Abstract
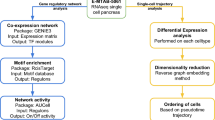
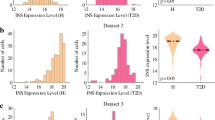
β cells are defined by the ability to produce and secret insulin. Recent studies have evaluated that human pancreatic β cells are heterogeneous and demonstrated the transcript alterations of β cell subpopulation in diabetes. Single-cell RNA sequence (scRNA-seq) analysis helps us to refine the cell types signatures and understand the role of the β cells during metabolic challenges and diseases. Here, we construct the pseudotime trajectory of β cells from publicly available scRNA-seq data in health and type 2 diabetes (T2D) based on highly dispersed and highly expressed genes using Monocle2. We identified three major states including 1) Normal branch, 2) Obesity-like branch and 3) T2D-like branch based on biomarker genes and genes that give rise to bifurcation in the trajectory. β cell function-maintain-related genes, insulin expression-related genes, and T2D-related genes enriched in three branches, respectively. Continuous pseudotime spectrum might suggest that β cells transition among different states. The application of pseudotime analysis is conducted to clarify the different cell states, providing novel insights into the pathology of β cells in T2D.




Similar content being viewed by others
Data Availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
Code Availability
The codes for performing the Seurat, Monocle2, and DEseq2 analyses are provided in the repository: https://github.com/finchbao/T2D_scRNA_seq/.
References
Alarcon C, Boland BB, Uchizono Y, Moore PC, Peterson B, Rajan S, Rhodes OS, Noske AB, Haataja L, Arvan P et al (2016) Pancreatic β-cell adaptive plasticity in obesity increases insulin production but adversely affects secretory function. Diabetes 65:438. https://doi.org/10.2337/db15-0792
Ashcroft FM, Rorsman P (2012) Diabetes mellitus and the β cell: the last ten years. Cell 148:1160–1171. https://doi.org/10.1016/j.cell.2012.02.010
Baron M, Veres A, Wolock SL, Faust AL, Gaujoux R, Vetere A, Ryu JH, Wagner BK, Shen-Orr SS, Klein AM et al (2016) A single-cell transcriptomic map of the human and mouse pancreas reveals inter- and intra-cell population structure. Cell Syst 3:346-360.e344. https://doi.org/10.1016/j.cels.2016.08.011
Cabrera O, Berman DM, Kenyon NS, Ricordi C, Berggren PO, Caicedo A (2006) The unique cytoarchitecture of human pancreatic islets has implications for islet cell function. Proc Natl Acad Sci USA 103:2334–2339. https://doi.org/10.1073/pnas.0510790103
Cheng K, Ho K, Stokes R, Scott C, Lau SM, Hawthorne WJ, O’Connell PJ, Loudovaris T, Kay TW, Kulkarni RN et al (2010) Hypoxia-inducible factor-1alpha regulates beta cell function in mouse and human islets. J Clin Investig 120:2171–2183. https://doi.org/10.1172/jci35846
Ebato C, Uchida T, Arakawa M, Komatsu M, Ueno T, Komiya K, Azuma K, Hirose T, Tanaka K, Kominami E et al (2008) Autophagy is important in islet homeostasis and compensatory increase of beta cell mass in response to high-fat diet. Cell Metab 8:325–332. https://doi.org/10.1016/j.cmet.2008.08.009
Eizirik DL, Pasquali L, Cnop M (2020) Pancreatic β-cells in type 1 and type 2 diabetes mellitus: different pathways to failure. Nat Rev Endocrinol 16:349–362. https://doi.org/10.1038/s41574-020-0355-7
Fadista J, Vikman P, Laakso EO, Mollet IG, Esguerra JL, Taneera J, Storm P, Osmark P, Ladenvall C, Prasad RB et al (2014) Global genomic and transcriptomic analysis of human pancreatic islets reveals novel genes influencing glucose metabolism. Proc Natl Acad Sci USA 111:13924–13929. https://doi.org/10.1073/pnas.1402665111
