Abstract
Background
Calcitriol, the active metabolite of vitamin D, is an essential regulator in the hematopoiesis and immunity. However, knowledge revealing its influence on the immune and hematologic reconstitution after hematopoietic stem cell transplantation (HSCT) in clinical trials is very limited.
Objectives
The effects of calcitriol on short-term and long-term hematopoietic recovery, relapse-free survival (RFS) and overall survival (OS) in multiple myeloma, Hodgkin’s and non-Hodgkin’s lymphoma following autologous peripheral blood HSCT were assessed.
Methods
Eighty patients (age: 18–68 years) in complete remission were allocated 1:1 to two groups by balanced block randomization. Calcitriol 0.25 μg or placebo capsule was administered three times daily from transplantation to day 30. Absolute neutrophil count (ANC), absolute lymphocyte count (ALC), and platelet count (PC) were determined daily from transplantation to day 30. White blood cell count (WBC), PC, and hemoglobin concentration (HC) of days 180 and 365 were extracted from clinic files. A thorough examination for oral mucositis (OM) was completed daily during hospital stay. Adverse drug reactions (ADRs) as well as two-year RFS and OS were evaluated.
Results
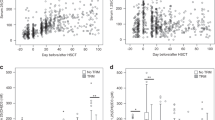
Median time to ANC engraftment (≥0.5 × 103/μl: 10.0 vs. 11.0 days; P = 0.98) and PC engraftment (≥20.0 × 103/μl: both 14.0 days; P = 0.58) was similar between groups. However, the median time to ALC recovery was significantly shorter in the calcitriol group (≥0.5 × 103/μl: 13.0 vs. 20.0 days; P < 0.001). Moreover, ALC recovery rates on day 15 (≥0.5 × 103/μl: 82.1% vs. 42.5%; P < 0.001) and on day 30 (≥1.0 × 103/μl: 91.7% vs. 57.5%; P = 0.001) was significantly higher with calcitriol. WBC, PC, and HC on days 180 and 365 were not significantly different between groups. None of the OM indices were modulated by calcitriol. All the ADRs were non-serious and mild, possibly or unlikely related to the intervention. In a median of 29 months follow-up, RFS was significantly better in the calcitriol group (77.0%, SE = 7.0% vs. 59.0%, SE = 8.0%; P = 0.03), albeit the OS was not affected (87.0%, SE = 5.0% vs. 92.0%, SE = 4.0%; P = 0.72).
Conclusion
Calcitriol could improve ALC recovery and RFS as a safe option post-HSCT.

Oral calcitriol 0.25 µg three times daily from transplantation to day 30 improved lymphocytes recovery and two-year relapse-free survival as a safe option in 80 patients of autologous hematopoietic stem cell transplantation in comparison with placebo.


Similar content being viewed by others
References
Relias V. Hematopoietic cell trasplantation. In: Zeind CS, Carvalho MG, editors. Applied therapeutics: the clinical use of drugs. 11th ed. Philadelphia: Lippincott Williams & Wilkins, Wolters Kluwer; 2018. p. 2102–6.
Porrata LF, Gertz MA, Inwards DJ, Litzow MR, Lacy MQ, Tefferi A, et al. Early lymphocyte recovery predicts superior survival after autologous hematopoietic stem cell transplantation in multiple myeloma or non-Hodgkin lymphoma. Blood. 2001;98(3):579–85.
Porrata LF, Inwards DJ, Micallef IN, Ansell SM, Geyer SM, Markovic SN. Early lymphocyte recovery post-autologous haematopoietic stem cell transplantation is associated with better survival in Hodgkin's disease. Br J Haematol. 2002;117(3):629–33.
Gordan LN, Sugrue MW, Lynch JW, Williams KD, Khan SA, Moreb JS. Correlation of early lymphocyte recovery and progression-free survival after autologous stem-cell transplant in patients with Hodgkin's and non-Hodgkin's lymphoma. Bone Marrow Transplant. 2003;31(11):1009–13.
Porrata LF, Inwards DJ, Ansell SM, Micallef IN, Johnston PB, Gastineau DA, et al. Early lymphocyte recovery predicts superior survival after autologous stem cell transplantation in non-Hodgkin lymphoma: a prospective study. Biol Blood Marrow Transplant. 2008;14(7):807–16.
Duggan P, Guo D, Luider J, Auer I, Klassen J, Chaudhry A, et al. Predictive factors for long-term engraftment of autologous blood stem cells. Bone Marrow Transplant. 2000;26(12):1299–304.
