Abstract
Background:
Carbonic anhydrase 1 (CA1) has been found to be involved in osteogenesis and osteoclast in various human diseases, but the molecular mechanisms are not completely understood. In this study, we aim to use siRNA and lentivirus to reduce or increase the expression of CA1 in Dental follicle stem cells (DFSCs), in order to further elucidate the role and mechanism of CA1 in osteogenesis, and provide better osteogenic growth factors and stem cell selection for the application of bone tissue engineering in alveolar bone fracture transplantation.
Methods:
The study used RNA interference and lentiviral vectors to manipulate the expression of the CA1 gene in DFSCs during in vitro osteogenic induction. The expression of osteogenic marker genes was evaluated and changes in CA1, alkaline phosphatase (ALP), Runt-related transcription factor 2 (RUNX2), and Bone morphogenetic proteins (BMP2) were measured using quantitative real-time polymerase chain reaction (qRT-PCR) and Western blotting (WB). The osteogenic effect was assessed through Alizarin Red staining.
Results:
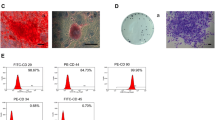
The mRNA and protein expression levels of CA1, ALP, RUNX2, and BMP2 decreased distinctly in the si-CA1 group than other groups (p < 0.05). In the Lentivirus-CA1 (LV-CA1) group, the mRNA and protein expressions of CA1, ALP, RUNX2, and BMP2 were amplified to varying degrees than other groups (p < 0.05). Apart from CA1, BMP2 (43.01%) and ALP (36.69%) showed significant upregulation (p < 0.05). Alizarin red staining indicated that the LV-CA1 group produced more calcified nodules than other groups, with a higher optical density (p < 0.05), and the osteogenic effect was superior.
Conclusions:
CA1 can impact osteogenic differentiation via BMP related signaling pathways, positioning itself upstream in osteogenic signaling pathways, and closely linked to osteoblast calcification and ossification processes.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
Gaihre B, Uswatta S, Jayasuriya AC. Reconstruction of craniomaxillofacial bone defects using tissue-engineering strategies with injectable and non-injectable scaffolds. J Funct Biomater. 2017;8:49.
Proussaefs P, Lozada J. The use of intraorally harvested autogenous block grafts for vertical alveolar ridge augmentation: a human study. Int J Periodontic Restor Dent. 2005;25:351–63.
Eppley BL, Pietrzak WS, Blanton MW. Allograft and alloplastic bone substitutes: a review of science and technology for the craniomaxillofacial surgeon. J Craniofac Surg. 2005;16:981–9.
Frenken JW, Bouwman WF, Bravenboer N, Zijderveld SA, Schulten EA, Ten Bruggenkate CM. The use of straumann bone ceramic in a maxillary sinus floor elevation procedure: a clinical, radiological, histological and histomorphometric evaluation with a 6-month healing period. Clin Oral Implants Res. 2010;21:201–8.
Holly D, Klein M, Mazreku M, Zamborsky R, Polak S, Danisovic L, et al. Stem cells and their derivatives-implications for alveolar bone regeneration: a comprehensive review. Int J Mol Sci. 2021;22.
Anitua E, Troya M, Zalduendo M. Progress in the use of dental pulp stem cells in regenerative medicine. Cytotherapy. 2018;20:479–98.
Tatullo M, Codispoti B, Paduano F, Nuzzolese M, Makeeva I. Strategic tools in regenerative and translational dentistry. Int J Mol Sci. 2019;20.
Abdel Meguid E, Ke Y, Ji J, El-Hashash AHK. Stem cells applications in bone and tooth repair and regeneration: New insights, tools, and hopes. J Cell Physiol. 2018;233:1825–35.
Ling LE, Feng L, Liu HC, Wang DS, Shi ZP, Wang JC, et al. The effect of calcium phosphate composite scaffolds on the osteogenic differentiation of rabbit dental pulp stem cells. J Biomed Mater Res A. 2015;103:1732–45.
Zhang W, Yelick PC. Tooth repair and regeneration: potential of dental stem cells. Trends Mol Med. 2021;27:501–11.
Zhang J, Ding H, Liu X, Sheng Y, Liu X, Jiang C. Dental follicle stem cells: tissue engineering and immunomodulation. Stem Cells Dev. 2019;28:986–94.
Morsczeck C, Schmalz G. Transcriptomes and proteomes of dental follicle cells. J Dent Res. 2010;89:445–56.
Luan X, Ito Y, Dangaria S, Diekwisch TG. Dental follicle progenitor cell heterogeneity in the develo** mouse periodontium. Stem Cells Dev. 2006;15:595–608.
