Abstract
Introduction
The effects of deep neuromuscular block (DNMB) on chronic postsurgical pain (CPSP) have not been conclusively determined. Moreover, a limited number of studies have examined the impact of DNMB on long-term recovery quality after spinal surgery. We investigated the impact of DNMB on CPSP and the quality of long-term recovery in patients who had been subjected to spinal surgery.
Methods
This was a randomized, controlled, double-blind, single-center study performed from May 2022 to November 2022. A total of 220 patients who underwent spinal surgery under general anesthesia were randomly assigned to receive either DNMB (post-tetanic count at 1–2) (the D group) or moderate NMB (MNMB) (train-of-four at 1–3) (the M group). The primary endpoint was the incidence of CPSP. The secondary endpoints included the visual analogue scale (VAS) score in the post-anesthesia recovery unit (PACU), at 12, 24, 48 h and 3 months after surgery; postoperative opioid consumption; quality of recovery-15 (QoR-15) scores on the second postoperative day, before discharge, and 3 months after surgery.
Results
The incidence of CPSP was significantly lower in the D group (30/104, 28.85%) than in the M group (45/105, 42.86%) (p = 0.035). Besides, VAS scores were significantly reduced at the third month in the D group (p = 0.016). In the PACU and 12 h after surgery, VAS scores were also significantly lower in the D group than in the M group (p < 0.001, p = 0.004, respectively). The total amount of postoperative opioid consumption (expressed in total oral morphine equivalents) was significantly less in D group than M group (p = 0.027). At 3 months after surgery, QoR-15 scores were significantly higher in D group than M group (p = 0.003).
Conclusions
Compared with MNMB, DNMB significantly reduced CPSP and postoperative opioid consumption in spinal surgery patients. Moreover, DNMB improved the long-term recovery of patients.
Trial Registration
Chinese Clinical Trial Registry (ChiCTR2200058454).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Chronic postsurgical pain (CPSP) is hard to treat and is increasingly recognized as a public health problem. |
Deep neuromuscular blockade (DNMB) can reduce acute postsurgical pain. However, there is still a lack of studies examining the effects of DNMB on CPSP. |
The most recent peri-operative management guidelines for neuromuscular blockade recommend that more well-designed studies are needed to investigate if deep neuromuscular blockade (DNMB) is superior to moderate neuromuscular blockade (MNMB). |
What was learned from this study? |
DNMB significantly reduced the occurrence of CPSP in patients undergoing spinal surgery. |
DNMB may relieve acute postsurgical pain and reduce postoperative opioid consumption. |
DNMB improved long-term recovery quality in patients who have undergone spinal surgery. |
Introduction
Lumbar degenerative disease, which causes pain and disability, is very common in the elderly population [1]. Currently, spinal surgery is performed to treat degenerative conditions of the lumbar spine [2]. The annual number of spinal surgeries is increasing [3], as are complex procedures and the age of patients [4]. Major spinal surgeries may cause significant postoperative pain [5]. An increased risk of chronic postsurgical pain (CPSP) is associated with high-intensity and persistent postoperative pain [6]. Pain lasting more than 3 months is defined as chronic pain [7], which is difficult to treat and is sometimes permanent [8]. Thus, an effective perioperative pain control strategy is essential to improving patient recovery after surgery and reducing the chronic pain risk [9].
Neuromuscular blocking agents are integral in general anesthesia [10]. Studies have shown that profound neuromuscular blockade during anesthesia can reduce postoperative pain scores [11,12,13,14] and improve conditions of surgical space [11, 12, 14,15,16]. Deep neuromuscular block (DNMB) in spinal surgery is associated with various advantages, such as lower airway pressure, lower pain and rescue opioid consumption [17], as well as less bleeding [18, 19].
However, the effects of DNMB on CPSP and long-term recovery quality have not been fully established. Therefore, we assessed whether DNMB can reduce the incidence of CPSP after spinal surgery. Further, the impact of DNMB on the quality of long-term recovery in patients undergoing spinal surgery was investigated.