Amigo ML, del Cañizo MC, Caballero MD, Vazquez L, Corral M, Vidriales B, et al. Factors that influence long-term hematopoietic function following autologous stem cell transplantation. Bone Marrow Transplant. 1999;24(3):289–93. https://doi.org/10.1038/sj.bmt.1701886.
Studzinski GP, Harrison JS, Wang X, Sarkar S, Kalia V, Danilenko M. Vitamin D control of hematopoietic cell differentiation and leukemia. J Cell Biochem. 2015;116(8):1500–12. https://doi.org/10.1002/jcb.25104.
Bunce CM, Brown G, Hewison M. Vitamin D and hematopoiesis. Trends Endocrinol Metab. 1997;8(6):245–51.
Ros-Soto J, Anthias C, Madrigal A, Snowden JA. Vitamin D: is it important in haematopoietic stem cell transplantation? A review. Bone Marrow Transplant. 2018. https://doi.org/10.1038/s41409-018-0377-0.
Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol. 2014;21(3):319–29. https://doi.org/10.1016/j.chembiol.2013.12.016.
Bikle D. Nonclassic actions of vitamin D. J Clin Endocrinol Metab. 2009;94(1):26–34.
Borella E, Nesher G, Israeli E, Shoenfeld Y. Vitamin D: a new anti-infective agent? Ann N Y Acad Sci. 2014;1317(1):76–83. https://doi.org/10.1111/nyas.12321.
Trump DL, Deeb KK, Johnson CS. Vitamin D: considerations in the continued development as an agent for cancer prevention and therapy. Cancer J. 2010;16(1):1–9. https://doi.org/10.1097/PPO.0b013e3181c51ee6.
Damoiseaux J, Smolders J. The engagement between vitamin D and the immune system: is consolidation by a marriage to be expected? EBioMedicine. 2018;31:9–10. https://doi.org/10.1016/j.ebiom.2018.04.013.
Hansson ME, Norlin A-C, Omazic B, Wikström A-C, Bergman P, Winiarski J, et al. Vitamin D levels affect outcome in pediatric hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2014;20(10):1537–43.
von Bahr L, Blennow O, Alm J, Björklund A, Malmberg KJ, Mougiakakos D, et al. Increased incidence of chronic GvHD and CMV disease in patients with vitamin D deficiency before allogeneic stem cell transplantation. Bone Marrow Transplant. 2015;50(9):1217–23. https://doi.org/10.1038/bmt.2015.123.
Beebe K, Magee K, McNulty A, Stahlecker J, Salzberg D, Miller H, et al. Vitamin D deficiency and outcomes in pediatric hematopoietic stem cell transplantation. Pediatr Blood Cancer. 2018;65(2):e26817. https://doi.org/10.1002/pbc.26817.
Wallace G, Jodele S, Howell J, Myers KC, Teusink A, Zhao X, et al. Vitamin D deficiency and survival in children after hematopoietic stem cell transplant. Biol Blood Marrow Transplant. 2015;21(9):1627–31. https://doi.org/10.1016/j.bbmt.2015.06.009.
Joseph RW, Alousi A, Konda B, Komanduri K, Neumann J, Trevino C, et al. High incidence of vitamin D deficiency in patients undergoing allogeneic stem cell transplantation. Am J Hematol. 2011;86(11):954–6. https://doi.org/10.1002/ajh.22143.
Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357(3):266–81.
Urbain P, Ihorst G, Biesalski H-K, Bertz H. Course of serum 25-hydroxyvitamin D3 status and its influencing factors in adults undergoing allogeneic hematopoietic cell transplantation. Ann Hematol. 2012;91(5):759–66. https://doi.org/10.1007/s00277-011-1365-2.
Wallace G, Jodele S, Myers KC, Dandoy CE, El-Bietar J, Nelson A, et al. Vitamin D deficiency in pediatric hematopoietic stem cell transplantation patients despite both standard and aggressive supplementation. Biol Blood Marrow Transplant 2016;22(7):1271–4. https://doi.org/10.1016/j.bbmt.2016.03.026.
Rosenblatt J, Bissonnette A, Ahmad R, Wu Z, Vasir B, Stevenson K, et al. Immunomodulatory effects of vitamin D: implications for GvHD. Bone Marrow Transplant. 2010;45(9):1463–8.
Glotzbecker B, Ho VT, Aldridge J, Kim HT, Horowitz G, Ritz J, et al. Low levels of 25-hydroxyvitamin D before allogeneic hematopoietic SCT correlate with the development of chronic GvHD. Bone Marrow Transplant. 2013;48(4):593–7. https://doi.org/10.1038/bmt.2012.177.
