Abstract
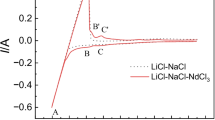
The electrochemical behavior of Al(III) ions was studied in molten LiCl-KCl melts on a molybdenum electrode. Cyclic voltammetry, chronopotentiometry and chronoamperometry were used to explore the deposition mechanism of Al and Li. Cyclic voltammetry expriment indicates that under potential deposition(UPD) of lithium on pre-deposited aluminium led to the formation of liquid Al-Li alloys at 853 K. The diffusion coefficient of Al(III) ions at 853 K in LiCl-KCl-AlF3(1%, mass fraction) melts was determined to be (2.79±0.05)×10−5 cm2/s. Chronopotentiograms and chronoamperograms demonstrate that the codeposition of Al(III) and Li(I) ions formed Al-Li alloys at cathodic current densities higher than −0.28 A/cm2 or cathodic potentials more negative than −2.20 V. X-Ray diffraction( XRD) pattern indicates that Al-Li alloys with different phases formed via galvanostatic electrolysis. Inductively coupled plasma(ICP) analyses of the samples obtained by electrolysis show that lithium and aluminium contents of Al-Li alloys could be controlled by AlF3 concentration and current intensity.
Similar content being viewed by others
References
Lavernia E. J., J. Mater. Sci., 1990, 25, 1137
Joh C. H., Yamada K., Miura Y., Mater. Trans., 1999, 40, 439
Gupta R. K., Nayan N., Nagasireesha G., Sharma S. C., Materials Science and Engineering A, 2006, 420, 228
Lagan T. J., Pickens J. R.; Ed.: Strakes E. A. Jr., Sanders T. H. Jr., Proceedings of Fifth International Al-Li Conference, Vol. II, Mater. and Comp. Engg. Pub., Williamsburg, 1989, 691
Li J. D., Zhang M. J., Wang Y. Y., Ai X. G., Advanced Materials Research, 2011, 233–235, 2913
Toyoshima M., Watanabe Y., Orito Y., Method of Producing a High Purity Aluminum-lithium Mother Alloy, US 4808283, 1989
Ye K., Zhang M. L., Chen Y., Han W., Yan Y. D., Cao P., Metallurgical and Materials Transactions B, 2010, 3(41), 691
Li Y. M, Wang F. L., Zhang M. L, Han W., Tian Y., Journal of Rare Earths, 2011, 29(4), 378
Yan Y. D, Zhang M. L., Xue Y., Han W., Cao D. X., Wei S. Q., Electrochimica Acta, 2009, 54, 3387
Han W., Tian Y., Zhang M. L., Ye K., Zhao Q. Y., Wei S. Q., Journal of Rare Earths, 2009, 27, 1046
Wei S. Q., Zhang M. L., Han W., Yan Y. D., Zhang M., Zhang B., Trans. Nonferrous Met. Soc. China, 2011, 21, 825
Bermejo M. R., de la Rosa F., Barrado E., Castrillejo Y., Journal of Electroanalytical Chemistry, 2007, 603, 81
Kuznetsov S. A., Hayashi H., Minato K., Gaune-Escard M., Electrochimica Acta, 2006, 51, 2463
Ødegard R., Bjørgum A., Sterten Å., Thonstad J., Tunold R., Electrochimica Acta, 1982, 27, 1595
Yan Y. D., Zhang M. L., Xue Y., Han W., Cao D. X., He L. Y., Journal of Applied Electrochemistry, 2009, 39, 455
Sum E., Skyllas-Kazacos M., Electrochimica Acta, 1989, 34, 677
Massalski T. B., Murray J. L., Benett L. H., Baker H., Binary Alloy Diagrams, American Society for Metals, OH, 1990
Massot L., Chamelot P., Taxil P., Electrochimica. Acta, 2005, 50, 5510
Nourry C., Massot L., Chamelot P., Taxil P., Electrochimca. Acta, 2008, 53, 2650
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the High Technology Research and Development Program of China(Nos.2009AA050702, 2011AA03A409) and the National Natural Science Foundation of China(No.21173060).
Rights and permissions
About this article
Cite this article
Sun, Y., Zhang, Ml., Han, W. et al. Electrochemical formation of Al-Li Alloys by codeposition of Al and Li from LiCl-KCl-AlF3 melts at 853 K. Chem. Res. Chin. Univ. 29, 324–328 (2013). https://doi.org/10.1007/s40242-013-2216-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-013-2216-6