Abstract
Purpose
Vitamin D3 (vit-D3) is a potent immunomodulator with anti-inflammatory and antioxidative properties. We used streptozotocin (STZ)-induced rat model of diabetes (DM) to evaluate the effects of vit-D3. We measured serum biochemical parameters, interleukin-17 (IL-17), osteocalcin (OC), malondialdehyde (MDA), and immune cell count on the 21st day of experiment.
Method
A total of 24 Wistar rats were randomly divided into three groups. Each group had eight rats. During the 1st day of the experiment, the control group was injected intraperitoneally with citrate buffer, while STZ group and STZ + vit-D3 group were injected by a single i.p. dose (35 mg/kg) of STZ dissolved in citrate buffer (pH 4,5; 0,1 M). Vitamin D3 was applied via oral gavage once daily to the STZ + vit-D3 group for a total period of 14 days, starting from the 7th day of the experiment.
Results
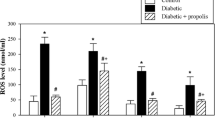
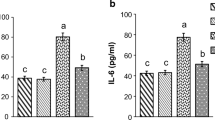
STZ rats showed a significant reduction in OC and an increase in MDA and IL-17 serum concentrations compared to the control rats. We also observed a significant STZ-associated decrease in the number of lymphocytes and a significant increase in monocyte and eosinophil number. Oral treatment with vit-D3 to STZ-induced diabetic rats significantly increased OC and decreased MDA serum levels. Furthermore, vit-D3 treatment resulted in a good regulation of hematopoiesis such as increase in the number of segmented granulocytes and lymphocytes and a reduction in the number of monocytes and eosinophils.
Conclusion
Vit-D3 treatment has important therapeutic effects; among many others it can attenuate oxidative stress and ameliorate the hyperglycemic state in the STZ-induced rat diabetic model, which is promising for further clinical trials.




Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Abdollahi M, Hosseini A. Streptozotocin. In: Phili W, editor. Encyclopedia of toxicology. 3rd ed. London: Academic; 2014. p. 402–4.
Schein PS, Cooney DA, Vernon ML. The use of nicotinamide to modify the toxicity of streptozotocin diabetes without loss of antitumor activity. Cancer Res. 1967;27:2324–32.
Lenzen S. The mechanisms of alloxan- and streptozotocin-induced diabetes. Diabetologia. 2008;51:216–26. https://doi.org/10.1007/s00125-007-0886-7.
Elsner M, Guldbakke B, Tiedge M, Munday R, Lenzen S. Relative importance of transport and alkylation for pancreatic beta-cell toxicity of streptozotocin. Diabetologia. 2000;43:1528–33. https://doi.org/10.1007/s001250051564.
Szkudelski T. The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiol Res. 2001;50:537–46.
Esposito K, Nappo F, Marfella R, Giugliano G, Giugliano F, Ciotola M, Quagliaro L, Ceriello A, Giugliano D. Inflammatory cytokine concentrations are acutely increased by hyperglycemia in humans. Circulation. 2002;106:2067–72. https://doi.org/10.1161/01.CIR.0000034509.14906.AE.
Pickup JC, Chusney GD, Thomas SM, Burt D. Plasma interleukin-6, tumor necrosis factor alpha and blood cytokine production in type 2 diabetes. Life Sci. 2000;67:291–300. https://doi.org/10.1016/s0024-3205(00)00622-6.
Fernánde-Real JM, Broch M, Vendrell J, Gutiérrez C, Casamitjana R, Pugeat M, Richart C, Ricart W. Interleukin-6 gene poly-morphism and insulin sensitivity. Diabetes. 2000;49:517–20. https://doi.org/10.2337/diabetes.49.3.517.
Mohamed-Ali V, Goodrick S, Rawesh A, Katz DR, Miles JM, Yudkin JS, Klein S, Coppack SW. Subcutaneous adiposetissue releases interleukin-6, but not tumor necrosis factor-alpha, in vivo. J Clin Endocrinol Metab. 1997;82:4196–200. https://doi.org/10.1210/jcem.82.12.4450.
