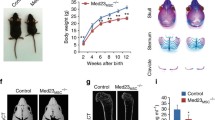
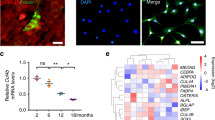
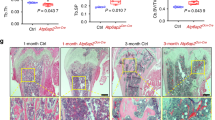
Abstract
Defects in stem cell renewal or progenitor cell expansion underlie ageing-related diseases such as osteoporosis. Yet much remains unclear about the mechanisms regulating progenitor expansion. Here we show that the tyrosine kinase c-Abl plays an important role in osteoprogenitor expansion. c-Abl interacts with and phosphorylates BMPRIA and the phosphorylation differentially influences the interaction of BMPRIA with BMPRII and the Tab1–Tak1 complex, leading to uneven activation of Smad1/5/8 and Erk1/2, the canonical and non-canonical BMP pathways that direct the expression of p16INK4a. c-Abl deficiency shunts BMP signalling from Smad1/5/8 to Erk1/2, leading to p16INK4a upregulation and osteoblast senescence. Mouse genetic studies revealed that p16INK4a controls mesenchymal stem cell maintenance and osteoblast expansion and mediates the effects of c-Abl deficiency on osteoblast expansion and bone formation. These findings identify c-Abl as a regulator of BMP signalling pathways and uncover a role for c-Abl in p16INK4a expression and osteoprogenitor expansion.







Similar content being viewed by others
References
Harada, S. & Rodan, G. A. Control of osteoblast function and regulation of bone mass. Nature 423, 349–355 (2003).
Olsen, B. R., Reginato, A. M. & Wang, W. Bone development. Annu. Rev. Cell Dev. Biol. 16, 191–220 (2000).
Ducy, P., Schinke, T. & Karsenty, G. The osteoblast: a sophisticated fibroblast under central surveillance. Science 289, 1501–1504 (2000).
Canalis, E., Economides, A. N. & Gazzerro, E. Bone morphogenetic proteins, their antagonists, and the skeleton. Endocr. Rev. 24, 218–235 (2003).
Sharpless, N. E. & DePinho, R. A. Telomeres, stem cells, senescence, and cancer. J Clin. Invest. 113, 160–168 (2004).
Sherr, C. J. & DePinho, R. A. Cellular senescence: mitotic clock or culture shock? Cell 102, 407–410 (2000).
Serrano, M. & Blasco, M. A. Putting the stress on senescence. Curr. Opin. Cell Biol. 13, 748–753 (2001).
Ivanov, A. & Adams, P. D. A damage limitation exercise. Nat. Cell Biol. 13, 193–195 (2011).
Lundberg, A. S., Hahn, W. C., Gupta, P. & Weinberg, R. A. Genes involved in senescence and immortalization. Curr. Opin. Cell Biol. 12, 705–709 (2000).
Takahashi, A. et al. Mitogenic signalling and the p16INK4a-Rb pathway cooperate to enforce irreversible cellular senescence. Nat. Cell Biol. 8, 1291–1297 (2006).
Schmitt, C. A. Senescence, apoptosis and therapy—cutting the lifelines of cancer. Nat. Rev. Cancer 3, 286–295 (2003).
Beausejour, C. M. & Campisi, J. Ageing: balancing regeneration and cancer. Nature 443, 404–405 (2006).
Van Riggelen, J. & Felsher, D. W. Myc and a Cdk2 senescence switch. Nat. Cell Biol. 12, 7–9 (2010).
Van Etten, R. A. Cycling, stressed-out and nervous: cellular functions of c-Abl. Trends Cell Biol. 9, 179–186 (1999).
Hantschel, O. & Superti-Furga, G. Regulation of the c-Abl and Bcr-Abl tyrosine kinases. Nat. Rev. Mol. Cell Biol. 5, 33–44 (2004).
Wong, S. & Witte, O. N. The BCR-ABL story: bench to bedside and back. Annu. Rev. Immunol. 22, 247–306 (2004).
Pendergast, A. M. The Abl family kinases: mechanisms of regulation and signaling. Adv. Cancer Res. 85, 51–100 (2002).