Saito MT, Silverio KG, Casati MZ, Sallum EA, Nociti FH Jr. Tooth-derived stem cells: update and perspectives. World J Stem Cells. 2015;7:399–407.
Takahashi K, Ogura N, Aonuma H, Ito K, Ishigami D, Kamino Y, et al. Bone morphogenetic protein 6 stimulates mineralization in human dental follicle cells without dexamethasone. Arch Oral Biol. 2013;58:690–8.
Qi J, Yu T, Hu B, Wu H, Ouyang H. Current biomaterial-based bone tissue engineering and translational medicine. Int J Mol Sci. 2021;22.
Salazar VS, Gamer LW, Rosen V. BMP signalling in skeletal development, disease and repair. Nat Rev Endocrinol. 2016;12:203–21.
Supuran CT. Carbonic anhydrases–an overview. Curr Pharm Des. 2008;14:603–14.
Boron WF. Evaluating the role of carbonic anhydrases in the transport of HCO3−-related species. Biochim Biophys Acta. 2010;1804:410–21.
Chang X, Zheng Y, Yang Q, Wang L, Pan J, **a Y, et al. Carbonic anhydrase I (CA1) is involved in the process of bone formation and is susceptible to ankylosing spondylitis. Arthr Res Ther. 2012;14:R176.
Zheng Y, Xu B, Zhao Y, Gu H, Li C, Wang Y, et al. CA1 contributes to microcalcification and tumourigenesis in breast cancer. BMC Cancer. 2015;15:679.
Toyota A, Shinagawa R, Mano M, Tokioka K, Suda N. Regeneration in experimental alveolar bone defect using human umbilical cord mesenchymal stem cells. Cell Transplant. 2021;30:963689720975391.
Bajestan MN, Rajan A, Edwards SP, Aronovich S, Cevidanes LHS, Polymeri A, et al. Stem cell therapy for reconstruction of alveolar cleft and trauma defects in adults: a randomized controlled, clinical trial. Clin Implant Dent Relat Res. 2017;19:793–801.
Sun XC, Wang H, Li JH, Zhang D, Yin LQ, Yan YF, et al. Repair of alveolar cleft bone defects by bone collagen particles combined with human umbilical cord mesenchymal stem cells in rabbit. Biomed Eng Online. 2020;19:62.
Liu J, Yu F, Sun Y, Jiang B, Zhang W, Yang J, et al. Concise reviews: characteristics and potential applications of human dental tissue-derived mesenchymal stem cells. Stem Cells. 2015;33:627–38.
Xu L, Liu Y, Sun Y, Wang B, **ong Y, Lin W, et al. Tissue source determines the differentiation potentials of mesenchymal stem cells: a comparative study of human mesenchymal stem cells from bone marrow and adipose tissue. Stem Cell Res Ther. 2017;8:275.
Khojasteh A, Kheiri L, Motamedian SR, Nadjmi N. Regenerative medicine in the treatment of alveolar cleft defect: a systematic review of the literature. J Craniomaxillofac Surg. 2015;43:1608–13.
Bar JK, Lis-Nawara A, Grelewski PG. Dental Pulp stem cell-derived secretome and its regenerative potential. Int J Mol Sci. 2021;22:12018.
Lucaciu O, Soritau O, Gheban D, Ciuca DR, Virtic O, Vulpoi A, et al. Dental follicle stem cells in bone regeneration on titanium implants. BMC Biotechnol. 2015;15:114.
Guo W, Chen L, Gong K, Ding B, Duan Y, ** Y. Heterogeneous dental follicle cells and the regeneration of complex periodontal tissues. Tissue Eng Part A. 2012;18:459–70.
Sun J, Li J, Li H, Yang H, Chen J, Yang B, et al. tBHQ Suppresses osteoclastic resorption in xenogeneic-treated dentin matrix-based scaffolds. Adv Healthc Mater. 2017. https://doi.org/10.1002/adhm.201700127.
Li Z, Jiang CM, An S, Cheng Q, Huang YF, Wang YT, et al. Immunomodulatory properties of dental tissue-derived mesenchymal stem cells. Oral Dis. 2014;20:25–34.
Yang C, Li X, Sun L, Guo W, Tian W. Potential of human dental stem cells in repairing the complete transection of rat spinal cord. J Neural Eng. 2017;14:026005.
Laird NZ, Acri TM, Tingle K, Salem AK. Gene- and RNAi-activated scaffolds for bone tissue engineering: current progress and future directions. Adv Drug Deliv Rev. 2021;174:613–27.