Manohar SM, Vaikasuvu SR, Deepthi K, Sachan A, Narasimha SR. An association of hyperglycemia with plasma malondialdehyde and atherogenic lipid risk factors in newly diagnosed Type 2 diabetic patients. J Res Med Sci. 2013;18:89–93.
Foroughi M, Maghsoudi Z, Askari G. The effect of vitamin D supplementation on blood sugar and different indices of insulin resistance in patients with non-alcoholic fatty liver disease (NAFLD). Iran J Nurs Midwifery Res. 2016;21:100–4. https://doi.org/10.4103/1735-9066.174759.
Alvarez JA, Ashraf A. Role of vitamin D in insulin secretion and insulin sensitivity for glucose homeostasis. Int J Endocrinol. 2010;2010:351–85. https://doi.org/10.1155/2010/351385.
Martin T, Campbell RK. Vitamin D and diabetes. Diabetes Spectr. 2011;24:113–8. https://doi.org/10.2337/diaspect.24.2.113.
Kim MR, Jeong SJ. Relationship between Vitamin D level and lipid profile in non-obese children. Metabolites. 2019;9:125. https://doi.org/10.3390/metabo9070125.
Akbarzadeh A, Norouzian D, Mehrabi MR, Jamshidi S, Farhangi A, Verdi AA, Mofidian SM, Rad BL. Induction of diabetes by Streptozotocin in rats. Indian J Clin Biochem. 2007;2:60–4. https://doi.org/10.1007/BF02913315.
Lee JH, Yang SH, Oh JM, Lee MG. Pharmacokinetics of drugs in rats with diabetes mellitus induced by alloxan or streptozocin: comparison with those in patients with type I diabetes mellitus. J Pharm Pharmacol. 2010;62:1–23. https://doi.org/10.1211/jpp.62.01.0001.
Cruz B, Flores RJ, Uribe KP, Espinoza EJ, Spencer CT, Serafine KM, Nazarian A, O’Dell LE. Insulin modulates the strong reinforcing effects of nicotine and changes in insulin biomarkers in a rodent model of diabetes. Neuropsychopharmacol. 2019;44:1141–51. https://doi.org/10.1038/s41386-018-0306-3.
Von Hurst PR, Sonehouse W, Coad J. Vitamin D supplementation reduces insulin resistance in South Asian women living in New Zealand who are insulin resistant and vitamin D deficient: a randomized, placebo-controlled trial. Br J Nutr. 2010;103:549–55. https://doi.org/10.1017/S0007114509992017.
Bourlon PM, Billaudel B, Faure-Dussert A. Influence of vitamin D3 deficiency and 1,25 dihydroxyvitamin D3 on de novo insulin biosynthesis in the islets of the rat endocrine pancreas. J Endocrinol. 1999;160:87–95. https://doi.org/10.1677/joe.0.1600087.
Pittas AG, Lau J, Hu FB, Dawson-Hughes B. The role of vitamin D and calcium in type 2 diabetes: a systematic review and meta-analysis. J Clin Endocrinol Metab. 2007;92:2017–29. https://doi.org/10.1210/jc.2007-0298.
Parker J, Hashmi O, Dutton D, Mavrodaris A, Stranges S, Kandala NB, Clarke A, Franco OH. Levels of vitamin D and cardiometabolic disorders: systematic review and meta-analysis. Maturitas. 2010;65:225–36. https://doi.org/10.1016/j.maturitas.2009.12.013.
Schwallenberg G. Vitamin D and diabetes: improvement of glycemic control with vitamin D3 repletion. Can Fam Phys. 2008;54:864–6.
Chiu KC, Chu A, Go VL, Saad MF. Hypovitaminosis D is associated with insulin resistance and beta cell dysfunction. Am J Clin Nutr. 2004;79:820–5. https://doi.org/10.1093/ajcn/79.5.820.
Riachy R, Vandewalle B, Moerman E, Belaich S, Lukowiak B, Gmyr V, Muharram G, Conte JK, Pattou F. 1,25-Dihydroxyvitamin D3 protects human pancreatic islets against cytokine-induced apoptosis via down-regulation of the Fas receptor. Apoptosis. 2006;11:151–9. https://doi.org/10.1007/s10495-006-3558-z.
