Abstract
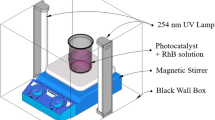
Tin (Sn) micro-nanoparticles with special pine tree dendritic morphology were synthesized by using tin foil as the anode and titanium as the cathode through simple anodization method. Surprisingly, it is found that the morphology of Sn particles is closely related to factors such as the type of electrolyte, the concentration of the electrolyte, and the different applied voltages, and briefly discussed the influence of various factors on the growth of Sn particles. In addition, Sn particles are calcined under different temperature conditions to obtain Sn/SnO2 hybrid materials with different tin dioxide (SnO2) contents. The changes in morphology and the phase of SnO2 crystal lattices were characterized by scanning electron microscopy (SEM) and X-ray diffraction (XRD), respectively, which proved the successful synthesis of Sn/SnO2 mixed materials. Finally, the Sn/SnO2 hybrid material with metal-doped modified semiconductor properties was used to photocatalytic degradation of simulated organic pollutants rhodamine B (RhB). It was found that the photocatalytic degradation efficiency of the Sn/SnO2 hybrid material under simulated sunlight conditions is near 90% in 5 h. Therefore, this work provides a convenient and effective environmental protection approach for the treatment of architecture and industrial dyes.
Graphical Abstract
Tin (Sn) micro-nanoparticles with special pine tree dendritic morphology are synthesized through simple anodization method, and the final product Sn/SnO2 particles after different heat treatments show superior photocatalytic degradation of RhB under simulated solar light.








Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article. Raw data that support the findings of this study are available from the corresponding author, upon reasonable request.
References
Bhat M, MR A, SV M, Das S, Roymahapatra G (2023) Photocatalytic degradation of dyes by titania/ferric oxide/polyvinyl alcohol nanocomposites. ES Gen 2:981. https://doi.org/10.30919/esg981
Kumar A, Singh M, Bhatt N, Jayaswal P, Sethi M, Burcar E, Wang Z (2024) The perils of heavy metal contamination in groundwater: impacts on human health in the northeastern region of Jhunjhunu, Rajasthan. ES Food Agrofor 6:1079. https://doi.org/10.30919/esfaf1079
Hou K, Xu X, **ang Y, Chen X, Lam SS, Naushad M, Sonne C, Ge S (2023) Rapid uptake of gold ions by sulfonated humic acid modified phenolic resin with high adsorption capacity and selectivity. Adv Compos Hybrid Mater 6:77. https://doi.org/10.1007/s42114-023-00647-y
Zhang H, Wang Z, Zhao Y, Cui W, Zhang X, Wang S, Walter ED, Sassi MJ, Pearce CI, Clark SB, Rosso KM (2023) Metal ion (Cr3+, Eu3+, UO22+) adsorption on gibbsite nanoplates. Eng Sci 4:896. https://doi.org/10.30919/es896
Balasubramaniyan S, Khan HA, Swaminathan M (2023) Microbial and solar photocatalytic degradation of pyridine. ES Energy Environ 22:971. https://doi.org/10.30919/esee971
Jasmine J, Ponvel KM (2023) Synthesis of Ag2CdS2/carbon nanocomposites for effective solar-driven ddye photodegradation and electrochemical application. ES Energy Environ 20:898. https://doi.org/10.30919/esee898
Ansari MZ, Ansari SA, Kim S-H (2022) Fundamentals and recent progress of Sn-based electrode materials for supercapacitors: A comprehensive review. J Energy Storage 53:105187. https://doi.org/10.1016/j.est.2022.105187
Shelke HD, Mohite AA, Torane AP, Madhale KV, Lokh CD (2022) Effect of Cu4SnS4 Layer Thickness on the Photovoltaic Parameters of Photoelectrochemical Solar Cells. ES Materi. Manuf 18:66–76. https://doi.org/10.30919/esmm5f724
Ahmad I, Idress A, Assiri MA, Ahmed SB, Shaban M, Ghadi YY (2023) Sn-based materials in photocatalysis: A review. Adv Colloid Interface Sci 321:103032. https://doi.org/10.1016/j.surfcoat.2018.10.011
