Abstract
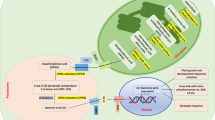
Cyclocarya paliurus (C. paliurus) leaves contain multiple health beneficial metabolites, and therefore are often used in functional foods and Chinese herbal medicines. However, breeding difficulties confines its utilization. Consequently, cell suspension cultures were developed to produce the bioactive secondary metabolites of C. paliurus leaves, but the content was comparatively low. In the present paper, Aspergillus niger elicitor (ANE) was used to stimulate the synthesis of triterpenoids in the suspension cultured C. paliurus cells, and the signal molecules crosstalk involved in this elicitation was further studied to interpret the underlying mechanism. Total triterpenoids accumulation in the cultured C. paliurus cells elicited by 200 µg/mL ANE was 9.17 times higher than that of the untreated cells. Nitric oxide (NO), hydrogen peroxide (H2O2) and jasmonic acid (JA) played important roles in the elicitation as signal molecules. Under the ANE stimulation, the concentrations of NO, H2O2 and JA all increased significantly, but presented different profiles and peaked at different time points. Based on series experiments on NO quenching by C-PTIO, H2O2 blocking by DMTU, and JA synthesis inhibition by IBU and NDGA, together with exogenous NO, H2O2 and JA addition experiments, it was concluded that ANE stimulated triterpenoid biosynthesis in the suspension culture of C. paliurus via a complex signal transduction network, in which three deduced and three hypothetical signal transduction pathways might be involved. JA was the critical point in the entire signal network, which connected with NO and H2O2 signal pathways. RNA-seq analysis showed that a total of three candidate JA synthesis pathway genes, including one LOX and two OPR genes, were significantly up-regulated under the ANE stimulation, along with five down-regulated JAZs and one up-regulated JAR1 regulating to JA signal transduction, which further validated the pivotal role of JA in ANE elicitation.







Similar content being viewed by others
Data availability
All relevant data are within the paper and its Supporting Information files.
Abbreviations
- SA:
-
Salicylic acid
- ANE:
-
Aspergillus niger elicitors
- SNP:
-
Sodium nitroprusside
- DEGs:
-
Differentially expressed genes
- JA:
-
Jasmonic acid
- NO:
-
Nitric oxide
- H2O2 :
-
Hydrogen peroxide
- FPKM:
-
Fragment per kilobases of exon model per million mapped reads
- FDR:
-
False discovery rate
- NOS:
-
Nitric oxide synthase
References
Afrin S, Huang JJ, Luo ZY (2015) JA-mediated transcriptional regulation of secondary metabolism in medicinal plants. Sci Bull 60:1062–1072. https://doi.org/10.1007/s11434-015-0813-0
Astier J, Loake G, Velikova V, Gaupels F (2016) Editorial: interplay between NO signaling, ROS and the antioxidant system in plants. Front Plant Sci 7:1731. https://doi.org/10.3389/fpls.2016.01731
Audic S, Claverie JM (1997) The significance of digital gene expression profiles. Genome Res 7:986–995. https://doi.org/10.1101/gr.7.10.986
Avanci N, Luche D, Goldman G, Goldman M (2010) Jasmonates are phytohormones with multiple functions, including plant defense and reproduction. Genet Mol Res 9:484–505. https://doi.org/10.4238/vol9-1gmr754
Bethke PC, Libourel IG, Reinöhl V, Jones RL (2006) Sodium nitroprusside, cyanide, nitrite, and nitrate break Arabidopsis seed dormancy in a nitric oxide-dependent manner. Planta 223:805–812. https://doi.org/10.1007/s00425-005-0116-9
