Abstract
Aim:
Some small molecules can induce mouse embryonic stem (ES) cells to differentiate into neuronal cells. Here, we explored the effect of isobavachin (IBA), a compound with a prenyl group at position 8 of ring A, on promoting neuronal differentiation and the potential role of its protein prenylation.
Methods:
The hanging drop method was employed for embryonic body (EB) formation to mimic embryo development in vivo. The EBs were treated with IBA at a final concentration of 10−7 mol/L from EB stage (d 4) to d 8+10. Geranylgeranyltransferase I inhibitor GGTI-298 was subsequently used to disrupt protein prenylation. Neuronal subtypes, including neurons and astrocytes, were observed by fluorescence microscopy. Gene and protein expression levels were detected using RT-PCR and Western blot analysis, respectively.
Results:
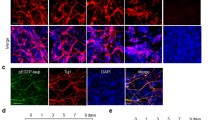
With IBA treatment, nestin was highly expressed in the neural progenitors generated from EBs (d 4, d 8+0). EBs then further differentiated into neurons (marked by β-tubulin III) and astrocytes (marked by GFAP), which were both up-regulated in a time-dependent manner on d 8+5 and d 8+10. Co-treatment with GGTI-298 selectively abolished the IBA-induced neuronal differentiation. Moreover, in the MAPK pathway, p38 and JNK phosphorylation were down-regulated, while ERK phosphorylation was up-regulated after IBA treatment at different neuronal differentiation passages.
Conclusion:
IBA can facilitate mouse ES cells differentiating into neuronal cells. The mechanism involved protein prenylation and, subsequently, phos-ERK activation and the phos-p38 off pathway.
Similar content being viewed by others
Introduction
Embryonic stem (ES) cells are pluripotent cells that have the capacity to continuously proliferate while retaining the potential to differentiate into three germ cells (cells derived from the end-, mes-, and ectoderm).
Mouse ES cells or their derivatives have been widely studied in the field of developmental biology, drug discovery and regenerative medicine1, 13, 34. It has also been reported that the sustained activation or phosphorylation of ERK is associated with differentiation35. In our study, the level of ERK phosphorylation was high in ES cells and was low in the early periods of d 4 and d 8+0 EB culture, while it increased greatly during late neuronal differentiation at d 8+5 and d 8+10 culture. These data indicate that ERK phosphorylation is a key event required for the survival of ES cells and for neurite formation and neuron survival during mouse ES cell differentiation16. At d 4 and d 8+0 culture, nestin was highly expressed in EBs, while ERK phosphorylation was inhibited, suggesting that the inhibition of ERK phosphorylation is associated with neural progenitor formation in mouse ES cells. p38 MAPK phosphorylation was shown to be a trigger in determining whether ES cells differentiated into neuronal cells or cardiomyocytes15. The control of p38 MAPK activity constitutes an early switch in ES cell commitment into cardiomyocytes (phos-p38 on) and neurons (phos-p38 off)18. In our study, p38 phosphorylation was expressed in the period of ES and EBs (d 4, d 8+0 culture), suggesting that p38 phosphorylation plays a critical role in maintaining mouse ES cell pluripotency and appears to promote neuroectoderm formation, while IBA can prevent p38 phosphorylation and subsequently promote neuronal differentiation. In the late period of neuronal differentiation, p38 phosphorylation was rare, indicating that p38 phosphorylation is associated with the trigger of mouse ES-cell differentiation and that its inhibition is important to neuronal differentiation. JNK phosphorylation was inhibited during neuronal differentiation compared with early stages of ES and EBs. It is noteworthy that the inhibitory effect of IBA on p38 is larger than on JNK phosphorylation. These results suggest that the regulation of the p38 and JNK MAPK signaling cascade may involve two mechanisms underlying the promoting effect of IBA on mouse ES cell neuronal differentiation19. Thus, a phosphatase-mediated crosstalk may exist between protein prenylation and the MAPK pathway in the regulation of neuronal differentiation from mouse ES cells promoted by IBA. Moreover, there may be a negative feedback regulation of their phosphorylation17, 36. Otherwise, ERK, p38, and JNK may be downstream targets of protein prenylation. However, how IBA regulates the ERK/JNK/p38 MAPK pathways and the specific relationship with protein prenylation needs to be further investigated.
In conclusion, this study suggests that IBA (prenyl group at position 8 of ring A) can facilitate the differentiation of mouse ES cells into multiple kinds of neuronal cell subtypes, such as neurons and astrocytes. The mechanism involved protein prenylation and, subsequently, phos-ERK activation and the phos-p38 off pathway.