Le Caër G, Matteazzi P, Fultz B (1992) A microstructural study of mechanical alloying of Fe and Sn powders. J Mater Res 7:1387–1395. https://doi.org/10.1557/JMR.1992.1387
**aohui W, Zhiliang L, Jie Z, **ngguo L, Zu** S (2017) Arc-discharge synthesis of dual-carbonaceous-layer-coated tin nanoparticles with tunable structures and high reversible lithium storage capacity. J Mater Chem A 5:13769–13775. https://doi.org/10.1039/C7TA03323K
Zaikovskii A, Novopashin S, Maltsev V, Kardash T, Shundrina I (2019) Tin–carbon nanomaterial formation in a helium atmosphere during arc-discharge. RSC Adv 9:36621–36630
Pintar A, Batista J, Muševič I (2004) Palladium-copper and palladium-tin catalysts in the liquid phase nitrate hydrogenation in a batch-recycle reactor. Appl Catal B 52:49–60. https://doi.org/10.1039/C9RA05485E
Deb NK, Kalita K, Giri PK, Abhilash S, Umapathy G, Biswas R, Das A, Kabiraj D, Chopra S, Bhuyan M (2020) Carbon-backed thin tin (116Sn) isotope target fabrication by physical vapor deposition technique. J Radioanal Nucl Chem 326:97–104. https://doi.org/10.1007/s10967-020-07316-0
Meléndrez M, Cárdenas G, Díaz VJ, Cruzat CC, Arbiol J (2009) Synthesis and aggregation study of tin nanoparticles and colloids obtained by chemical liquid deposition. Colloid Polym Sci 287:13–22. https://doi.org/10.1007/s00396-008-1950-7
Rozovskis G, Mockus Z, Pautienien V, Survila A (2002) Electrochemical determination of Sn (IV)/Sn(II) ratio in tin sol formed in copper–tin sulphate solution containing laprol 2402C. Electrochem commun 4:76–79. https://doi.org/10.1016/S1388-2481(01)00277-6
Poinern GE, Ng Y-J, Fawcett D (2010) Fine-tuning of catalytic tin nanoparticles by the reverse micelle method for direct deposition of silicon nanowires by a plasma-enhanced chemical vapour technique. J Colloid Interf Sci 352:259–264. https://doi.org/10.1016/j.jcis.2010.08.085
Chee S-S, Lee J-H (2014) Synthesis of sub-10-nm Sn nanoparticles from Sn(II) 2-ethylhexanoate by a modified polyol process and preparation of AgSn film by melting of the Sn nanoparticles. Thin Solid Films 562:211–217. https://doi.org/10.1016/j.tsf.2014.04.061
Chaudhari Y, Tarkas H, Tak S, Punde A, Shah S, Rahane S, Waghmare A, Hase Y, Sali J, Jadkar S (2022) Influence of Mixed-solvent System and Reaction Temperature on In situ, Surfactant-free Synthesis of P3HT:SnS Nanocomposites. ES Mater Manuf 18:45–55. https://doi.org/10.30919/esmm5f726
Biswas S, Doherty J, Saladukha D, Ramasse Q, Majumdar D, Upmanyu M, Singha A, Ochalski T, Morris MA, Holmes JD (2016) Non-equilibrium induction of tin in germanium: towards direct bandgap Ge1−xSnx nanowires. Nat commun 7:1–12. https://doi.org/10.1038/ncomms11405
Brumbach M, Veneman PA, Marrikar FS, Schulmeyer T, Simmonds A, **a W, Lee P, Armstrong NR (2007) Surface composition and electrical and electrochemical properties of freshly deposited and acid-etched indium tin oxide electrodes. Langmuir 23:11089–11099. https://doi.org/10.1021/la701754u
Brande PV, Weymeersch A (2000) Steel coating by self-induced ion plating, a new high throughput metallization ion plating technique. J Vac Sci Technol 18:1555–1560. https://doi.org/10.1116/1.582384
Veeraraghavan B, Durairajan A, Haran B, Popov B, Guidotti R (2002) Study of Sn-coated graphite as anode material for secondary lithium-ion batteries. J Electrochem Soc 149:A675. https://doi.org/10.1149/1.1470653
Rokade A, Tarkas H, Rokade A, Waman V, Upasani D, Sali JV, Jadkar SR (2023) Effect of Incorporation of SnS Nanorods on the Growth Mechanism, Crystallinity, Optoelectronic Performance, and Stability of CH3NH3PbI3 Perovskite Active Layer. ES Mater. Manuf. 22:937. https://doi.org/10.30919/esmm937