Methods
Study Design and Patients
This randomized, double-blind study was registered on https://www.chictr.org.cn/ (ChiCTR2200058454) and approved by the ethics committee of the Affiliated Hospital of Xuzhou Medical University. This study was conducted between May 2022 and November 2022 at the Affiliated Hospital of Xuzhou Medical University. Written informed consent was obtained from all participants prior to their inclusion in the study.
A total of 220 patients aged > 18 years old who underwent spinal surgery under general anesthesia and scored between I and III on the American Society of Anesthesiologists (ASA) physical status scale were recruited. The exclusion criteria were: (i) Known rocuronium allergies; (ii) Known allergies to cisatracurium or neostigmine; (iii) Contraindications to the use of neostigmine, such as bronchial asthma, glaucoma, myocardial ischemia and epilepsy; (iv) Preoperative hyperalgesia; (v) History of spinal surgery or neuromuscular disease that may interfere with data collection; (vi) Communication disability; (vii) Peripheral neuropathy caused by diabetes; (viii) History of chronic analgesics treatment; (ix) History of drug or alcohol abuse and (x) body mass index (BMI) ≥ 35 kg/m2.
Patients were randomly assigned (1:1 ratio) to the DNMB group (D group) or the moderate NMB (MNMB) group (M group) using an online randomization system (https://www.randomizer.org) to generate a random number table. Two unblinded personnel that were not otherwise involved in the study packed 220 opaque envelopes based on a computer-generated randomization protocol. The participants were screened and included by blinded researchers. Each participant was randomly assigned an envelope on-site at the end of the screening process. All participants were blinded. Each randomization envelope was opened by an unblinded individual who ensured that the NMB level was appropriate according to allocation of treatments.
Perioperative Management
Before surgery, all participants fasted for 8 h. After entering the operating room, they were monitored using basic monitors such as electrocardiograms, pulse oximeters, and invasive blood pressure (IBP). A general anesthetic with endotracheal intubation was used for all surgical procedures. Anesthesia was induced using midazolam (0.05–0.10 mg/kg), etomidate (0.2–0.3 mg/kg), sufentanil (0.5 µg/kg), and rocuronium (1.0 mg/kg). The bispectral index (BIS) monitor (BIS VISTATM, Aspect Medical System, USA) was used to measure the depth of anesthesia during surgery. Neuromuscular functions were monitored using the Philips IntelliVue neuromuscular transmission (NMT) module (connected to Philips IntelliVue MP70/MX800 patient monitor, J. 10. 52; Philips, Amsterdam, The Netherlands) or a train-of-four (TOF)-Cuff NMT monitor (RGB Medical Devices S.A., Madrid, Spain).
Propofol (3–6 mg/kg h), remifentanil (0.1–0.3 µg/kg min), and sevoflurane (0–1 vol%) were utilized for anesthesia maintenance. Pum** rates of propofol and remifentanil were continuously adjusted to maintain the hemodynamic stability and reach a BIS value of 40–60. Cisatracurium was continuously infused at a rate of 0.06–0.12 mg/kg h to maintain muscle relaxation within the target range. The volume control mode was used to ventilate the patients, with a tidal volume of 6–8 ml/kg. A 35–45 mmHg end-tidal CO2 pressure was maintained by controlling the respiratory rate. Core temperatures were maintained above 36 °C and continuously monitored. Pump delivery of cisatracurium in the M group was started when the TOF count returned to 2. The TOF count was maintained between 1 and 3 by adjusting the pump rate. In the D group, after tracheal intubation for 15 min, a cisatracurium pump was started, and its rate was adjusted to maintain a post-tetanic count (PTC) in the 1–2 range. Cisatracurium infusion was temporarily stopped when muscle relaxation deepened in both groups until they returned to predefined levels. Muscle relaxation monitoring was on standby during transition from the supine-to-prone position and prone-to-supine position. Monitoring resumed when positional change was completed. Pump delivery of cisatracurium was stopped approximately 30 min before the end of surgery.
