J. Clin. Invest. 117, 3271–3282 (2007).
Liu, Q. et al. Lipoprotein receptor LRP1 regulates leptin signaling and energy homeostasis in the adult central nervous system. PLoS Biol. 9, e1000575 (2011).
Andras, I. E. et al. HIV-1-induced amyloid beta accumulation in brain endothelial cells is attenuated by simvastatin. Mol. Cell. Neurosci. 43, 232–243 (2010).
Castellano, J. et al. Hypoxia stimulates low-density lipoprotein receptor-related protein-1 expression through hypoxia-inducible factor-{alpha} in human vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 31, 1411–1420 (2011).
Mao, H., Lockyer, P., Townley-Tilson, D., **e, L. & Pi, X. LRP1 regulates retinal angiogenesis by inhibiting PARP-1 activity and endothelial cell proliferation. Arterioscler. Thromb. Vasc. Biol. 36, 350–360 (2016).
Nakajima, C. et al. The lipoprotein receptor LRP1 modulates sphingosine-1-phosphate signaling and is essential for vascular development. Development 141, 4513–4525 (2014).
Pi, X. et al. LRP1-dependent endocytic mechanism governs the signaling output of the Bmp system in endothelial cells and in angiogenesis. Circ. Res. 111, 564–574 (2012).
Shinohara, M. et al. Reduction of brain beta-amyloid (Abeta) by fluvastatin, a hydroxymethylglutaryl-CoA reductase inhibitor, through increase in degradation of amyloid precursor protein C-terminal fragments (APP-CTFs) and Abeta clearance. J. Biol. Chem. 285, 22091–22102 (2010).
Strickland, D. K. & Muratoglu, S. C. LRP in endothelial cells: a little goes a long way. Arterioscler. Thromb. Vasc. Biol. 36, 213–216 (2016).
Beisiegel, U., Weber, W., Ihrke, G., Herz, J. & Stanley, K. K. The LDL-receptor-related protein, LRP, is an apolipoprotein E-binding protein. Nature 341, 162–164 (1989).
May, P., Woldt, E., Matz, R. L. & Boucher, P. The LDL receptor-related protein (LRP) family: an old family of proteins with new physiological functions. Ann. Med. 39, 219–228 (2007).
Kisanuki, Y. Y. et al. Tie2-Cre transgenic mice: a new model for endothelial cell-lineage analysis in vivo. Dev. Biol. 230, 230–242 (2001).
Ginsberg, H. N., Zhang, Y. L. & Hernandez-Ono, A. Regulation of plasma triglycerides in insulin resistance and diabetes. Arch. Med. Res. 36, 232–240 (2005).
May, P., Reddy, Y. K. & Herz, J. Proteolytic processing of low density lipoprotein receptor-related protein mediates regulated release of its intracellular domain. J. Biol. Chem. 277, 18736–18743 (2002).
Zurhove, K., Nakajima, C., Herz, J., Bock, H. H. & May, P. Gamma-secretase limits the inflammatory response through the processing of LRP1. Sci. Signal. 1, ra15 (2008).
Kanda, T. et al. PPARgamma in the endothelium regulates metabolic responses to high-fat diet in mice. J. Clin. Invest. 119, 110–124 (2009).
Yu, C. et al. The nuclear receptor corepressors NCoR and SMRT decrease peroxisome proliferator-activated receptor gamma transcriptional activity and repress 3T3-L1 adipogenesis. J. Biol. Chem. 280, 13600–13605 (2005).
Westin, S. et al. Interactions controlling the assembly of nuclear-receptor heterodimers and co-activators. Nature 395, 199–202 (1998).
Nolte, R. T. et al. Ligand binding and co-activator assembly of the peroxisome proliferator-activated receptor-gamma. Nature 395, 137–143 (1998).
Nicholson, A. C., Han, J., Febbraio, M., Silversterin, R. L. & Hajjar, D. P. Role of CD36, the macrophage class B scavenger receptor, in atherosclerosis. Ann. NY Acad. Sci. 947, 224–228 (2001).