Buraphaka H, Putalun W (2020) Stimulation of health-promoting triterpenoids accumulation in Centella asiatica (L.) Urban leaves triggered by postharvest application of methyl jasmonate and salicylic acid elicitors. Ind Crop Prod 146:112171. https://doi.org/10.1016/j.indcrop.2020.112171
Commault AS, Fabris M, Kuzhiumparambil U, Adriaans J, Pernice M, Ralph PJ (2019) Methyl jasmonate treatment affects the regulation of the 2-C-methyl-d-erythritol 4-phosphate pathway and early steps of the triterpenoid biosynthesis in Chlamydomonas reinhardtii. Algal Res 39:101462. https://doi.org/10.1016/j.algal.2019.101462
Conesa A, Götz S, García-Gómez JM, Terol J, Talón M, Robles M (2005) Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21:3674–3676. https://doi.org/10.1093/bioinformatics/bti610
Creelman RA, Tierney ML, Mullet JE (1992) Jasmonic acid/methyl jasmonate accumulate in wounded soybean hypocotyls and modulate wound gene expression. Proc Natl Acad Sci 89:4938–4941. https://doi.org/10.1073/pnas.89.11.4938
Domingos P, Prado AM, Wong A, Gehring C, Feijo JA (2015) Nitric oxide: a multitasked signaling gas in plants. Mol Plant 8:506–520. https://doi.org/10.1016/j.molp.2014.12.010
El-Bialy HA, El-Bastawisy HS (2020) Elicitors stimulate paclitaxel production by endophytic fungi isolated from ecologically altered Taxus baccata. J Radiat Res Appl Sci 13:79–87. https://doi.org/10.1080/16878507.2019.1702244
Ge Q, Zhang Y, Hua WP, Wu YC, ** XX, Song SH, Wang ZZ (2015) Combination of transcriptomic and metabolomic analyses reveals a JAZ repressor in the jasmonate signaling pathway of Salvia miltiorrhiza. Sci Rep 5:14048. https://doi.org/10.1038/srep14048
Guo CM, Yin ZP, Shangguan XC, Chen JG, Fu X (2014) Effect of sodium niterprusside on the biosynthesis of triterpenic acids in suspended cultured Cyclocarya paliurus cells. Mod Food Sci Technol 30:68–92. https://doi.org/10.13982/j.mfst.1673-9078.2014.04.017
Halder M, Sarkar S, Jha S (2019) Elicitation: a biotechnological tool for enhanced production of secondary metabolites in hairy root cultures. Eng Life Sci 19:880–895. https://doi.org/10.1002/elsc.201900058
Hidalgo D, Sanchez R, Lalaleo L, Bonfill M, Corchete P, Palazon J (2018) Biotechnological production of pharmaceuticals and biopharmaceuticals in plant cell and organ cultures. Curr Med Chem 25:3577–3596. https://doi.org/10.2174/0929867325666180309124317
Hu WB, Ouyang KH, Wu GQ, Chen H, **ong L, Liu X, Wang WJ (2018) Hepatoprotective effect of flavonoid-enriched fraction from Cyclocarya paliurus leaves on LPS/D-GalN-induced acute liver failure. J Funct Foods 48:337–350. https://doi.org/10.1016/j.jff.2018.07.031
Jiang C, Wang Y, ** Q, Zhang D, Gao M, Yao N, Ma S (2019) Cyclocarya paliurus triterpenoids improve diabetes-induced hepatic inflammation via the Rho-kinase dependent pathway. Front Pharmacol 10:811. https://doi.org/10.3389/fphar.2019.00811
Kümmritz S, Louis M, Haas C, Oehmichen F, Gantz S, Delenk H, Steingroewer J (2016) Fungal elicitors combined with a sucrose feed significantly enhance triterpene production of a Salvia fruticosa cell suspension. Appl Microbiol Biol 100:7071–7082. https://doi.org/10.1007/s00253-016-7432-9
Kunkel BN, Brooks DM (2002) Cross talk between signaling pathways in pathogen defense. Curr Opin Plant Boil 5:325–331. https://doi.org/10.1016/S1369-5266(02)00275-3
Li J, Brader G, Palva ET (2004) The WRKY70 transcription factor: a node of convergence for jasmonate-mediated and salicylate-mediated signals in plant defense. Plant Cell 16:319–331