James AW, LaChaud G, Shen J, Asatrian G, Nguyen V, Zhang X, et al. A review of the clinical side effects of bone morphogenetic protein-2. Tissue Eng Part B Rev. 2016;22:284–97.
Wang T, Zhang X, Bikle DD. Osteogenic differentiation of periosteal cells during fracture healing. J Cell Physiol. 2017;232:913–21.
Yuan L, Wang M, Liu T, Lei Y, Miao Q, Li Q, et al. Carbonic anhydrase 1-mediated calcification is associated with atherosclerosis, and methazolamide alleviates its pathogenesis. Front Pharmacol. 2019;10:766.
Vuola J, Bhling T, Kinnunen J, Hirvensalo E, Asko-Seljavaara S. Natural coral as bone-defect-filling material. J Biomed Mater Res. 2000;51:117–22.
Ripamonti U, Crooks J, Khoali L, Roden L. The induction of bone formation by coral-derived calcium carbonate/hydroxyapatite constructs. Biomaterials. 2009;30:1428–39.
Siller AF, Whyte MP. Alkaline phosphatase: discovery and naming of our favorite enzyme. J Bone Miner Res. 2018;33:362–4.
Millan JL, Whyte MP. Alkaline phosphatase and hypophosphatasia. Calcif Tissue Int. 2016;98:398–416.
Kemoun P, Laurencin-Dalicieux S, Rue J, Farges JC, Gennero I, Conte-Auriol F, et al. Human dental follicle cells acquire cementoblast features under stimulation by BMP-2/-7 and enamel matrix derivatives (EMD) in vitro. Cell Tissue Res. 2007;329:283–94.
Bottini M, Mebarek S, Anderson KL, Strzelecka-Kiliszek A, Bozycki L, Simao AMS, et al. Matrix vesicles from chondrocytes and osteoblasts: their biogenesis, properties, functions and biomimetic models. Biochim Biophys Acta Gen Subj. 2018;1862:532–46.
Florencio-Silva R, Sasso GR, Sasso-Cerri E, Simoes MJ, Cerri PS. Biology of bone tissue: structure, function, and factors that influence bone cells. Biomed Res Int. 2015;2015:421746.
Gamez B, Rodriguez-Carballo E, Ventura F. BMP signaling in telencephalic neural cell specification and maturation. Front Cell Neurosci. 2013;7:87.
Silverio KG, Davidson KC, James RG, Adams AM, Foster BL, Nociti Jr FH, et al. Wnt/beta-catenin pathway regulates bone morphogenetic protein (BMP2)-mediated differentiation of dental follicle cells. J Periodontal Res. 2012;47:309–19.
Pan K, Sun Q, Zhang J, Ge S, Li S, Zhao Y, et al. Multilineage differentiation of dental follicle cells and the roles of Runx2 over-expression in enhancing osteoblast/cementoblast-related gene expression in dental follicle cells. Cell Prolif. 2010;43:219–28.
Koo KT, Lee SW, Lee MH, Kim KH, Jung SH, Kang YG. Time-dependent expression of osteoblast marker genes in human primary cells cultured on microgrooved titanium substrata. Clin Oral Implants Res. 2014;25:714–22.
Rendl M, Polak L, Fuchs E. BMP signaling in dermal papilla cells is required for their hair follicle-inductive properties. Genes Dev. 2008;22:543–57.
Acknowledgements
Research was funded by grants from the National Natural Science Foundation of China (81070817) and Natural Science Foundation of Shandong Province (ZR2010HM054; ZR2015HM022). The funding body played no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Funding
National Natural Science Foundation of China, 81070817, Wen-lin **ao,Natural Science Foundation of Shandong Province, ZR2010HM054, Wen-lin **ao, ZR2015HM022, Wen-lin **ao
Author information
Authors and Affiliations
Contributions
**-ze Zhao and Ying-Ying Ge: conducted the experiments, performed data collection and/or assembly; data analysis and interpretation; and wrote the manuscript. Cong Li: data collection and analysis. Ling-fa Xue, Yao-xiang Xu, ** Yue and Wen-lin **ao: developed the study conception and design; performed data analysis and interpretation; administrative and manuscript proof.All authorsread and approvedthe manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical statement
This study was approved by the Ethics Committee of Qingdao University (Approval Number: QYFYWZLL27632), and all samples were obtained with informed consent from patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, Jz., Ge, YY., Xue, Lf. et al. CA1 Modulates the Osteogenic Differentiation of Dental Follicle Stem Cells by Activating the BMP Signaling Pathway In Vitro. Tissue Eng Regen Med (2024). https://doi.org/10.1007/s13770-024-00642-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13770-024-00642-4