Wang, X. et al. A positive role for c-Abl in Atm and Atr activation in DNA damage response. Cell Death Diff. 18, 5–15 (2011).
Tybulewicz, V. L., Crawford, C. E., Jackson, P. K., Bronson, R. T. & Mulligan, R. C. Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell 65, 1153–1163 (1991).
Schwartzberg, P. L. et al. Mice homozygous for the ablm1 mutation show poor viability and depletion of selected B and T cell populations. Cell 65, 1165–1175 (1991).
Li, B. et al. Mice deficient in Abl are osteoporotic and have defects in osteoblast maturation. Nat Genet. 24, 304–308 (2000).
Li, B. et al. Distinct roles of c-Abl and Atm in oxidative stress response are mediated by protein kinase C delta. Genes Dev. 18, 1824–1837 (2004).
Silberman, I. et al. T cell survival and function requires the c- Abl tyrosine kinase. Cell Cycle 7, 3847–3857 (2008).
Huang, Y. et al. The c-Abl tyrosine kinase regulates actin remodeling at the immune synapse. Blood 112, 111–119 (2008).
Tzeng, S. J., Bolland, S., Inabe, K., Kurosaki, T. & Pierce, S. K. The B cell inhibitory Fc receptor triggers apoptosis by a novel c-Abl family kinase-dependent pathway. J. Biol. Chem. 280, 35247–35254 (2005).
Zipfel, P. A., Zhang, W., Quiroz, M. & Pendergast, A. M. Requirement for Abl kinases in T cell receptor signaling. Curr. Biol. 14, 1222–1231 (2004).
Koleske, A. J. et al. Essential roles for the Abl and Arg tyrosine kinases in neurulation. Neuron 21, 1259–1272 (1998).
Schlatterer, S. D., Acker, C. M. & Davies, P. c-Abl in neurodegenerative disease. J. Mol. Neurosci. 45, 445–452 (2011).
Ko, H. S. et al. Phosphorylation by the c-Abl protein tyrosine kinase inhibits parkin’s ubiquitination and protective function. Proc. Natl Acad. Sci. USA 107, 16691–16696 (2010).
De Arce, K. P. et al. Synaptic clustering of PSD-95 is regulated by c-Abl through tyrosine phosphorylation. J. Neurosci. 30, 3728–3738 (2010).
Michael, M., Vehlow, A., Navarro, C. & Krause, M. c-Abl, Lamellipodin, and Ena/VASP proteins cooperate in dorsal ruffling of fibroblasts and axonal morphogenesis. Curr. Biol. 20, 783–791 (2010).
Woodring, P. J. et al. Modulation of the F-actin cytoskeleton by c-Abl tyrosine kinase in cell spreading and neurite extension. J Cell Biol. 156, 879–892 (2002).
Zukerberg, L. R. et al. Cables links Cdk5 and c-Abl and facilitates Cdk5 tyrosine phosphorylation, kinase upregulation, and neurite outgrowth. Neuron 26, 633–646 (2000).
Wang, J. Y. Controlling Abl: auto-inhibition and co-inhibition? Nat. Cell Biol. 6, 3–7 (2004).
Chen, D., Zhao, M. & Mundy, G. R. Bone morphogenetic proteins. Growth Factors 22, 233–241 (2004).
Varga, A. C. & Wrana, J. L. The disparate role of BMP in stem cell biology. Oncogene 24, 5713–5721 (2005).
Randle, D. H., Zindy, F., Sherr, C. J. & Roussel, M. F. Differential effects of p19(Arf) and p16(Ink4a) loss on senescence of murine bone marrow -derived preB cells and macrophages. Proc. Natl Acad. Sci. USA 98, 9654–9659 (2001).
Dimri, G. P. et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl Acad. Sci. USA 92, 9363–9367 (1995).
O’Neill, A. J., Cotter, T. G., Russell, J. M. & Gaffney, E. F. Abl expression in human fetal and adult tissues, tumours, and tumour microvessels. J. Pathol. 183, 325–329 (1997).