Li T, Shangguan XC, Yin ZP, Chen JG, Jiang Y (2013) The separation and identification of triterpeniods from leaves of Cyclocarya paliurus. Acta Agric Univ Jiangxiensis 33:338–349. https://doi.org/10.3969/j.issn.1000-2286.2013.05.027
Li J, Liu S, Wang J, Li J, Liu D, Li J, Gao W (2016a) Fungal elicitors enhance ginsenosides biosynthesis, expression of functional genes as well as signal molecules accumulation in adventitious roots of Panax ginseng C. A. Mey. J Biotechnology 239:106–114. https://doi.org/10.1016/j.jbiotec.2016.10.011
Li J, Wang J, Li JX, Liu DH, Li HF, Gao WY, Li JL, Liu SJ (2016b) Aspergillus niger enhance bioactive compounds biosynthesis as well as expression of functional genes in adventitious roots of Glycyrrhiza uralensis Fisch. Appl Biochem Biotechnol 178:576–593. https://doi.org/10.1007/s12010-015-1895-5
Liang C, Chen C, Zhou P, Xu L, Zhu J, Liang J, Yu R (2018) Effect of Aspergillus flavus fungal elicitor on the production of terpenoid indole alkaloids in Catharanthus roseus cambial meristematic cells. Molecules 23:3276. https://doi.org/10.3390/molecules23123276
Liechti R, Farmer EE (2006) Jasmonate biochemical pathway. Sci Signal 2006:322. https://doi.org/10.1126/stke.3222006cm3
Lin WL, Hu XY, Zhang WQ, Rogers WJ, Cai WM (2005) Hydrogen peroxide mediates defence responses induced by chitosans of different molecular weights in rice. J Plant Physiol 162:937–944. https://doi.org/10.1016/j.jplph.2004.10.003
Liu ZB, Xu XX, Yin ZP, **ong QQ, Zhao WJ, Wang M, Chen JG, Shangguan XC (2016) Incentive mechanism of triterpenoid synthesis by antioxidant response in the cultured Cyclocarya paliurus cells under the elicitation of Aspergillus niger elicitor. Sci Technol Food Ind 3:7–13. https://doi.org/10.13386/j.issn1002-0306.2018.24.021
Liu Y, Chen P, Zhou M, Wang T, Fang S, Shang X, Fu X (2018a) Geographic variation in the chemical composition and antioxidant properties of phenolic compounds from Cyclocarya paliurus (Batal) Il**skaja leaves. Molecules 23:2440. https://doi.org/10.3390/molecules23102440
Liu ZB, Chen JG, Yin ZP, Shangguan XC, Peng DY, Lu T, Lin P (2018b) Methyl jasmonate and salicylic acid elicitation increase content and yield of chlorogenic acid and its derivatives in Gardenia jasminoides cell suspension cultures. PCTOC 134:79–93. https://doi.org/10.1007/s11240-018-1401-1
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using realtime quantitative PCR and the 2-△△Ct method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lv F, Li S, Feng J, Liu P, Gao Z, Yang Y, Wei J (2019) Hydrogen peroxide burst triggers accumulation of jasmonates and salicylic acid inducing sesquiterpene biosynthesis in wounded Aquilaria sinesis. J Plant Physiol 234:167–175. https://doi.org/10.1016/j.jplph.2019.02.006
Major I, Yoshida Y, Campos M, Kapali G, **n X, Sugimoto K, Howe G (2017) Regulation of growth–defense balance by the JASMONATE ZIM-DOMAIN (JAZ)-MYC transcriptional module. New Phytol 215:1533–1547. https://doi.org/10.1111/nph.14638
Menke FL, Parchmann S, Mueller MJ, Kijne JW, Memelink J (1999) Involvement of the octadecanoid pathway and protein phosphorylation in fungal elicitor-induced expression of terpenoid indole alkaloid biosynthetic genes in Catharanthus roseus. Plant Physiol 119:1289–1296. https://doi.org/10.1104/pp.119.4.1289
Mortazavi A, Williams BA, McCue K, Schaeffer L, Wold B (2008) Map** and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5:621–628. https://doi.org/10.1038/NMETH.1226