Hu Z, Chen J, Sun X, Wang L, Wang A. Efficacy of vitamin D supplementation on glycemic control in type 2 diabetes patients: A meta-analysis of interventional studies. Medicine. 2019;98(14):e14970. https://doi.org/10.1097/MD.0000000000014970.
El Khoury G, Mansour H, Kabbara W, Chamoun N, Attalah N, Salameh P. Hyperglycemia in hospitalized diabetic non-critically ill patients: Prevalence, correlates, management and nurses’ attitudes. Curr Diabetes Rev. 2018;15(2):133–40. https://doi.org/10.2174/1573399814666180119142254.
Arif S, Moore F, Marks K, Bouckenooghe T, Dayan CM. Peripheral and islet interleukin-17 pathway activation characterizes human autoimmune diabetes and promotes cytokine-mediated β-cell death. Diabetes. 2011;60:2112–29. https://doi.org/10.2337/db10-1643.
Fatima N, Faisal SM, Zubair S, Ajmal M, Siddiqui SS, Moin S, Owais M. Role of pro-inflammatory cytokines and biochemical markers in the pathogenesis of type 1 diabetes: Correlation with age and glycemic condition in diabetic human subjects. PLoS ONE. 2016;11(8):e0161548. https://doi.org/10.1371/journal.pone.0161548.
Mohammed FM, Pambuk CIA, Al-Kadh NA. Evaluation of interleukin 17 A level and lipid profile in diabetic female patients treated by Metformin and Glimepiride in Kirkuk City Iraq. Biomed Pharmacol J. 2019;12(4):1849–55. https://doi.org/10.13005/bpj/1815.
Zúñiga LA, Shen WJ, Joyce-Shaikh B, Pyatnova EA, Richards AG, Thom C, Andrade SM, Cua DJ, Kraemer FB, Butcher EC. IL-17 regulates adipogenesis, glucose homeostasis, and obesity. J Immunol. 2010;11:6947–59. https://doi.org/10.4049/jimmunol.1001269.
**a Y, Chen H, **ao H, Yang J, Li Z, Wang Y, Yang T, Wang B. Immune regulation mechanism of vitamin D level and IL-17/IL-17R pathway in Crohn’s disease. Exp Therapy Med. 2019;5:3423–8. https://doi.org/10.3892/etm.2019.7389.
Seckin D, Ilhan N, Ilhan N, Ertugrul S. Glycaemic control, markers of endothelial cell activation and oxidative stress in children with type 1 diabetes mellitus. Diabetes Res Clin Pract. 2006;73:191–7. https://doi.org/10.1016/j.diabres.2006.01.001.
Halliwell B, Chirico S. Lipid peroxidation: its mechanism, measurement, and significance. Am J Clin Nutr. 1993;57:715–24. https://doi.org/10.1093/ajcn/57.5.715S.
Firoozrai M, Nourbakhsh M, Razzaghy-Azar M. Erythrocyte susceptibility to oxidative stress and antioxidant status in patients with type 1 diabetes. Diabetes Res Clin Pract. 2007;77:427–32. https://doi.org/10.1016/j.diabres.2007.02.001.
Esterbauer H, Eckl P, Ortner A. Possible mutagens derived from lipids and lipid precursors. Mutat Res. 1990;238:223–33. https://doi.org/10.1016/0165-1110(90)90014-3.
Alatawi FS, Faridi UA, Alatawi MS. Effect of treatment with vitamin D plus calcium on oxidative stress in streptozotocin-induced diabetic rats. Saudi Pharma J. 2018;8:1208–13. https://doi.org/10.1016/j.jsps.2018.07.012.
Hwang YC, Jeong IK, Ahm KJ, Chung HY. Circulating osteocalcin level is associated with improved glucose tolerance, insulin secretion and sensitivity independent of the plasma adiponectin level. Osteoporosis Int. 2012;23:1337–42. https://doi.org/10.1007/s00198-011-1679-x.
Lee NK, Sowa H, Hinoi E, Ferron M, Ahn JD, Confavreux C, Dacquin R, Mee PJ, McKee MD, Jung DY, Zhang Z, Kim JK, Mauvais-Jarvis F, Ducy P, Karsenty G. Endocrine regulation of energy metabolism by the skeleton. Cell. 2007;130:456–69. https://doi.org/10.1016/j.cell.2007.05.047.