Arain A, Matthiesen C. Vitamin D deficiency and graft-versus-host disease in hematopoietic stem cell transplant population. Hematol Oncol Stem Cell Ther 2018. https://doi.org/10.1016/j.hemonc.2018.08.001.
Hamidieh AA, Sherafatmand M, Mansouri A, Hadjibabaie M, Ashouri A, Jahangard-Rafsanjani Z, et al. Calcitriol for oral mucositis prevention in patients with Fanconi anemia undergoing hematopoietic SCT: a double-blind, randomized, placebo-controlled trial. Am J Ther. 2016;23(6):e1700–e8. https://doi.org/10.1097/MJT.0000000000000269.
Caballero-Velazquez T, Montero I, Sanchez-Guijo F, Parody R, Saldana R, Valcarcel D et al. Immunomodulatory effect of vitamin D after allogeneic stem cell transplantation: results of a prospective multicenter clinical trial. Clin Cancer Res. 2016;22(23):5673-81. https://doi.org/10.1158/1078-0432.ccr-16-0238.
Lips P. Relative value of 25(OH)D and 1,25(OH)2D measurements. J Bone Miner Res. 2007;22(11):1668–71. https://doi.org/10.1359/jbmr.070716.
Kreutz M, Eissner G, Hahn J, Andreesen R, Drobnik W, Holler E. Variations in 1α,25-dihydroxyvitamin D3 and 25-hydroxyvitamin D3 serum levels during allogeneic bone marrow transplantation. Bone Marrow Transplant. 2004;33(8):871–3. https://doi.org/10.1038/sj.bmt.1704448.
Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911–30.
NCCN guidelines. National Comprehensive Cancer Network, Plymouth Meeting, PA, USA. 2018. https://www.nccn.org/professionals/physician_gls/default.aspx. Accessed Jan 13 2019.
Hamadani M, Craig M, Awan FT, Devine SM. How we approach patient evaluation for hematopoietic stem cell transplantation. Bone Marrow Transplant. 2010;45:1259. https://doi.org/10.1038/bmt.2010.94.
Calcitriol capsules 0.25 mcg capsule. DailyMed, National Library of Medicine, USA. 2018. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=bebefb2a-7365-48e2-9681-83ed4b41e26e. Accessed Jan 13 2019.
Calcitriol (systemic): drug information. In: Post T, editor. UpToDate. Waltham, MA, USA: UpToDate Inc., Wolters Kluwer; 2018.
The use of the WHO-UMC system for standardised case causality assessment. WHO–UMC, Uppsala, Sweden. 2000. https://who-umc.org/media/164200/who-umc-causality-assessment_new-logo.pdf. Accessed Jan 13 2019.
Hartwig SC, Siegel J, Schneider PJ. Preventability and severity assessment in reporting adverse drug reactions. Am J Hosp Pharm. 1992;49(9):2229–32.
FDA. What is a serious adverse event? FDA, USA. 2016. http://www.fda.gov/Safety/MedWatch/HowToReport/ucm053087.htm. Accessed Jul 25 2019.
Bensinger W, Schubert M, Ang K, Brizel D, Brown E, Eilers J, et al. NCCN task force report: prevention and management of mucositis in cancer care. J Natl Compr Canc Netw 2008;6:S1–21.
Antiinfectives for systemic use. WHO, Oslo, Norway. 2018. http://www.whocc.no/atc_ddd_index/?code=J. Accessed Jan 13 2019.
Dudakov JA, Perales MA, van den Brink MRM. Immune reconstitution following hematopoietic cell transplantation. In: Forman SJ, Negrin RS, Antin JH, Appelbaum FR, editors. Thomas’ hematopoietic cell transplantation. 5th ed. UK: Wiley; 2016. p. 161–2.
Skrobot A, Demkow U, Wachowska M. Immunomodulatory role of vitamin D: a review. Current Trends in Immunity and Respiratory Infections. Springer; 2018. p. 13–23.
Balogh G, de Boland AR, Boland R, Barja P. Effect of 1,25(OH)(2)-vitamin D(3) on the activation of natural killer cells: role of protein kinase C and extracellular calcium. Exp Mol Pathol. 1999;67(2):63–74. https://doi.org/10.1006/exmp.1999.2264.
Ravid A, Koren R, Maron L, Liberman UA. 1,25(OH)2D3 increases cytotoxicity and exocytosis in lymphokine-activated killer cells. Mol Cell Endocrinol. 1993;96(1–2):133–9. https://doi.org/10.1016/0303-7207(93)90103-q.