Kadam V, Jagtap C, Kumkale V, Rednam U, Lokhande P, Pathan H (2024) Photocatalytic degradation of rose bengal dye using chemically synthesized pristine and molybdenum doped zinc oxide. Eng Sci 8:1077. https://doi.org/10.30919/es1077
Sagadevan S, Lett JA, Weldegebrieal GK, Imteyaz S, Johan MR (2021) Synthesis, characterization, and photocatalytic activity of PPy/SnO2 nanocomposite. Chem Phys Lett 783:139051. https://doi.org/10.1016/j.cplett.2021.139051
Arif HS, Murtaza G, Hanif H, Ali HS, Yaseen M, Khalid N (2017) Effect of La on structural and photocatalytic activity of SnO2 nanoparticles under UV irradiation. J Environ Chem Eng 5:3844–3851. https://doi.org/10.1016/j.jece.2017.07.043
Abdelkader E, Nadjia L, Naceur B, Noureddine B (2016) SnO2 foam grain-shaped nanoparticles: Synthesis, characterization and UVA light induced photocatalysis. J Alloy Compound 679:408–419. https://doi.org/10.1016/j.jallcom.2016.04.016
Dhanalakshmi M, Saravanakumar K, Prabavathi SL, Abinaya M, Muthuraj V (2018) Fabrication of novel surface plasmon resonance induced visible light driven iridium decorated SnO2 nanorods for degradation of organic contaminants. J Alloy Compound 763:512–524. https://doi.org/10.1016/j.jallcom.2018.05.340
Li X, Yan J, Zhu K (2021) Fabrication and Characterization of Pt doped Ti/Sb-SnO2 Electrode and its Efficient Electro-Catalytic Activity toward Phenol. Eng. Sci 15:38–46. https://doi.org/10.30919/es8d432
Elango G, Kumaran SM, Kumar SS, Muthuraja S, Roopan SM (2015) Green synthesis of SnO2 nanoparticles and its photocatalytic activity of phenolsulfonphthalein dye. Spectrochim Acta Part A 145:176–180. https://doi.org/10.1016/j.saa.2015.03.033
Drouiche N, Aoudj S, Hecini M, Ghaffour N, Lounici H, Mameri N (2009) Study on the treatment of photovoltaic wastewater using electrocoagulation: Fluoride removal with aluminium electrodes—Characteristics of products. J Hazard Mater 169:65–69. https://doi.org/10.1016/j.jhazmat.2009.03.073
Allam NK, Grimes CA (2011) Electrochemical fabrication of complex copper oxide nanoarchitectures via copper anodization in aqueous and non-aqueous electrolytes. Mater Lett 65:1949–1955. https://doi.org/10.1016/j.matlet.2011.03.105
Deen K, Farooq A, Raza M, Haider W (2014) Effect of electrolyte composition on TiO2 nanotubular structure formation and its electrochemical evaluation. Electrochim Acta 117:329–335. https://doi.org/10.1016/j.electacta.2013.11.108
Su JF, Kuan W-F, Liu H, Huang C (2019) Mode of electrochemical deposition on the structure and morphology of bimetallic electrodes and its effect on nitrate reduction toward nitrogen selectivity. Appl Catal B: Environ 257:117909. https://doi.org/10.1016/j.apcatb.2019.117909
Zhang X, Wan K, Subramanian P, Xu M, Luo J, Fransaer J (2020) Electrochemical deposition of metal–organic framework films and their applications. J Mater Chem A 8:7569–7587. https://doi.org/10.1039/D0TA00406E
McKaveney JP, Byrnes C (1970) Apparatus using semiconductor electrodes for the measurement of acid concentrations. Anal Chem 42:1023–1028. https://doi.org/10.1021/ac60291a018
Han Z, Niu Y, Shi X, Pan D, Liu H, Qiu H, Chen W, Xu BB, El-Bahy ZM, Hou H, Elsharkawy ER, Amin MA, Liu C, Guo Z (2024) MXene@c-MWCNT adhesive silica nanofiber membranes enhancing electromagnetic interference shielding and thermal insulation performance in extreme environments. Nano-Micro Lett 16:195. https://doi.org/10.1007/s40820-024-01398-1
Sun B, Zhou G, Zhang Y, Liu R, Li T (2015) Photocatalytic properties of exposed crystal surface-controlled rutile TiO2 nanorod assembled microspheres. Chem Eng J 264:125–133. https://doi.org/10.1016/j.cej.2014.11.070