Mueller M (1997) Enzymes involved in jasmonic acid biosynthesis. Physiol Plantarum 100:653–663. https://doi.org/10.1111/j.1399-3054.1997.tb03072.x
Nojiri H, Sugimori M, Yamane H, Nishimura Y, Yamada A, Shibuya N, Omori T (1996) Involvement of jasmonic acid in elicitor-induced phytoalexin production in suspension-cultured rice cells. Plant Physiol 110:387–392. https://doi.org/10.1104/pp.110.2.387
Reiner A, Yekutieli D, Benjamini Y (2003) Identifying differentially expressed genes using false discovery rate controlling procedures. Bioinformatics 19:368–375. https://doi.org/10.1093/bioinformatics/btf877
Ren M, Shangguan XC, Chen JG, Jiang Y, Wu S, Yin ZP (2012) Effects of several factors on the growth of Cyclocarya paliurus suspension cells. Acta Agriculturae Boreali-Occidentalis Sinica 21:179–184. https://doi.org/10.1093/bioinformatics/btf877
Saxena I, Srikanth S, Chen Z (2016) Cross talk between H2O2 and interacting signal molecules under plant stress response. Front Plant Sci 7:570. https://doi.org/10.3389/fpls.2016.00570
Shangguan XC, Guo CL, Yang WY, Jiang Y, Shen YG (2006) Effects of basic media and culture conditions on callus growth and flavonoid content of Cyclocarya paliurus. J Fujian Agric for Univ. https://doi.org/10.3969/j.issn.1671-5470.2006.06.007
Staswick PE, Tiryaki I (2004) The oxylipin signal jasmonic acid is activated by an enzyme that conjugates it to isoleucine in Arabidopsis. Plant Cell 16:2117–2127. https://doi.org/10.1105/tpc.104.023549
Sun H, Tan J, Lv W, Li J, Wu J, Xu J, Xuan T (2019) Hypoglycemic triterpenoid glycosides from Cyclocarya paliurus (Sweet Tea Tree). Bioorg Chem 95:103493. https://doi.org/10.1016/j.bioorg.2019.103493
Toyoda K, Kawanishi Y, Kawamoto Y, Kurihara C, Yamagishi N, Tamura A, Shiraishi T (2013) Suppression of mRNAs for lipoxygenase (LOX), allene oxide synthase (AOS), allene oxide cyclase (AOC) and 12-oxo-phytodienoic acid reductase (OPR) in pea reduces sensitivity to the phytotoxin coronatine and disease development by Mycosphaerella pinodes. J Gen Plant Pathol 79:321–334
Turner JG, Ellis C, Devoto A (2002) The jasmonate signal pathway. Plant Cell 14(suppl 1):S153–S164. https://doi.org/10.1105/tpc.000679
**e MY, **e JH (2008) Review about the research on Cyclocarya paliurus (Batal.) Il**skaja. J Food Sci Biotechnol 1:113–121. https://doi.org/10.3321/j.issn:1673-1689.2008.01.021
**e JH, Liu X, Shen MY, Nie SP, Zhang H, Li C, **e MY (2013) Purification, physicochemical characterisation and anticancer activity of a polysaccharide from Cyclocarya paliurus leaves. Food Chem 136:1453–1460. https://doi.org/10.1016/j.foodchem.2012.09.078
**e J, Wang W, Dong C, Huang L, Wang H, Li C, **e M (2018) Protective effect of flavonoids from Cyclocarya paliurus leaves against carbon tetrachloride-induced acute liver injury in mice. Food Chem Toxicol 119:392–399. https://doi.org/10.1016/j.fct.2018.01.016
**ong QQ, Yin ZP, Chen JG, Shangguang XC, Jiang Y, Peng DY (2016) Aspergillus Niger inducer stimulate the biosynthesis of triterpenoids in suspension-cultured Cyclocarya paliurus cells. Food Sci Technol 41:7–13. https://doi.org/10.13684/j.cnki.spkj.2016.03.002
**ong L, Ouyang KH, Jiang Y, Yang ZW, Hu WB, Chen H, Wang WJ, (2018) Chemical composition of Cyclocarya paliurus polysaccharide and inflammatory effects in lipopolysaccharide-stimulated RAW264.7 macrophage. Int J Biol Macromol 107:1898–1907. https://doi.org/10.1016/j.ijbiomac.2017.10.055
Xu MJ, Dong JF (2006) Nitric oxide mediates the fungal elicitor-induced Taxol biosynthesis of Taxus chinensis suspension cells through the reactive oxygen species-dependent and-independent signal pathways. Chin Sci Bull 51:1967–1975. https://doi.org/10.1007/s11434-006-2081-5