Janghorbani M, Feskanich D, Willett WC, Hu F. Prospective study of diabetes and risk of hip fracture: the Nurses’ Health Study. Diabetes Care. 2006;29:1573–8. https://doi.org/10.2337/dc06-0440.
Fadini GP, de Kreutzenberg SV, Boscaro E, Albiero M, Cappellari R, Krankel N, Landmesser U, Toniolo A, Bolego C, Cignarella A, Seeger F, Dimmeler S, Zeiher A, Agostini C, Avogaro A. An unbalanced monocyte polarisation in peripheral blood and bone marrow of patients with type 2 diabetes has an impact on microangiopathy. Diabetologia. 2013;8:1856–66. https://doi.org/10.1007/s00125-013-2918-9.
Venneri MA, Giannetta E, Panio G, De Gaetano R, Gianfrilli D, Pofi R, Masciarelli S, Fazi F, Pellegrini M, Lenzi A, Naro F, Isidori AM. Chronic inhibition of PDE5 limits pro-inflammatory monocyte-macrophage polarization in streptozotocin-induced diabetic mice. PLoS One. 2015;10(5):e0126580. https://doi.org/10.1371/journal.pone.0126580.
Min D, Brooks B, Wong J, Salomon R, Bao W, Harrisberg B, Twigg SM, Yue DK, McLennan SV. Alterations in monocyte CD16 in as-sociation with diabetes complications. Mediat Inflamm. 2021;2012:649083. https://doi.org/10.1155/2012/649083.
Muller YD, Golshayan D, Ehirchiou D, Wyss JC, Giovannoni L, Meier R, Serre-Beinier V, Puga Yung G, Morel P, Bühler LH, Seebach JD. Immunosuppressive effects of streptozotocin-induced diabetes result in absolute lymphopenia and a relative increase of T regulatory cells. Diabetes. 2011;60:2331–40. https://doi.org/10.2337/db11-0159.
Nagaraju S, Bertera S, Funair A, Wijkstrom M, Trucco M, Cooper DKC, Bottino R. Streptozotocin-associated lymphopenia in cynomolgus monkeys. Islets. 2014;6(3):e944441. https://doi.org/10.4161/19382014.2014.944441.
Stohr R, Federici M. Insulin resistance and atherosclerosis: convergence between metabolic pathways and inflammatory nodes. Biochem J. 2013;1:1–11. https://doi.org/10.1042/bj20130121.
Pifarre P, Gutierrez-Mecinas M, Prado J, Usero L, Roura-Mir C, Giralt M, Hidalgo J, Garcia A. Phosphodiesterase 5 in-hibition at disease onset prevents experimental autoimmune encephalomyelitis progression through immunoregulatory and neuroprotective actions. Experiment Neurol. 2014;251:58–71. https://doi.org/10.1016/j.expneurol.2013.10.021.
Venneri MA, De Palma M, Ponzoni M, Pucci F, Scielzo C, Zonari E, Mazzieri R, Doglioni C, Naldini L. Identification of proangiogenicTIE2-expressing monocytes (TEMs) in human peripheral blood and cancer. Blood. 2007;12:276–85. https://doi.org/10.1182/blood-2006-10-053504.
Acknowledgements
Authors want to acknowledge Dr. Andi Alijagic for critical reading of the manuscript.
Author information
Authors and Affiliations
Contributions
MMB, MF, AV: Methodology, validation, formal analysis; DS, AD, AZ: Conceptualization, data curation, writing original draft, writing review and editing. AMDŽ: writing original draft. The experiments were per-formed in-house, and no paper mill was used.
Corresponding author
Ethics declarations
Ethics approval
All applicable international, national, and institutional guidelines for the care and use of animals were followed by the authors.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mitrašinović-Brulić, M., Dervišević, A., Začiragić, A. et al. Vitamin D3 attenuates oxidative stress and regulates glucose level and leukocyte count in a semi-chronic streptozotocin-induced diabetes model. J Diabetes Metab Disord 20, 771–779 (2021). https://doi.org/10.1007/s40200-021-00814-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-021-00814-2