Kotake, Y. et al. pRB family proteins are required for H3K27 trimethylation and Polycomb repression complexes binding to and silencing p16INK4α tumor suppressor gene. Genes Dev. 21, 49–54 (2007).
Bracken, A. P. et al. The Polycomb group proteins bind throughout theINK4A-ARF locus and are disassociated in senescent cells. Genes Dev. 21, 525–530 (2007).
Lin, A. W. et al. Premature senescence involving p53 and p16 is activated in response to constitutive MEK/MAPK mitogenic signaling. Genes Dev. 12, 3008–3019 (1998).
Cagnol, S. & Chambard, J. C. ERK and cell death: mechanisms of ERK—induced cell death—apoptosis, autophagy and senescence. FEBS J. 277, 2–21 (2010).
Von Kriegsheim, A. et al. Cell fate decisions are specified by the dynamic ERK interactome. Nat. Cell Biol. 11, 1458–1464 (2009).
Yamaguchi, K. et al. XIAP, a cellular member of the inhibitor of apoptosis protein family, links the receptors to TAB1-TAK1 in the BMP signaling pathway. EMBO J. 18, 179–187 (1999).
Sorrentino, A. et al. The type I TGF-beta receptor engages TRAF6 to activate TAK1 in a receptor kinase-independent manner. Nat. Cell Biol. 10, 1199–1207 (2008).
Miyazono, K. & Miyazawa, K. Id: a target of BMP signaling. Sci. STKE 2002, pe40 (2002).
Zhang, Y. & Derynck, R. Regulation of Smad signalling by protein associations and signalling crosstalk. Trends Cell Biol. 9, 274–279 (1999).
Zebedee, Z. & Hara, E. Id proteins in cell cycle control and cellular senescence. Oncogene 20, 8317–8325 (2001).
Ruzinova, M. B. & Benezra, R. Id proteins in development, cell cycle and cancer. Trends Cell Biol. 13, 410–418 (2003).
Zhao, M. et al. Bone morphogenetic protein receptor signaling is necessary for normal murine postnatal bone formation. J. Cell Biol. 157, 1049–1060 (2002).
Mishina, Y. et al. Bone morphogenetic protein type IA receptor signaling regulates postnatal osteoblast function and bone remodeling. J. Biol. Chem. 279, 27560–27566 (2004).
Kamiya, N. et al. BMP signaling negatively regulates bone mass through sclerostin by inhibiting the canonical Wnt pathway. Development 135, 3801–3811 (2008).
Kamiya, N. et al. Disruption of BMP signaling in osteoblasts through type IA receptor (BMPRIA) increases bone mass. J. Bone Miner. Res. 23, 2007–2017 (2008).
Rodda, S. J. & McMahon, A. P. Distinct roles for Hedgehog and canonical Wnt signaling in specification, differentiation and maintenance of osteoblast progenitors. Development 133, 3231–3244 (2006).
Usui, M. et al. Murine and chicken chondrocytes regulate osteoclastogenesis by producing RANKL in response to BMP2. J. Bone Miner. Res. 23, 314–325 (2008).
Whang, Y. E. et al. c-Abl is required for development and optimal cell proliferation in the context of p53 deficiency. Proc. Natl Acad. Sci. USA 97, 5486–5491 (2000).
Manolagas, S. C. & Jilka, R. L. Bone marrow, cytokines, and bone remodeling. Emerging insights into the pathophysiology of osteoporosis. N. Engl. J. Med. 332, 305–311 (1995).
Huot, T. J. et al. Biallelic mutations in p16(INK4a) confer resistance to Ras-and Ets-induced senescence in human diploid fibroblasts. Mol. Cell Biol. 22, 8135–8143 (2002).
Alani, R. M., Young, A. Z. & Shifflett, C. B. Id1 regulation of cellular senescence through transcriptional repression of p16/Ink4a. Proc. Natl Acad. Sci. USA 98, 7812–7816 (2001).
Molofsky, A. V. et al. Increasing p16INK4a expression decreases forebrain progenitors and neurogenesis during ageing. Nature 443, 448–452 (2006).