Author contribution
Yi-jia LOU: Conception and design, financial support, final approval of manuscript; Dan-yin WANG: Provision of study material, acquisition of data and data analysis, manuscript writing; Yu-zhe HU and Si-si KONG: Partial acquisition of data; Yong-** YU: Partial provision of study molecule; Dan-yan ZHU: Partial conception and new skills; all authors have read and approved the final manuscript.
References
MacLaren RE, Pearson RA . Stem cell therapy and the retina. Eye (Lond) 2007; 21: 1352–9.
**ng F, Fang Z, Qin B, Li Y, Hou J, Chen X . Parthenogenetic embryonic stem cells derived from cryopreserved newborn mouse ovaries: a new approach to autologous stem cell therapy. Fertil Steril 2009; 91: 1238–44.
Emre N, Coleman R, Ding S . A chemical approach to stem cell biology. Curr Opin Chem Biol 2007; 11: 252–8.
Trompouki E, Zon LI . Small molecule screen in zebrafish and HSC expansion. Methods Mol Biol 2010; 636: 301–16.
Xu Y, Shi Y, Ding S . A chemical approach to stem-cell biology and regenerative medicine. Nature 2008; 453: 338–44.
Dantuma E, Merchant S, Sugaya K . Stem cells for the treatment of neurodegenerative diseases. Stem Cell Res Ther 2010; 1: 37.
Ding S, Schultz PG . Small molecules and future regenerative medicine. Curr Top Med Chem 2005; 5: 383–95.
Tavakoli-Nezhad M, Arbogast LA . Mu and kappa opioid receptor expression in the mediobasal hypothalamus and effectiveness of selective antagonists on prolactin release during lactation. Neuroscience 2010; 166: 359–67.
Wang Z, Wang H, Wu J, Zhu D, Zhang X, Ou L, et al. Enhanced co-expression of beta-tubulin III and choline acetyltransferase in neurons from mouse embryonic stem cells promoted by icaritin in an estrogen receptor-independent manner. Chem Biol Interact 2009; 179: 375–85.
Ochocki JD, Igbavboa U, Gibson Wood W, Wattenberg EV, Distefano MD . Enlarging the scope of cell-penetrating prenylated peptides to include farnesylated 'CAAX' box sequences and diverse cell types. Chem Biol Drug Des 2010; 76: 107–15.
Chen S, Fu L, Raja SM, Yue P, Khuri FR, Sun SY . Dissecting the roles of DR4, DR5 and c-FLIP in the regulation of geranylgeranyltransferase I inhibition-mediated augmentation of TRAIL-induced apoptosis. Mol Cancer 2010; 9: 23.
Seabra MC . Membrane association and targeting of prenylated Ras-like GTPases. Cell Signal 1998; 10: 167–72.
Kim HS, Kim JW, Gang J, Wen J, Koh SS, Koh JS, et al. The farnesyltransferase inhibitor, LB42708, inhibits growth and induces apoptosis irreversibly in H-ras and K-ras-transformed rat intestinal epithelial cells. Toxicol Appl Pharmacol 2006; 215: 317–29.
Binetruy B, Heasley L, Bost F, Caron L, Aouadi M . Concise review: regulation of embryonic stem cell lineage commitment by mitogen-activated protein kinases. Stem Cells 2007; 25: 1090–5.
Roux PP, Blenis J . ERK and p38 MAPK-activated protein kinases: a family of protein kinases with diverse biological functions. Microbiol Mol Biol Rev 2004; 68: 320–44.
Li Z, Theus MH, Wei L . Role of ERK 1/2 signaling in neuronal differentiation of cultured embryonic stem cells. Dev Growth Differ 2006; 48: 513–23.
Wang Z, Yang H, Tachado SD, Capo-Aponte JE, Bildin VN, Koziel H, et al. Phosphatase-mediated crosstalk control of ERK and p38 MAPK signaling in corneal epithelial cells. Invest Ophthalmol Vis Sci 2006; 47: 5267–75.
Aouadi M, Bost F, Caron L, Laurent K, Le Marchand Brustel Y, Binetruy B . p38 mitogen-activated protein kinase activity commits embryonic stem cells to either neurogenesis or cardiomyogenesis. Stem Cells 2006; 24: 1399–406.
Zeng KW, Fu H, Liu GX, Wang XM . Icariin attenuates lipopolysaccharide-induced microglial activation and resultant death of neurons by inhibiting TAK1/IKK/NF-kappaB and JNK/p38 MAPK pathways. Int Immunopharmacol 2010; 10: 668–78.