After surgery, the muscle relaxant monitoring mode was adjusted to a TOF mode in all patients. Then, they were administered with neostigmine 1 mg and atropine 0.5 mg when the TOF count returned to 2 or TOF exceeded 70%. The endotracheal tube was removed when the TOF returned to 90% and the patient completed movements, such as eye-opening and handshaking, as instructed by the anesthesiologist. After extubation, a mask of high-flow oxygen was administered. When blood oxygen saturation stabilized above 95%, patients were transferred to a post-anesthesia recovery unit (PACU) where routine monitoring was performed. Sufentanil (2 µg/kg) and tropisetron (6 mg) were used for patient-controlled analgesia (PCA). Moderate pain (VAS score 4–6) was treated with fentanyl 0.05 mg, and severe pain (VAS score 7–10) with fentanyl 0.1 mg, with an aim to reduce pain scores to less than or equal to 3. All patients were discharged to the ward after fulfilling the PACU discharge criteria.
Outcomes
Patients’ baseline characteristics and intraoperative data were carefully documented. The primary outcome was the incidence of CPSP, which was defined as pain with VAS ≥ 4 and lasting more than 3 months [20]. At the third month after surgery, patients were followed up over the telephone, and VAS scores were used to measure the current and past three months’ pain status. Secondary outcomes included VAS scores in PACU, at 12, 24, 48 h and 3 months after surgery; postoperative opioid consumption expressed in cumulative oral morphine equivalents; quality of recovery-15 (QoR-15) scores [21] on the second postoperative day, before discharge, and 3 months after surgery; consumption of propofol and remifentanil; time to extubation; hypoxemia incidences after extubation and readmission incidences due to pain.
Sample Size
The primary objective of this study was to determine whether DNMB can prevent CPSP after spinal surgery. In the pre-experiment, the incidence of CPSP in the MNMB group was about 35%. To detect a 50% difference in CPSP incidence, 97 patients in each arm were required at a two-sided level of significance of 0.05 and a power of 80%. Therefore, assuming a 10% dropout rate, we included 110 patients in each group.
Statistical Analysis
All statistical analyses were conducted using SPSS version 25.0 (SPSS Inc., Chicago, IL, USA). Kolmogorov–Smirnov tests were used to assess the normality of continuous variables. Normally distributed variables were expressed as mean ± standard deviation (mean ± SD) while non-normally distributed variables were expressed as medians (interquartile range [IQR]). Continuous variables were compared between groups using the two-sample t test and Mann–Whitney U test, where appropriate. Categorical variables were expressed as numbers (percentages). Fisher’s exact test or chi-square test, where appropriate, were used to compare categorical variables. Analysis of continuous variables for intra-group comparisons over time was performed using repeated measures ANOVA followed by Bonferroni’s post hoc test. Repeated measures ANOVA models were sphericity tested using the Mauchly test and corrected for Greenhouse–Geisser sphericity as necessary. P values less than 0. 05 were considered statistically significant.
Results
Patient Selection
A total of 308 patients were assessed for eligibility, of which 220 patients were included and randomized. Among the 308 patients, 39 declined participation, seven had a history of spinal surgery, 28 had a history of chronic analgesic treatment and 14 had BMI ≥ 35 kg/m2. Then, 220 patients were randomized into two groups. Five patients in the M group and six in the D group were lost to follow-up. Therefore, 104 patients in the DNMB group and 105 patients in the MNMB group were finally analyzed (Fig. 1).
Patient Characteristics
Differences in demographic characteristics, including age, sex, height, weight, Charlson Comorbidity Index (CCI), and analgesic treatment between the groups were insignificant. Moreover, differences in surgical duration, type of surgery, and number of lumbar segments between the groups were insignificant. The total amount of rocuronium was similar between the two groups (p = 0.281), while the cisatracurium consumption was significantly more in the D group than M group (p < 0.001) (Table 1).
Primary Outcomes
There was CPSP in 45 of 105 patients (42.86%) in the M group, a significantly higher rate than in the D group (30/104, 28.85%, p = 0.035) (Table 2).