Pan D, Ge S, Zhao J, Shao Q, Guo L, Zhang X, Lin J, Xu G, Guo Z (2018) Synthesis, characterization and photocatalytic activity of mixed-metal oxides derived from NiCoFe ternary layered double hydroxides. Dalton Trans 47:9765–9778. https://doi.org/10.1039/C8DT01045E
Gu Y, Pan Z, Zhang H, Zhu J, Yuan B, Pan D, Wu C, Dong B, Guo Z (2020) Synthesis of high performance diesel oxidation catalyst using novel mesoporous AlLaZrTiOx mixed oxides by a modified sol-gel method. Adv Compos Hybrid Mater 3:583–593. https://doi.org/10.1007/s42114-020-00193-x
Wu L, Hu X, Qian J, Pei F, Wu F, Mao R, Ai X, Yang H, Cao Y (2013) A Sn–SnS–C nanocomposite as anode host materials for Na-ion batteries. J Mater Chem A 1:7181–7184. https://doi.org/10.1039/C3TA10920H
Wu L, Lu H, **ao L, Qian J, Ai X, Yang H, Cao Y (2014) A tin (ii) sulfide–carbon anode material based on combined conversion and alloying reactions for sodium-ion batteries. J Mater Chem A 2:16424–16428. https://doi.org/10.1039/C4TA03365E
Zhang N, Zhao Q, Han X, Yang J, Chen J (2014) Pitaya-like Sn@C nanocomposites as high-rate and long-life anode for lithium-ion batteries. Nanoscale 6:2827–2832. https://doi.org/10.1039/C3NR05523J
Guo Y, Tan R, Li Y, Song W (2012) Solution route to SnO2 crystals with controllable morphology. Appl Surf Sci 258:1958–1963. https://doi.org/10.1016/j.apsusc.2011.06.037
Wang WW, Zhu YJ, Yang LX (2007) ZnO–SnO2 hollow spheres and hierarchical nanosheets: hydrothermal preparation, formation mechanism, and photocatalytic properties. Adv Funct Mater 17:59–64. https://doi.org/10.1002/adfm.200600431
Cheng B, Russell JM, Shi W, Zhang L, Samulski ET (2004) Large-scale, solution-phase growth of single-crystalline SnO2 nanorods. J Am Chem Soc 126:5972–5973. https://doi.org/10.1021/ja0493244
Derikvandi H, Nezamzadeh-Ejhieh A (2017) Synergistic effect of pn heterojunction, supporting and zeolite nanoparticles in enhanced photocatalytic activity of NiO and SnO2. J Colloid Interf Sci 490:314–327. https://doi.org/10.1016/j.jcis.2016.11.069
Kim SP, Choi MY, Choi HC (2016) Photocatalytic activity of SnO2 nanoparticles in methylene blue degradation. Mater Res Bull 74:85–89. https://doi.org/10.1016/j.materresbull.2015.10.024
Kawade AN, Bhujbal PK, Supekar AT, Sonawane KM, Pathan HM, Shaikh SF, Al-Kaitani AAA (2021) Comparative Study of Eosin-Y and Rose Bengal Sensitized SnO2-ZnO Composite Electrode for Dye-Sensitized Solar Cell. ES Energy Environ 14:73–78. https://doi.org/10.30919/esee8c495
Roy S, Luitel H, Sanyal D (2019) Magnetic properties of transition metal doped SnO2: A detailed theoretical study. Computat Condens Matte 21:e00393. https://doi.org/10.1016/j.cocom.2019.e00393
Soitah TN, Yang C, Sun L (2010) Structural, optical and electrical properties of Fe-doped SnO2 fabricated by sol–gel dip coating technique. Mat Sci Semicon Proc 13:125–131. https://doi.org/10.1016/j.mssp.2010.03.002
Pan D, Ge S, Zhao J, Tian J, Shao Q, Guo L, Mai X, Wu T, Murugadoss V, Liu H (2018) Synthesis and characterization of ZnNiIn layered double hydroxides derived mixed metal oxides with highly efficient photoelectrocatalytic activities. Ind Eng Chem Res 58:836–848. https://doi.org/10.1021/acs.iecr.8b04829
Kumar R, Umar A, Kumar R, Chauhan M, Kumar G, Chauhan S (2021) Spindle-like Co3O4-ZnO nanocomposites scaffold for hydrazine sensing and photocatalytic degradation of rhodamine B dye. Eng. Sci 16:288–300. https://doi.org/10.30919/es8d548
Zhang F, Cheng W, Yu Z, Ge S, Shao Q, Pan D, Liu B, Wang X, Guo Z (2021) Microwave hydrothermally synthesized WO3/UiO-66 nanocomposites toward enhanced photocatalytic degradation of rhodamine B. Adv Compos Hybrid Mater 4:1330–1342. https://doi.org/10.1007/s42114-021-00346-6