Krishnamurthy, J. et al. p16INK4a induces an age-dependent decline in islet regenerative potential. Nature 443, 453–457 (2006).
Janzen, V. et al. Stem-cell ageing modified by the cyclin-dependent kinase inhibitor p16INK4a. Nature 443, 421–426 (2006).
Tsuji, K. et al. BMP2 activity, although dispensable for bone formation, is required for the initiation of fracture healing. Nat. Genet. 38, 1424–1429 (2006).
Chang, J. et al. Inhibition of osteoblastic bone formation by nuclear factor-κB. Nat. Med. 15, 682–689 (2009).
Kawahara, T. L. et al. SIRT6 links histone H3 lysine 9 deacetylation toNF-κB-dependent gene expression and organismal life span. Cell 136, 62–74 (2009).
Serrano, M. et al. Role of the INK4a locus in tumor suppression and cell mortality. Cell 85, 27–37 (1996).
Yates, J. R. 3rd, Eng, J. K., McCormack, A. L. & Schieltz, D. Method to correlate tandem mass spectra of modified peptides to amino acid sequences in the protein database. Anal. Chem. 67, 1426–1436 (1995).
Wang, L.H. et al. pFind 2.0: a software package for peptide and protein identification via tandem mass spectrometry. Rapid Commun. Mass Spec. 21, 2985–2991 (2007).
Acknowledgements
We would like to thank A. Nur-E-Kamal, Y. Wan and X-H. Sun for helpful discussions, I. H. In, D. Cai, G. C. Hong, L. Soh, J. Lin and T. Cheng for technical assistance, and Novartis, J. Campisi (Buck Institute for Research on Aging, USA), J. Wang (UCSD, USA), A. Koleske (Yale University, USA), X-H. Sun (Oklahoma Medical Research Foundation, USA), C. Cepko (Harvard Medical School, USA), D. Bulavin (IMCB, Singapore) and R. DePinho (University of Texas, MD Anderson Cancer Center, USA) for reagents and mice. S.P.G. is an investigator of Howard Hughes Medical Institute. The work was supported by grants from the Ministry of Science and Technology of China (The National Key Scientific Program (2012CB966901, to B.L.)), the National Natural Science Foundation of China (81130039, 31071229 and 81121001), Shanghai Pujiang Program (10PJ1405000), Changjiang Scholars Program of the Ministry of Education and the Agency for Science, Technology and Research of Singapore.
Author information
Authors and Affiliations
Contributions
B.L. and L.H. conceived the project. H-Y.K., H.L, W.F.L., L.L., D.J., Y.H., X.W., J.F.L.C., J.Y., L.X. and G.M. carried out the experiments. Y-G.C, S.P.G., S.B. and Y. M. contributed the knockout mouse lines and constructs. H.L. and H-Y.K. prepared the figures. B.L., S.P.G., G-Q.C., S.B., L.H. and Y.M. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2198 kb)
Rights and permissions
About this article
Cite this article
Kua, HY., Liu, H., Leong, W. et al. c-Abl promotes osteoblast expansion by differentially regulating canonical and non-canonical BMP pathways and p16INK4a expression. Nat Cell Biol 14, 727–737 (2012). https://doi.org/10.1038/ncb2528
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2528
- Springer Nature Limited
This article is cited by
-
The roles and regulatory mechanisms of TGF-β and BMP signaling in bone and cartilage development, homeostasis and disease
Cell Research (2024)
-
BMP9 reduces age-related bone loss in mice by inhibiting osteoblast senescence through Smad1-Stat1-P21 axis
Cell Death Discovery (2022)
-
Gut stem cell aging is driven by mTORC1 via a p38 MAPK-p53 pathway
Nature Communications (2020)
-
DNA damage response manages cell cycle restriction of senile multipotent mesenchymal stromal cells
Molecular Biology Reports (2020)
-
Low concentration flufenamic acid enhances osteogenic differentiation of mesenchymal stem cells and suppresses bone loss by inhibition of the NF-κB signaling pathway
Stem Cell Research & Therapy (2019)