Secondary Outcomes
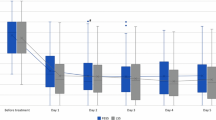
As shown in Table 2, lower VAS scores were observed at the third month (p = 0.016) in the D group. Regarding acute postoperative pain, there were significant interactions between the groups over time in terms of VAS scores (all p < 0.05). Pairwise comparisons in the PACU and 12 h after surgery revealed significantly higher VAS scores in the M group than in the D group (p < 0.001, p = 0.004, respectively) (Fig. 2). The total amount of opioid consumption postoperatively was more in the M group, than in the D group (p = 0.027) (Table 2). In Fig. 3, at 3 months after surgery, the D group had significantly higher quality of recovery-15 (QoR-15) scores than the M group (p = 0.003).
Differences between the groups in terms of total amount of propofol and remifentanil used during surgery were insignificant (p = 0.103, p = 0. 079, respectively). The D group had a significantly longer time to extubation (p < 0.001). Incidences of hypoxemia after extubation and readmission due to pain were comparable between the groups (p = 0.070, p = 0.277, respectively) (Table 2).
Discussion
Compared with MNMB, DNMB significantly reduced the incidence of CPSP and improved the long-term quality of recovery.
Physiologically, CPSP is pain (VAS score ≥ 4) that persists longer than 3 months after surgery and limits social activities [22]. It worsens the quality of life for patients, causing distress and impairment of functions [23]. Thus, there is an urgent need to develop better options for prevention and management of chronic pain [24]. There is a positive relationship between chronic pain and severe postoperative pain. Moreover, it is risky for chronic pain to develop after surgery when severe postoperative pain still exists [25, 26]. Reduced postoperative pain hastens functional recovery and hospital discharge, decreases acute morbidity, and may prevent chronic pain [27].
During anesthesia, DNMB reduces postoperative pain scores [11,12,13,14]. Moreover, DNMB confers several advantages in spinal surgery, such as lower airway pressure, lower pain and rescue opioid consumption [17], as well as less bleeding [18, 19]. However, a limited number of studies have assessed the effects of DNMB on CPSP and long-term recovery after spinal surgery. In this study, we found significantly reduced incidences of CPSP in DNMB patients. The incidence of CPSP was 28.85% in the D group and 42.86% in the M group. It was also found that DNMB reduced postoperative pain in the PACU and 12 h after surgery, and reduced postoperative opioid consumption. We speculate that the possible mechanism is that deep neuromuscular block can improve the surgical condition, reduce muscle traction, and decrease injury to muscle caused by surgery. In terms of QoR, patients in the DNMB group scored higher at 3 months after surgery. Nevertheless, the VAS scores at 24, 48 h after surgery and QoR-15 scores 2 days after surgery, before discharge were not significantly different between the two groups.
The extubation time was significantly earlier in the M group (p < 0.001) because longer time was needed for TOF count to return to 2 or for TOF% to exceed 70% in the D group. To reverse the blockade of neuromuscular functions, all participants were administered with neostigmine. Thus, deep NMB did not increase the incidence of hypoxemia after extubation.
The mechanisms by which acute pain becomes chronic pain have not been fully established [28]. The exact mechanisms that sustain chronic pain and underlie the transition between acute and chronic pain are unknown. Since postoperative pain can be a marker of chronic pain, reducing acute pain may reduce the transition from acute to chronic pain [29, 30]. We found that maintaining DNMB intraoperatively alleviated acute postoperative pain, which may be the key reason of low CPSP incidence in the D group.
Unlike previous studies, we investigated not only the effect of DNMB on postoperative pain, but also on CPSP and long-term QoR, which is a strength of this study. To the best of our knowledge, this is the first study to investigate the effects of DNMB on CPSP and long-term QoR. This study has several limitations. First, our study population only included patients who underwent lumbar surgery, while those who underwent cervical or thoracic surgery were excluded. Therefore, future studies should explore the effect of DNMB on cervical or thoracic surgery patients, which may be informative. Second, the mechanism and mediators of the interventions were not the focus of this study and need further research. Third, for simplicity, we chose a relatively simple QoR-15 scale rather than the QoR-40 scale. Fourth, a follow-up period longer than 3 months is required to evaluate the QoR. Finally, we did not record the surgeons’ scores for surgical working conditions.
