Abstract
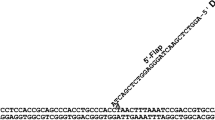
Flap endonuclease-1 (FEN-1), a structure specific nuclease, is an essential enzyme for eukaryotic DNA replication and repair. The crystal structure of FEN-1 from Methanococcus jannaschii, determined at 2.0 Å resolution, reveals an active site with two metal ions residing on top of a deep cleft where several conserved acidic residues are clustered. Near the active site, a long flexible loop comprised of many basic and aromatic residues forms a hole large enough to accommodate the DNA substrate. Deletion mutations in this loop significantly decreased the nuclease activity and specificity of FEN-1, suggesting that the loop is critical for recognition and cleavage of the junction between single and double-stranded regions of flap DNA.




Similar content being viewed by others
References
Harrington, J. J. & Lieber, M. R. Functional domains within FEN-1 and RAD2 define a family of structure-specific endonucleases: Implications for nucleotide excision repair. Genes Dev. 8 1344– 1355 (1994).
Harrington, J. J. & Lieber, M. R. The characterization of a mammalian structure-specific DNA endonuclease. EMBO J. 13, 1235– 1246 (1994).
Robins, P., Pappin, D., Wood, R. D. & Lindahl, T. Structural and functional homology between mammalian DNase IV and the 5'-nuclease domain of E. coli DNA polymerase I. J. Biol. Chem. 269, 28535– 28538 (1994).
Ishimi, Y., Claude, A., Bullock, P. & Hurwitz, J. Complete enzymatic synthesis of DNA containing the SV40 origin of replication. J. Biol. Chem. 263, 19723– 19733 (1988).
Waga, S., Baue, G. & Stillman, B. Reconstitution of complete SV40 DNA replication using purified proteins. J. Biol. Chem. 269, 10923– 10934 (1994).
Goulian, M., Richards, S. H., Heard, C. J. & Bigsby, B. M. Discontinuous DNA synthesis by purified mammalian proteins. J. Biol. Chem. 265, 18461– 18471 (1990).
Murante, R. S., Rumbaugh, J. A., Barnes, C. J., Norton, J. R., Bambara, R. A. Calf RTH1 nuclease can remove initiator RNAs of Okazaki fragments by endonuclease action. J. Biol. Chem. 271, 25888– 25897 (1996).
Sommers, C. H., Miller, E. J., Dujon, B., Prakash, L. & Prakash, S. Conditional lethality of null mutations in RTH1 that encodes the yeast counterpart of a mammalian 5' - to 3' exonuclease required for lagging strand DNA synthesis in reconstituted systems. J. Biol. Chem. 270, 4193– 4196 ( 1995).
Turchi, J. J. & Bambara, R. A. Completion of lagging strand DNA replication using purified proteins. J. Biol. Chem. 269, 1191– 1196 (1993).
Li, X., Li, J., Harrington, J. J., Lieber, M. R. & Burgers, P. M. J. Lagging strand DNA synthesis at the eukaryotic replication fork involves binding and stimulation of FEN-1 by PCNA. J. Biol. Chem. 270, 22109– 22112 (1995).
Wu, X., Li, J., Li, X., Hsieh, C -L., Burgers, P. M. J. & Lieber, M. R. Processing of branched DNA intermediates by a complex of human FEN-1 and PCNA . Nuc. Acids Res. 24, 2036– 2043 (1996).
Klungland, A. & Lindahl, T. Second pathway for completion of human DNA base excision-repair: reconstitution with purified proteins and requirement for DNase IV (FEN-1). EMBO J. 16, 3341– 3348 (1997).
Johnson, R., Kovvali, G., Prakash, L. & Prakash, S. Requirement of the yeast RTH1 5' to 3' exonuclease for the stability of simple repetitive DNA. Science 269, 238– 240 (1995).
Tishkoff, D. X., Fisoli, N., Gaida, N., Kolodner, R. D. A novel mutation avoidence mechanim dependent on S. cerevisiae RAD27 is distinct from DNA mismatch repair. Cell 88, 253– 263 ( 1997).
Eshleman, J. R. & Markowitz, S. D. Microsatellite instability in inherited and sporadic neoplasms. Curr. Opin. Oncol. 7, 83– 89 (1995).
Scherly, D. et al. Complementation of the DNA repair defect in xeroderma pigmentation group G cells by a human cDNA related to yeast RAD2. Nature 363, 182– 185 (1993).
Madura, K., & Prakash, P. Nucleotide sequence, transcript map**, and regulation of the rad2 gene of Saccharomyces cerevisiae. J. Bacteriol. 166, 914– 923 (1986).