Jeoung, N. H. & Harris, R. A. Pyruvate dehydrogenase kinase-4 deficiency lowers blood glucose and improves glucose tolerance in diet-induced obese mice. Am. J. Physiol. Endocrinol. Metab. 295, E46–E54 (2008).
Tao, R., **ong, X., Harris, R. A., White, M. F. & Dong, X. C. Genetic inactivation of pyruvate dehydrogenase kinases improves hepatic insulin resistance induced diabetes. PLoS ONE 8, e71997 (2013).
Wang, Q. A. et al. Distinct regulatory mechanisms governing embryonic versus adult adipocyte maturation. Nat. Cell Biol. 17, 1099–1111 (2015).
Wang, N. D. et al. Impaired energy homeostasis in C/EBP alpha knockout mice. Science 269, 1108–1112 (1995).
Hayashi, M. et al. Targeted deletion of BMK1/ERK5 in adult mice perturbs vascular integrity and leads to endothelial failure. J. Clin. Invest. 113, 1138–1148 (2004).
Jones, J. R. et al. Deletion of PPARgamma in adipose tissues of mice protects against high fat diet-induced obesity and insulin resistance. Proc. Natl Acad. Sci. USA 102, 6207–6212 (2005).
Home, P. Safety of PPAR agonists. Diabetes Care 34, (Suppl 2): S215–S219 (2011).
Wright, M. B., Bortolini, M., Tadayyon, M. & Bopst, M. Minireview: Challenges and opportunities in development of PPAR agonists. Mol. Endocrinol. 28, 1756–1768 (2014).
Biserni, A. et al. In vivo imaging reveals selective peroxisome proliferator activated receptor modulator activity of the synthetic ligand 3-(1-(4-chlorobenzyl)-3-t-butylthio-5-isopropylindol-2-yl)-2,2-dimethylpropanoic acid (MK-886). Mol. Pharmacol. 73, 1434–1443 (2008).
Obermoeller-McCormick, L. M. et al. Dissection of receptor folding and ligand-binding property with functional minireceptors of LDL receptor-related protein. J. Cell Sci. 114, 899–908 (2001).
Sobczak, M., Dargatz, J. & Chrzanowska-Wodnicka, M. Isolation and culture of pulmonary endothelial cells from neonatal mice. J. Vis. Exp. 46, 2316 (2010).
Adamczak, R., Porollo, A. & Meller, J. Combining prediction of secondary structure and solvent accessibility in proteins. Proteins 59, 467–475 (2005).
Acknowledgements
We thank the Baylor Phenoty** Core, Optical Imaging and Vital Microscopy Core and Histology Core Laboratories for their help. This work was supported by NIH R01s HL122736 (to L.X.), HL112890 and HL061656 (to X.P.).
Author information
Authors and Affiliations
Contributions
H.M., C.M.B., C.P., L.X. and X.P. conceived the research and designed the experiments. H.M., P.L., L.L. and X.P. performed the experiments. H.M., C.M.B., L.X. and X.P. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures, supplementary table and supplementary references. (PDF 14127 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Mao, H., Lockyer, P., Li, L. et al. Endothelial LRP1 regulates metabolic responses by acting as a co-activator of PPARγ. Nat Commun 8, 14960 (2017). https://doi.org/10.1038/ncomms14960
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms14960
- Springer Nature Limited
This article is cited by
-
Modulation of endothelium function by fatty acids
Molecular and Cellular Biochemistry (2022)
-
Brain endothelial LRP1 maintains blood–brain barrier integrity
Fluids and Barriers of the CNS (2021)
-
Loss of bone morphogenetic protein-binding endothelial regulator causes insulin resistance
Nature Communications (2021)
-
Endothelium-specific depletion of LRP1 improves glucose homeostasis through inducing osteocalcin
Nature Communications (2021)
-
Selection and validation of differentially expressed metabolic and immune genes in weaned Ghurrah versus crossbred piglets
Tropical Animal Health and Production (2021)