Sun Z, Zhang Y, Guo S, Shi J, Shi C, Qu K, Qi H, Huang Z, Murugadoss V, Huang M, Guo Z (2022) Confining FeNi nanoparticles in biomass-derived carbon for effectively photo-Fenton catalytic reaction for polluted water treatment. Adv Compos Hybrid Mater 5:1566–1581. https://doi.org/10.1007/s42114-022-00477-4
Xu Z, Yu J (2011) Visible-light-induced photoelectrochemical behaviors of Fe-modified TiO2 nanotube arrays. Nanoscale 3:3138–3144. https://doi.org/10.1039/C1NR10282F
Huang K, Zhang Q, Yang F, He D (2010) Ultraviolet photoconductance of a single hexagonal WO3 nanowire. Nano Res 3:281–287. https://doi.org/10.1007/s12274-010-1031-3
Shinde DR, Quraishi IS, Pawar RA (2021) An efficient visible light driven photocatalytic removal of dyes from the dye effluent using metal halide lamp based slurry reactor. ES Energy Environ 14:54–62. https://doi.org/10.30919/esee8c504
Pan D, Ge S, Zhang X, Mai X, Li S, Guo Z (2018) Synthesis and photoelectrocatalytic activity of In2O3 hollow microspheres via a bio-template route using yeast templates. Dalton Trans 47:708–715. https://doi.org/10.1039/C7DT03878J
Khan SH, Pathak B, Fulekar MH (2020) A study on the influence of metal (Fe, Bi, and Ag) do** on structural, optical, and antimicrobial activity of ZnO nanostructures. Adv Compos Hybrid Mater 3:551–569. https://doi.org/10.1007/s42114-020-00174-0
Sun D, Jang S, Yim SJ, Ye L, Kim DP (2018) Metal Doped Core-Shell Metal-Organic Frameworks@ Covalent Organic Frameworks (MOFs@COFs) Hybrids as a Novel Photocatalytic Platform. Adv Funct Mater 28:1707110. https://doi.org/10.1002/adfm.201707110
Xu T, Zhang L, Cheng H, Zhu Y (2011) Significantly enhanced photocatalytic performance of ZnO via graphene hybridization and the mechanism study. Appl Catal B: Environ 101:382–387. https://doi.org/10.1016/j.apcatb.2010.10.007
Wang W-L, Wu Q-Y, Huang N, Xu Z-B, Lee M-Y, Hu H-Y (2018) Potential risks from UV/H2O2 oxidation and UV photocatalysis: a review of toxic, assimilable, and sensory-unpleasant transformation products. Water Res 141:109–125. https://doi.org/10.1016/j.watres.2018.05.005
Song Y, Tian J, Gao S, Shao P, Qi J, Cui F (2017) Photodegradation of sulfonamides by g-C3N4 under visible light irradiation: effectiveness, mechanism and pathways. Appl Catal B: Environ 210:88–96. https://doi.org/10.1016/j.apcatb.2017.03.059
**e K, Wei S, Alhadhrami A, Liu J, Zhang P, Elnaggar AY, Zhang F, Mahmoud MHH, Murugadoss V, El-Bahy SM, Wang F, Li C, Li G (2022) Synthesis of CsPbBr3/CsPb2Br5@silica yolk-shell composite microspheres: precisely controllable structure and improved catalytic activity for dye degradation. Adv Compos Hybrid Mater 5:1423–1432. https://doi.org/10.1007/s42114-022-00520-4
Acknowledgements
The authors extend their appreciation to Taif University, Saudi Arabia for supporting this work through project number (TU-DSPP-2024-01).
Funding
The research was funded by Taif University, Taif, Saudi Arabia (TU-DSPP-2024-01).
Author information
Authors and Affiliations
Contributions
Yukun Lu prepared the materials and conducted most of the measurements and data analysis. Yaojie Zhang and Jiale Zhang contributed to the data analysis. Zhaoyang Li, Feiyang Hu and Duo Pan conceived the idea, wrote the paper, and coordinated the overall project. Saad Melhi and Xuetao Shi revised the paper. Mohammed A. Amin provided supervision and resources. Zeinhom M. El-Bahy reviewed and revised the manuscript. Qian Shao revised the manuscript and coordinated the overall project. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lu, Y., Zhang, Y., Zhang, J. et al. Electrochemically synthesized Tin micro-nanometer powders for visible light photocatalytic degradation of Rhodamine B dye from polluted water. Adv Compos Hybrid Mater 7, 110 (2024). https://doi.org/10.1007/s42114-024-00921-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42114-024-00921-7