Rajan P, McKay RD . Multiple routes to astrocytic differentiation in the CNS. J Neurosci 1998; 18: 3620–9.
Liu J, Ye H, Lou Y . Determination of rat urinary metabolites of icariin in vivo and estrogenic activities of its metabolites on MCF-7 cells. Pharmazie 2005; 60: 120–5.
Doetschman TC, Eistetter H, Katz M, Schmidt W, Kemler R . The in vitro development of blastocyst-derived embryonic stem cell lines: formation of visceral yolk sac, blood islands and myocardium. J Embryol Exp Morphol 1985; 87: 27–45.
Stewart R, Stojkovic M, Lako M . Mechanisms of self-renewal in human embryonic stem cells. Eur J Cancer 2006; 42: 1257–72.
Zhu DY, Lou YJ . Inducible effects of icariin, icaritin, and desmethylicaritin on directional differentiation of embryonic stem cells into cardiomyocytes in vitro. Acta Pharmacol Sin 2005; 26: 477–85.
Metzger JM, Lin WI, Johnston RA, Westfall MV, Samuelson LC . Myosin heavy chain expression in contracting myocytes isolated during embryonic stem cell cardiogenesis. Circ Res 1995; 76: 710–9.
Scholz G, Pohl I, Genschow E, Klemm M, Spielmann H . Embryotoxicity screening using embryonic stem cells in vitro: correlation to in vivo teratogenicity. Cells Tissues Organs 1999; 165: 203–11.
Bain G, Kitchens D, Yao M, Huettner JE, Gottlieb DI . Embryonic stem cells express neuronal properties in vitro. Dev Biol 1995; 168: 342–57.
Murashov AK, Pak ES, Hendricks WA, Tatko LM . 17beta-Estradiol enhances neuronal differentiation of mouse embryonic stem cells. FEBS Lett 2004; 569: 165–8.
Zhang SC, Wernig M, Duncan ID, Brustle O, Thomson JA . In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nat Biotechnol 2001; 19: 1129–33.
Wobus AM . Potential of embryonic stem cells. Mol Aspects Med 2001; 22: 149–64.
Canals M, Angulo E, Casado V, Canela EI, Mallol J, Vinals F, et al. Molecular mechanisms involved in the adenosine A and A receptor-induced neuronal differentiation in neuroblastoma cells and striatal primary cultures. J Neurochem 2005; 92: 337–48.
Li BS, Zhang L, Gu J, Amin ND, Pant HC . Integrin alpha(1) beta(1)-mediated activation of cyclin-dependent kinase 5 activity is involved in neurite outgrowth and human neurofilament protein H Lys-Ser-Pro tail domain phosphorylation. J Neurosci 2000; 20: 6055–62.
Martin GR, Evans MJ . Differentiation of clonal lines of teratocarcinoma cells: formation of embryoid bodies in vitro. Proc Natl Acad Sci U S A 1975; 72: 1441–5.
Solomon CS, Goalstone ML . Dominant negative alpha-subunit of FTase inhibits effects of insulin and IGF-I in MCF-7 cells. Biochem Biophys Res Commun 2002; 291: 458–65.
Marshall CJ . Specificity of receptor tyrosine kinase signaling: transient versus sustained extracellular signal-regulated kinase activation. Cell 1995; 80: 179–85.
Estrada Y, Dong J, Ossowski L . Positive crosstalk between ERK and p38 in melanoma stimulates migration and in vivo proliferation. Pigment Cell Melanoma Res 2009; 22: 66–76.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (NSFC No 30973600, No 90813026).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, Dy., Hu, Yz., Kong, Ss. et al. Promoting effects of isobavachin on neurogenesis of mouse embryonic stem cells were associated with protein prenylation. Acta Pharmacol Sin 32, 425–432 (2011). https://doi.org/10.1038/aps.2011.5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2011.5
- Springer Nature Singapore Pte Ltd.
Keywords
This article is cited by
-
Flavonoids in Combination with Stem Cells for the Treatment of Neurological Disorders
Neurochemical Research (2023)
-
Shen-**g as a Chinese Medicine Concept Might Be a Counterpart of Stem Cells in Regenerative Medicine
Chinese Journal of Integrative Medicine (2019)
-
Structure Based docking studies towards exploring potential anti-androgen activity of selected phytochemicals against Prostate Cancer
Scientific Reports (2017)
-
The Role of Geranylgeranyltransferase I-Mediated Protein Prenylation in the Brain
Molecular Neurobiology (2016)