Conclusions
In conclusion, compared with MNMB, DNMB significantly reduced CPSP and postoperative opioid consumption in spinal surgery patients. Moreover, DNMB improved the long-term recovery of patients.
References
Gong J, Huang X, Luo L, et al. Radiation dose reduction and surgical efficiency improvement in endoscopic transforaminal lumbar interbody fusion assisted by intraoperative O-arm navigation: a retrospective observational study. Neurospine. 2022;19:376–84.
Yee A, Adjei N, Do J, Ford M, Finkelstein J. Do patient expectations of spinal surgery relate to functional outcome? Clin Orthop Relat Res. 2008;466:1154–61.
Martin BI, Mirza SK, Spina N, Spiker WR, Lawrence B, Brodke DS. Trends in lumbar fusion procedure rates and associated hospital costs for degenerative spinal diseases in the United States, 2004 to 2015. Spine (Phila Pa 1976). 2019;44:369–76.
Deyo RA, Mirza SK, Martin BI, Kreuter W, Goodman DC, Jarvik JG. Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA. 2010;303:1259–65.
Sice PJ, Chan D, Macintyre PA. Epidural analgesia after spinal surgery via intervertebral foramen. Br J Anaesth. 2005;94:378–80.
Macrae WA. Chronic pain after surgery. Br J Anaesth. 2001;87:88–98.
Classification of chronic pain. Descriptions of chronic pain syndromes and definitions of pain terms. Prepared by the International Association for the Study of Pain, Subcommittee on Taxonomy. Pain Suppl 1986;3:S1–226.
Reddi D. Preventing chronic postoperative pain. Anaesthesia. 2016;71(Suppl 1):64–71.
Cruz JJ, Kather A, Nicolaus K, et al. Acute postoperative pain in 23 procedures of gynaecological surgery analysed in a prospective open registry study on risk factors and consequences for the patient. Sci Rep. 2021;11:22148.
Sunaga H, Zhang Y, Savarese JJ, Emala CW. Gantacurium and CW002 do not potentiate muscarinic receptor-mediated airway smooth muscle constriction in guinea pigs. Anesthesiology. 2010;112:892–9.
Kim MH, Lee KY, Lee KY, Min BS, Yoo YC. Maintaining optimal surgical conditions with low insufflation pressures is possible with deep neuromuscular blockade during laparoscopic colorectal surgery: a prospective, randomized, double-blind, parallel-group clinical trial. Medicine (Baltimore). 2016;95: e2920.
Koo BW, Oh AY, Seo KS, Han JW, Han HS, Yoon YS. Randomized clinical trial of moderate versus deep neuromuscular block for low-pressure pneumoperitoneum during laparoscopic cholecystectomy. World J Surg. 2016;40:2898–903.
Madsen MV, Istre O, Staehr-Rye AK, et al. Postoperative shoulder pain after laparoscopic hysterectomy with deep neuromuscular blockade and low-pressure pneumoperitoneum: a randomised controlled trial. Eur J Anaesthesiol. 2016;33:341–7.
Torensma B, Martini CH, Boon M, et al. Deep neuromuscular block improves surgical conditions during bariatric surgery and reduces postoperative pain: a randomized double blind controlled trial. PLoS One. 2016;11: e0167907.
Martini CH, Boon M, Bevers RF, Aarts LP, Dahan A. Evaluation of surgical conditions during laparoscopic surgery in patients with moderate vs deep neuromuscular block. Br J Anaesth. 2014;112:498–505.
Yoo YC, Kim NY, Shin S, et al. The intraocular pressure under deep versus moderate neuromuscular blockade during low-pressure robot assisted laparoscopic radical prostatectomy in a randomized trial. PLoS One. 2015;10: e0135412.
Oh SK, Kwon WK, Park S, et al. Comparison of operating conditions, postoperative pain and recovery, and overall satisfaction of surgeons with deep vs. no neuromuscular blockade for spinal surgery under general anesthesia: a prospective randomized controlled trial. J Clin Med. 2019;8:498.