Lyamichev, V., Brow, M. A. D. & Dahlberg, J. E. Structure-specific endonucleolytic cleavage of nucleic acids by eubacterial DNA polymerase. Science 260, 778– 783 (1993).
Gutman, P. D. & Minton, K. W. Conserved sites in the 5'-3' exonuclease domain of Escherichia coli DNA polymerase. Nuc. Acids Res. 21, 4406– 4407 ( 1993).
Kim, Y et al. Crystal structure of Thermus aquaticus DNA polymerase. Nature 376, 612– 616 (1995).
Ceska, T. A., Sayers, J. R., Stier, G. & Suck, D. A helical arch allowing single-stranded DNA to thread through T5 5' exonuclease. Nature 382, 90– 93 ( 1996).
Sander, C. and Schneider, R. Data base of homology derived protein structures and the structural meaning of sequence alignment. Proteins Struct. Funct. Genet. 9, 56– 68 (1991).
Mueser, T. C., Nossal, N. G. and Hyde, C. C. Structure of bacteriophage T4 RNase H, a 5' to 3' RNA-DNA and DNA-DNA exonuclease with sequence similarity to the RAD2 family of eukaryotic protein. Cell 95, 1101– 1112 ( 1996).
Artymiuk, P. J., Ceska, T. A., Suck, D. & Sayers, J. R. Prokaryotic 5'-3' exonucleases share a common core structure with gamma-delta resolvase . Nucleic Acids Res 25, 4224– 4229 (1997).
Shen, B., Nolan, J. P., Sklar, L. A. & Park, M. S. Functional analysis of point mutations in human flap endonuclease-1 active site. Nucleic Acids Res. 25, 3332– 3338 (1997).
Beese, L. S. & Steitz, T. A. Structural basis for the 3'-5' exonuclease activity of Escherichia coli polymerase I: a two metal ion mechanism. EMBO J. 10. 25– 33 (1991).
Mol, C. D., Kuo, C. F., Thayer, M. M., Cunningham, R. P. & Tainer, J. A. Structure and function of the multifunctional DNA-repair enzyme exonuclease III. Nature 374, 381– 386 (1995).
Barzilay, G. et al. Identification of critical active site residues in the multifunctional human DNA repair enzyme HAP1. Nature Struct Biol. 2, 562– 567 (1995).
Murante, R. S., Rust, L & Bambara, R. A. Calf 5' to 3' exo/endonuclease must slide from 5' end of the substarte to perform structure-specific cleavage. J. Biol. Chem., 270, 30377– 30383 (1995).
Otwinowski, Z & Minor, W. Processing of x-ray diffraction data collected in oscillation mode. Meth. Enz. 276, 307– 326 (1997).
Collaborative Computational Project Number 4. The CCP4 Suite: Program for protein crystallography. Acta. Crystallogr. D 50, 760– 763 (1994).
Sack, J. S. CHAIN: a crystallographic modelling program. J. Molec. Graphics 6, 224– 225 (1988).
Brünger, A. T. X-PLOR, a system for crystallography and NMR, Version 3.1 (Yale Univ. Press, New Haven, CT, 1992).
Kabsch, W. & Sander, C. Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 22, 2577– 2637 (1983).
Nicholls, A., Sharp, K. A. & Honig, B. Protein folding and association: insight from the interfacial and thermodynamic properties of hydrocarbons. Proteins Struct. Funct. Genet. 11, 281– 296 (1991).
Acknowledgements
We are grateful to M. S. Park (Los Alamos National Lab), S. S. Kim, E. Kim (L.G. Biotech) for their invaluable comments. We thanks to K. W. Bae and C. S. Cho for technical assistance. This work was supported by Biotech 2000 program from MOST and KIST 2000 program from KIST.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hwang, K., Baek, K., Kim, HY. et al. The crystal structure of flap endonuclease-1 from Methanococcus jannaschii . Nat Struct Mol Biol 5, 707–713 (1998). https://doi.org/10.1038/1406
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/1406
- Springer Nature America, Inc.
This article is cited by
-
Phosphate steering by Flap Endonuclease 1 promotes 5′-flap specificity and incision to prevent genome instability
Nature Communications (2017)
-
Evolution of the archaeal and mammalian information processing systems: towards an archaeal model for human disease
Cellular and Molecular Life Sciences (2017)
-
A virtual pebble game to ensemble average graph rigidity
Algorithms for Molecular Biology (2015)
-
Functional region prediction with a set of appropriate homologous sequences-an index for sequence selection by integrating structure and sequence information with spatial statistics
BMC Structural Biology (2012)
-
Rapid progress of DNA replication studies in Archaea, the third domain of life
Science China Life Sciences (2012)