Fang Z, Weng C, Li H, Tao R, Mai W, Liu X, Lu L, Lai S, Duan Q, Alvarez C et al (2019) Single-cell heterogeneity analysis and CRISPR screen identify key β-cell-specific disease genes. Cell Rep 26:3132-3144.e3137. https://doi.org/10.1016/j.celrep.2019.02.043
Good AL, Haemmerle MW, Oguh AU, Doliba NM, Stoffers DA (2019) Metabolic stress activates an ERK/hnRNPK/DDX3X pathway in pancreatic β cells. Mol Metab 26:45–56. https://doi.org/10.1016/j.molmet.2019.05.009
Hamada S, Hara K, Hamada T, Yasuda H, Moriyama H, Nakayama R, Nagata M, Yokono K (2009) Upregulation of the mammalian target of rapamycin complex 1 pathway by Ras homolog enriched in brain in pancreatic beta-cells leads to increased beta-cell mass and prevention of hyperglycemia. Diabetes 58:1321–1332. https://doi.org/10.2337/db08-0519
Jung HS, Chung KW, Won Kim J, Kim J, Komatsu M, Tanaka K, Nguyen YH, Kang TM, Yoon K-H, Kim J-W et al (2008) Loss of autophagy diminishes pancreatic β cell mass and function with resultant hyperglycemia. Cell Metab 8:318–324. https://doi.org/10.1016/j.cmet.2008.08.013
Kahn SE (2003) The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of Type 2 diabetes. Diabetologia 46:3–19. https://doi.org/10.1007/s00125-002-1009-0
Karmaus PWF, Chen X, Lim SA, Herrada AA, Nguyen T-LM, Xu B, Dhungana Y, Rankin S, Chen W, Rosencrance C et al (2019) Metabolic heterogeneity underlies reciprocal fates of TH17 cell stemness and plasticity. Nature 565:101–105. https://doi.org/10.1038/s41586-018-0806-7
Kim KH, Jeong YT, Oh H, Kim SH, Cho JM, Kim YN, Kim SS, Kim DH, Hur KY, Kim HK et al (2013) Autophagy deficiency leads to protection from obesity and insulin resistance by inducing Fgf21 as a mitokine. Nat Med 19:83–92. https://doi.org/10.1038/nm.3014
Kubota N, Terauchi Y, Tobe K, Yano W, Suzuki R, Ueki K, Takamoto I, Satoh H, Maki T, Kubota T et al (2004) Insulin receptor substrate 2 plays a crucial role in beta cells and the hypothalamus. J Clin Investig 114:917–927. https://doi.org/10.1172/jci21484
Kusminski CM, Ghaben AL, Morley TS, Samms RJ, Adams AC, An Y, Johnson JA, Joffin N, Onodera T, Crewe C et al (2020) A novel model of diabetic complications: adipocyte mitochondrial dysfunction triggers massive β-cell hyperplasia. Diabetes 69:313–330. https://doi.org/10.2337/db19-0327
Lam PP, Leung YM, Sheu L, Ellis J, Tsushima RG, Osborne LR, Gaisano HY (2005) Transgenic mouse overexpressing syntaxin-1A as a diabetes model. Diabetes 54:2744–2754. https://doi.org/10.2337/diabetes.54.9.2744
Lawlor N, George J, Bolisetty M, Kursawe R, Sun L, Sivakamasundari V, Kycia I, Robson P, Stitzel ML (2017) Single-cell transcriptomes identify human islet cell signatures and reveal cell-type-specific expression changes in type 2 diabetes. Genome Res 27:208–222. https://doi.org/10.1101/gr.212720.116
Lin X, Taguchi A, Park S, Kushner JA, Li F, Li Y, White MF (2004) Dysregulation of insulin receptor substrate 2 in beta cells and brain causes obesity and diabetes. J Clin Investig 114:908–916. https://doi.org/10.1172/jci22217
Liu H, Javaheri A, Godar RJ, Murphy J, Ma X, Rohatgi N, Mahadevan J, Hyrc K, Saftig P, Marshall C et al (2017) Intermittent fasting preserves beta-cell mass in obesity-induced diabetes via the autophagy-lysosome pathway. Autophagy 13:1952–1968. https://doi.org/10.1080/15548627.2017.1368596
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550. https://doi.org/10.1186/s13059-014-0550-8
Matsuoka T-A, Artner I, Henderson E, Means A, Sander M, Stein R (2004) The MafA transcription factor appears to be responsible for tissue-specific expression of insulin. Proc Natl Acad Sci USA 101:2930. https://doi.org/10.1073/pnas.0306233101
Ohtsubo K, Chen MZ, Olefsky JM, Marth JD (2011) Pathway to diabetes through attenuation of pancreatic beta cell glycosylation and glucose transport. Nat Med 17:1067–1075. https://doi.org/10.1038/nm.2414