Ota K, Dambaeva S, Kim MW, Han AR, Fukui A, Gilman-Sachs A, et al. 1,25-Dihydroxy-vitamin D3 regulates NK-cell cytotoxicity, cytokine secretion, and degranulation in women with recurrent pregnancy losses. Eur J Immunol. 2015;45(11):3188–99. https://doi.org/10.1002/eji.201545541.
Bochen F, Balensiefer B, Körner S, Bittenbring JT, Neumann F, Koch A, et al. Vitamin D deficiency in head and neck cancer patients–prevalence, prognostic value and impact on immune function. Oncoimmunology. 2018;7(9):e1476817.
Lemire JM, Adams J, Sakai R, Jordan S. 1 alpha, 25-dihydroxyvitamin D3 suppresses proliferation and immunoglobulin production by normal human peripheral blood mononuclear cells. J Clin Invest. 1984;74(2):657–61.
Quesada JM, Serrano I, Borrego F, Martin A, Pena J, Solana R. Calcitriol effect on natural killer cells from hemodialyzed and normal subjects. Calcif Tissue Int. 1995;56(2):113–7.
Dogan M, Erol M, Cesur Y, Yuca SA, Doğan Ş. The effect of 25-hydroxyvitamin D3 on the immune system. J Pediatr Endocrinol Metab. 2009;22(10):929–36.
Cortes M, Chen Michael J, Stachura David L, Liu Sarah Y, Kwan W, Wright F, et al. Developmental vitamin D availability impacts hematopoietic stem cell production. Cell Rep. 2016;17(2):458–68. https://doi.org/10.1016/j.celrep.2016.09.012.
Weeres MA, Robien K, Ahn Y-O, Neulen M-L, Bergerson R, Miller JS, et al. The effects of 1, 25-dihydroxyvitamin D3 on in vitro human NK cell development from hematopoietic stem cells. J Immunol. 2014;193(7):3456–62.
Hiwase DK, Hiwase S, Bailey M, Bollard G, Schwarer AP. Higher infused lymphocyte dose predicts higher lymphocyte recovery, which in turn, predicts superior overall survival following autologous hematopoietic stem cell transplantation for multiple myeloma. Biol Blood Marrow Transplant. 2008;14(1):116–24.
Kim H, Sohn H, Kim S, Kang H, Park S, Kim S, et al. Lymphocyte recovery as a positive predictor of prolonged survival after autologous peripheral blood stem cell transplantation in T-cell non-Hodgkin's lymphoma. Bone Marrow Transplant. 2004;34(1):43–9.
Kim H, Sohn H, Kim S, Lee J, Kim W, Suh C. Early lymphocyte recovery predicts longer survival after autologous peripheral blood stem cell transplantation in multiple myeloma. Bone Marrow Transplant. 2006;37(11):1037–42.
Porrata LF, Litzow MR, Markovic SN. Immune reconstitution after autologous hematopoietic stem cell transplantation. Mayo Clin Proc. 2001;76(4):407–12. https://doi.org/10.4065/76.4.407.
Medrano M, Carrillo-Cruz E, Montero I, Perez-Simon JA. Vitamin D: effect on haematopoiesis and immune system and clinical applications. Int J Mol Sci. 2018;19(9):2663. https://doi.org/10.3390/ijms19092663.
Zhou K, Chen D, ** H, Wu K, Wang X, Xu H, et al. Effects of calcitriol on experimental spinal cord injury in rats. Spinal Cord. 2016;54(7):510–6. https://doi.org/10.1038/sc.2015.217.
Stiff P. Mucositis associated with stem cell transplantation: current status and innovative approaches to management. Bone Marrow Transplant. 2001;27(S2):S3–S11. https://doi.org/10.1038/sj.bmt.1702863.
Acknowledgements
We greatly appreciate the participants without whom this investigational study would not be possible. We thank Ms. Ashraf Sadat Mousavi, Ms. Zahra Shahriari, BMT wards staff of Shariati Hospital, and Dr. Hamid Khoee (drug supply and randomization) for their kind assistances.
Funding
This study was funded through an educational grant to the researchers from the Hematology-Oncology and Stem Cell Transplantation Research Center, Shariati Hospital, Tehran University of Medical Sciences, Tehran, Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raoufinejad, K., Shamshiri, A.R., Pezeshki, S. et al. Oral calcitriol in hematopoietic recovery and survival after autologous stem cell transplantation: a randomized clinical trial. DARU J Pharm Sci 27, 709–720 (2019). https://doi.org/10.1007/s40199-019-00306-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40199-019-00306-y