Xu MJ, Dong JF, Zhu MY (2005) Nitric oxide mediates the fungal elicitor-induced hypericin production of Hypericum perforatum cell suspension cultures through a jasmonic-acid-dependent signal pathway. Plant Physiol 139:991–998. https://doi.org/10.1104/pp.105.066407
Xu MJ, Dong JF, Zhu MY (2006) Nitric oxide mediates the fungal elicitor-induced puerarin biosynthesis in Pueraria thomsonii Benth. suspension cells through a salicylic acid (SA)-dependent and a jasmonic acid (JA)-dependent signal pathway. Sci China Ser C 49:379–389. https://doi.org/10.1007/s11427-006-2010-5
Xuan TY, Tan J, Sun HH, Yang C, Lv WY, Zhang JH, Zhu GZ (2019) Cyclocarioside O-Q, three novel seco-dammarane triterpenoid glycosides from the leaves of Cyclocarya paliurus. Nat Prod Res. https://doi.org/10.1080/14786419.2019.1616722
Yan K, He L, Yang K (2020) Effects of SA and H2O2 mediated endophytic fungal elicitors on essential oil in suspension cells of cinnamomum longepaniculatum. Oalib J 7:1–12. https://doi.org/10.4236/oalib.1106009
Yang ZW, Ouyang KH, Zhao J, Chen H, **ong L, Wang WJ (2016) Structural characterization and hypolipidemic effect of Cyclocarya paliurus polysaccharide in rat. Int J Biol Macromol 91:1073–1080. https://doi.org/10.1016/j.ijbiomac.2016.06.063
Yang Z, Wang J, Li J, **ong L, Chen H, Liu X, Wang W (2018) Antihyperlipidemic and hepatoprotective activities of polysaccharide fraction from Cyclocarya paliurus in high-fat emulsion-induced hyperlipidaemic mice. Carbohydr Polym 183:11–20. https://doi.org/10.1016/j.carbpol.2017.11.033
Ye J, Fang L, Zheng H, Zhang Y, Chen J, Zhang Z, Wang J (2006) WEGO: a web tool for plotting GO annotations. Nucleic Acids Res 34:293–297. https://doi.org/10.1093/nar/gkl031
Yin ZP, Shangguan XC, Chen JG, Wu S, Li D (2013a) Separation and identification of triterpenic acids from suspended cultured cells of Cyclocarya paliurus. Scientia Silvae Sinicae 49:23–28
Yin ZP, Shangguan XC, Chen JG, Zhao Q, Li DM (2013b) Growth and triterpenic acid accumulation of Cyclocarya paliurus cell suspension cultures. Biotechnol Bioprocess Eng 18:606–614. https://doi.org/10.1007/s12257-012-0751-5
Yin ZP, **ong QQ, Shangguan XC, Chen JG (2017) Chemical fingerprint and determination of triterpenoids in cultured Cyclocarya paliurus cells by high-performance liquid chromatography. Anal Lett 50:748–760. https://doi.org/10.1080/00032719.2016.1199026
Zhang Y, Li X (2019) Salicylic acid: biosynthesis, perception, and contributions to plant immunity. Curr Opinn Plant Boil 50:29–36. https://doi.org/10.1016/j.pbi.2019.02.004
Acknowledgements
The authors are grateful for the assistance in RNA-seq determination and its related analysis from Sangon Biotech (Shanghai) Co., Ltd.
Funding
The authors are grateful for the support from the National Natural Science Foundation of China (31960515), Jiangxi Province Natural Science Foundation (20192BAB204004) and Guangzhou Science and Technology Project (201909020001).
Author information
Authors and Affiliations
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, M., Tang, D., **ong, Q. et al. Mechanism of molecules crosstalk and the critical role of jasmonic acid on triterpenoid synthesis in Cyclocarya Paliurus cells under Aspergillus niger elicitor. J. Plant Biochem. Biotechnol. 31, 864–879 (2022). https://doi.org/10.1007/s13562-021-00764-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13562-021-00764-z