Qiu X, Mao Q, Tang Y, Wang L, Chawla R, Pliner HA, Trapnell C (2017) Reversed graph embedding resolves complex single-cell trajectories. Nat Methods 14:979–982. https://doi.org/10.1038/nmeth.4402
R Core Team (2020) R: a language and environment for statistical computing. Austria, Vienna
Rasouli N, Younes N, Utzschneider KM, Inzucchi SE, Balasubramanyam A, Cherrington AL, Ismail-Beigi F, Cohen RM, Olson DE, DeFronzo RA et al (2021) Association of baseline characteristics with insulin sensitivity and β-cell function in the glycemia reduction approaches in diabetes: a comparative effectiveness (GRADE) study cohort. Diabetes Care 44:340–349. https://doi.org/10.2337/dc20-1787
Rocha M, Apostolova N, Diaz-Rua R, Muntane J, Victor VM (2020) Mitochondria and T2D: role of autophagy, ER stress, and inflammasome. Trends Endocrinol Metab 31:725–741. https://doi.org/10.1016/j.tem.2020.03.004
Sarparanta J, García-Macia M, Singh R (2017) Autophagy and mitochondria in obesity and type 2 diabetes. Curr Diabetes Rev 13:352–369. https://doi.org/10.2174/1573399812666160217122530
Segerstolpe Å, Palasantza A, Eliasson P, Andersson EM, Andréasson AC, Sun X, Picelli S, Sabirsh A, Clausen M, Bjursell MK et al (2016) Single-cell transcriptome profiling of human pancreatic islets in health and type 2 diabetes. Cell Metab 24:593–607. https://doi.org/10.1016/j.cmet.2016.08.020
Sidarala V, Pearson GL, Parekh VS, Thompson B, Christen L, Gingerich MA, Zhu J, Stromer T, Ren J, Reck EC et al (2020) Mitophagy protects β cells from inflammatory damage in diabetes. JCI Insight. https://doi.org/10.1172/jci.insight.141138
Sivaskandarajah GA, Jeansson M, Maezawa Y, Eremina V, Baelde HJ, Quaggin SE (2012) Vegfa protects the glomerular microvasculature in diabetes. Diabetes 61:2958–2966. https://doi.org/10.2337/db11-1655
Soleimanpour SA, Gupta A, Bakay M, Ferrari AM, Groff DN, Fadista J, Spruce LA, Kushner JA, Groop L, Seeholzer SH et al (2014) The diabetes susceptibility gene Clec16a regulates mitophagy. Cell 157:1577–1590. https://doi.org/10.1016/j.cell.2014.05.016
Staels W, Heremans Y, Heimberg H, De Leu N (2019) VEGF-A and blood vessels: a beta cell perspective. Diabetologia 62:1961–1968. https://doi.org/10.1007/s00125-019-4969-z
Stuart T, Butler A, Hoffman P, Hafemeister C, Papalexi E, Mauck WM 3rd, Hao Y, Stoeckius M, Smibert P, Satija R (2019) Comprehensive integration of single-cell data. Cell 177:1888-1902.e1821. https://doi.org/10.1016/j.cell.2019.05.031
Sudhakar JN, Lu HH, Chiang HY, Suen CS, Hwang MJ, Wu SY, Shen CN, Chang YM, Li FA, Liu FT et al (2020) Lumenal Galectin-9-Lamp2 interaction regulates lysosome and autophagy to prevent pathogenesis in the intestine and pancreas. Nat Commun 11:4286. https://doi.org/10.1038/s41467-020-18102-7
Takamoto I, Terauchi Y, Kubota N, Ohsugi M, Ueki K, Kadowaki T (2008) Crucial role of insulin receptor substrate-2 in compensatory β-cell hyperplasia in response to high fat diet-induced insulin resistance. Diabetes Obes Metab 10:147–156. https://doi.org/10.1111/j.1463-1326.2008.00951.x
Taneera J, Lang S, Sharma A, Fadista J, Zhou Y, Ahlqvist E, Jonsson A, Lyssenko V, Vikman P, Hansson O et al (2012) A systems genetics approach identifies genes and pathways for type 2 diabetes in human islets. Cell Metab 16:122–134. https://doi.org/10.1016/j.cmet.2012.06.006
Wang YJ, Kaestner KH (2019) Single-cell RNA-Seq of the pancreatic islets—a promise not yet fulfilled? Cell Metab 29:539–544. https://doi.org/10.1016/j.cmet.2018.11.016
Wang H, Brun T, Kataoka K, Sharma AJ, Wollheim CB (2007) MAFA controls genes implicated in insulin biosynthesis and secretion. Diabetologia 50:348–358. https://doi.org/10.1007/s00125-006-0490-2
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer, New York