Koh JC, Lee JS, Han DW, Choi S, Chang CH. Increase in airway pressure resulting from prone position patient placing may predict intraoperative surgical blood loss. Spine (Phila Pa 1976). 2013;38:E678-682.
Kang WS, Oh CS, Kwon WK, et al. Effect of mechanical ventilation mode type on intra- and postoperative blood loss in patients undergoing posterior lumbar interbody fusion surgery: a randomized controlled trial. Anesthesiology. 2016;125:115–23.
Schug SA, Lavand’homme P, Barke A, Korwisi B, Rief W, Treede RD. The IASP classification of chronic pain for ICD-11: chronic postsurgical or posttraumatic pain. Pain. 2019;160:45–52.
Stark PA, Myles PS, Burke JA. Development and psychometric evaluation of a postoperative quality of recovery score: the QoR-15. Anesthesiology. 2013;118:1332–40.
Treede RD, Rief W, Barke A, et al. Chronic pain as a symptom or a disease: the IASP classification of chronic pain for the International Classification of Diseases (ICD-11). Pain. 2019;160:19–27.
Guimarães AG, Serafini MR, Quintans-Júnior LJ. Terpenes and derivatives as a new perspective for pain treatment: a patent review. Expert Opin Ther Pat. 2014;24:243–65.
Alexander JK, Cox GM, Tian JB, et al. Macrophage migration inhibitory factor (MIF) is essential for inflammatory and neuropathic pain and enhances pain in response to stress. Exp Neurol. 2012;236:351–62.
Bisgaard T, Rosenberg J, Kehlet H. From acute to chronic pain after laparoscopic cholecystectomy: a prospective follow-up analysis. Scand J Gastroenterol. 2005;40:1358–64.
Perkins FM, Kehlet H. Chronic pain as an outcome of surgery. A review of predictive factors. Anesthesiology. 2000;93:1123–33.
Barreveld A, Witte J, Chahal H, Durieux ME, Strichartz G. Preventive analgesia by local anesthetics: the reduction of postoperative pain by peripheral nerve blocks and intravenous drugs. Anesth Analg. 2013;116:1141–61.
Kuner R, Flor H. Structural plasticity and reorganisation in chronic pain. Nat Rev Neurosci. 2016;18:20–30.
Dansie EJ, Turk DC. Assessment of patients with chronic pain. Br J Anaesth. 2013;111:19–25.
Niesters M, Aarts L, Sarton E, Dahan A. Influence of ketamine and morphine on descending pain modulation in chronic pain patients: a randomized placebo-controlled cross-over proof-of-concept study. Br J Anaesth. 2013;110:1010–6.
Acknowledgements
Funding
This study was supported by the Natural Science Research Fund of Higher Education Institutions in Jiangsu Province (22KJA320007). The Rapid Service Fee was funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship of this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
Su Liu, **nghe Wang, **hui Tang, Yan Wu, Qingsong Chen, and Yuqing Xu designed the study. Qingsong Chen and Yuqing Xu recruited the study patients. **hui Tang and Yan Wu performed the statistical analyses and wrote the manuscript. Su Liu and **nghe Wang revised the manuscript. All authors are responsible for the research data. All authors read and approved the final version of the manuscript.
Disclosures
**hui Tang, Yan Wu, Qingsong Chen, Yuqing Xu, **nghe Wang, and Su Liu declare that they have no conflicts of interest.
Compliance with Ethical Guidelines
The trial plan was approved by the ethical committee of the Affiliated Hospital of Xuzhou Medical University (XYFY2022-KL173-01). The study protocol was in accordance with the Declaration of Helsinki. This study was registered in the Chinese Clinical Trial Registry (ChiCTR2200058454) on April 9, 2022. Written informed consent was obtained from all participants in this study.
Data Availability
The data sets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Tang, X., Wu, Y., Chen, Q. et al. Deep Neuromuscular Block Attenuates Chronic Postsurgical Pain and Enhances Long-Term Postoperative Recovery After Spinal Surgery: A Randomized Controlled Trial. Pain Ther 12, 1055–1064 (2023). https://doi.org/10.1007/s40122-023-00528-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40122-023-00528-8