Wirostko B, Wong TY, Simó R (2008) Vascular endothelial growth factor and diabetic complications. Prog Retin Eye Res 27:608–621. https://doi.org/10.1016/j.preteyeres.2008.09.002
**n Y, Kim J, Okamoto H, Ni M, Wei Y, Adler C, Murphy AJ, Yancopoulos GD, Lin C, Gromada J (2016) RNA sequencing of single human islet cells reveals type 2 diabetes genes. Cell Metab 24:608–615. https://doi.org/10.1016/j.cmet.2016.08.018
**n Y, Dominguez Gutierrez G, Okamoto H, Kim J, Lee AH, Adler C, Ni M, Yancopoulos GD, Murphy AJ, Gromada J (2018) Pseudotime ordering of single human β-cells reveals states of insulin production and unfolded protein response. Diabetes 67:1783–1794. https://doi.org/10.2337/db18-0365
Yang L, Li P, Fu S, Calay ES, Hotamisligil GS (2010) Defective hepatic autophagy in obesity promotes ER stress and causes insulin resistance. Cell Metab 11:467–478. https://doi.org/10.1016/j.cmet.2010.04.005
Yang X, Graff SM, Heiser CN, Ho KH, Chen B, Simmons AJ, Southard-Smith AN, David G, Jacobson DA, Kaverina I et al (2020) Coregulator Sin3a promotes postnatal murine β-cell fitness by regulating genes in Ca(2+) homeostasis, cell survival, vesicle biosynthesis, glucose metabolism, and stress response. Diabetes 69:1219–1231. https://doi.org/10.2337/db19-0721
Zhou Q, Brown J, Kanarek A, Rajagopal J, Melton DA (2008) In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature 455:627–632. https://doi.org/10.1038/nature07314
Zhou Y, Zhou B, Pache L, Chang M, Khodabakhshi AH, Tanaseichuk O, Benner C, Chanda SK (2019) Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat Commun 10:1523. https://doi.org/10.1038/s41467-019-09234-6
Zhu D, Zhang Y, Lam PP, Dolai S, Liu Y, Cai EP, Choi D, Schroer SA, Kang Y, Allister EM et al (2012) Dual role of VAMP8 in regulating insulin exocytosis and islet β cell growth. Cell Metab 16:238–249. https://doi.org/10.1016/j.cmet.2012.07.001
Acknowledgements
This research was supported by the National Key R&D Program of China (2019YFA0801900, 2018YFA0800300), the National Natural Science Foundation of China (31971074), the Science and Technology Innovation Action Plan of Shanghai Science and Technology Committee (18140901300), the Open Research Fund of the National key laboratory of genetic engineering (SKLGE1803), the Shanghai Municipal Science and Technology Major Project (2017SHZDZX01).
Funding
This research was supported by the National Key R&D Program of China (2019YFA0801900, 2018YFA0800300), the National Natural Science Foundation of China (31971074), the Science and Technology Innovation Action Plan of Shanghai Science and Technology Committee (18140901300), the Open Research Fund of the National key laboratory of genetic engineering (SKLGE1803), the Shanghai Municipal Science and Technology Major Project (2017SHZDZX01), and Shanghai Frontiers Science Research Base of Exercise and Metabolic Health.
Author information
Authors and Affiliations
Contributions
KB contributed to the data analysis and manuscript writing. ZC contributed to scientific discussion and manuscript writing. TL and XK conceived the idea and contributed to the writing of the paper. HW and HX contributed to the final revision of the paper.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare no competing financial interests.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All the participants approved to publish.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bao, K., Cui, Z., Wang, H. et al. Pseudotime Ordering Single-Cell Transcriptomic of β Cells Pancreatic Islets in Health and Type 2 Diabetes. Phenomics 1, 199–210 (2021). https://doi.org/10.1007/s43657-021-00024-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43657-021-00024-z