Abstract
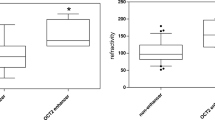
Organic cation transporter 2 (rOCT2) and multidrug and toxin extrusion protein 1 (rMATE1) are mainly expressed in rat renal proximal tubules and mediate urinary excretion of cationic drugs, such as metformin. Accumulated evidence indicated that renal rOCT2 expression in male rats is much higher than that of female rats. However, it is unclear whether the gender-related differences in rOCT2 expression between male and female rats can affect the urinary excretion of metformin. The aim of this study was to investigate the effect of gender on the pharmacokinetics of metformin and to clarify the effect of gender-related differences on renal rOCT2 expression and its role in urinary excretion of metformin. Renal rOCT2 levels, but not rOCT1 and rMATE1, were significantly lowered in female rats when compared to that of male rats (P < 0.01), while the pharmacokinetic parameters, i.e., AUC0→t, t 1/2, CL/F, and cumulative urinary excretion of metformin, did not show any significant differences between female and male rats following oral administration of metformin at l00 mg/kg (P > 0.05). However, when metformin was orally administered at the dose of 500 mg/kg, the cumulative urinary excretion and renal tissue-to-plasma concentration ratio of metformin in female rats (26,689 ± 1266 μg and 2.96 ± 0.47 mL/g, respectively) were markedly lowered compared to that of male rats (32,949 ± 1384 μg and 4.20 ± 0.31 mL/g, respectively), and the plasma concentration of metformin in female rats (55.9 ± 4.5 μg/mL) was significantly increased compared to that of male rats (43.5 ± 3.1 μg/mL) at 2 h after oral administration. These results indicated that effect of gender-related differences on renal rOCT2 expression indeed contributes to the decreased urinary excretion of metformin in female rats when metformin was administered at relatively high doses.




Similar content being viewed by others
References
Gleiter CH, Gundert-Remy U. Gender differences in pharmacokinetics. Eur J Drug Metab Pharmacokinet. 1996;21(2):123–8.
Harris RZ, Benet LZ, Schwartz JB. Gender effects in pharmacokinetics and pharmacodynamics. Drugs. 1995;50(2):222–39.
Tanaka E. Gender-related differences in pharmacokinetics and their clinical significance. J Clin Pharm Ther. 1999;24(5):339–46.
Franconi F, Brunelleschi S, Steardo L, Cuomo V. Gender differences in drug responses. Pharmacol Res. 2007;55(2):81–95.
Meibohm B, Beierle I, Derendorf H. How important are gender differences in pharmacokinetics? Clin Pharmacokinet. 2002;41(5):329–42.
Bebawy M, Chetty M. Gender differences in p-glycoprotein expression and function: effects on drug disposition and outcome. Curr Drug Metab. 2009;10(4):322–8.
Buckley DB, Klaassen CD. Tissue- and gender-specific mRNA expression of UDP-glucuronosyltransferases (UGTs) in mice. Drug Metab Dispos. 2007;35(1):121–7.
Morris ME, Lee HJ, Predko LM. Gender differences in the membrane transport of endogenous and exogenous compounds. Pharmacol Rev. 2003;55(2):229–40.
Ou-Yang DS, Huang SL, Wang W, **e HG, Xu ZH, Shu Y, Zhou HH. Phenotypic polymorphism and gender-related differences of CYP1A2 activity in a Chinese population. Br J Clin Pharmacol. 2000;49(2):145–51.
Rademaker M. Do women have more adverse drug reactions? Am J Clin Dermatol. 2001;2(6):349–51.
Koepsell H. Polyspecific organic cation transporters: their functions and interactions with drugs. Trends Pharmacol Sci. 2004;25(7):375–81.
Koepsell H, Endou H. The SLC22 drug transporter family. Pflugers Arch. 2004;447(5):666–76.
Yonezawa A, Inui K. Organic cation transporter OCT/SLC22A and H(+)/organic cation antiporter MATE/SLC47A are key molecules for nephrotoxicity of platinum agents. Biochem Pharmacol. 2011;81(5):563–8.
Li X, Zhang N, Li Y, Shi Y, Li D, **e Y, Ming J. Effects of metformin and rosiglitazone on peripheral insulin resistance and beta-cell function in obesity: a double-blind, randomized, controlled study. J Int Med Res. 2011;39(2):358–65.
Ou HY, Cheng JT, Yu EH, Wu TJ. Metformin increases insulin sensitivity and plasma beta-endorphin in human subjects. Horm Metab Res. 2006;38(2):106–11.
Graham GG, Punt J, Arora M, Day RO, Doogue MP, Duong JK, Furlong TJ, Greenfield JR, Greenup LC, Kirkpatrick CM, Ray JE, Timmins P, Williams KM. Clinical pharmacokinetics of metformin. Clin Pharmacokinet. 2011;50(2):81–98.
Scheen AJ. Clinical pharmacokinetics of metformin. Clin Pharmacokinet. 1996;30(5):359–71.
Kimura N, Masuda S, Tanihara Y, Ueo H, Okuda M, Katsura T, Inui K. Metformin is a superior substrate for renal organic cation transporter OCT2 rather than hepatic OCT1. Drug Metab Pharmacokinet. 2005;20(5):379–86.
Takane H, Shikata E, Otsubo K, Higuchi S, Ieiri I. Polymorphism in human organic cation transporters and metformin action. Pharmacogenomics. 2008;9(4):415–22.
Urakami Y, Nakamura N, Takahashi K, Okuda M, Saito H, Hashimoto Y, Inui K. Gender differences in expression of organic cation transporter OCT2 in rat kidney. FEBS Lett. 1999;461(3):339–42.
Urakami Y, Okuda M, Saito H, Inui K. Hormonal regulation of organic cation transporter OCT2 expression in rat kidney. FEBS Lett. 2000;473(2):173–6.
Lickteig AJ, Cheng X, Augustine LM, Klaassen CD, Cherrington NJ. Tissue distribution, ontogeny and induction of the transporters multidrug and toxin extrusion (MATE) 1 and MATE2 mRNA expression levels in mice. Life Sci. 2008;83(1–2):59–64.
Ito S, Kusuhara H, Yokochi M, Toyoshima J, Inoue K, Yuasa H, Sugiyama Y. Competitive inhibition of the luminal efflux by multidrug and toxin extrusions, but not basolateral uptake by organic cation transporter 2, is the likely mechanism underlying the pharmacokinetic drug–drug interactions caused by cimetidine in the kidney. J Pharmacol Exp Ther. 2012;340(2):393–403.
Li Q, Guo D, Dong Z, Zhang W, Zhang L, Huang S-M, Polli JE, Shu Y. Ondansetron can enhance cisplatin-induced nephrotoxicity via inhibition of multiple toxin and extrusion proteins (MATEs). Toxicol Appl Pharmacol. 2013;273(1):100–9.
Konig J, Zolk O, Singer K, Hoffmann C, Fromm MF. Double-transfected MDCK cells expressing human OCT1/MATE1 or OCT2/MATE1: determinants of uptake and transcellular translocation of organic cations. Br J Pharmacol. 2011;163(3):546–55.
Lepist EI, Zhang X, Hao J, Huang J, Kosaka A, Birkus G, Murray BP, Bannister R, Cihlar T, Huang Y, Ray AS. Contribution of the organic anion transporter OAT2 to the renal active tubular secretion of creatinine and mechanism for serum creatinine elevations caused by cobicistat. Kidney Int. 2014;86(2):350–7.
Strobel J, Muller F, Zolk O, Endress B, Konig J, Fromm MF, Maas R. Transport of asymmetric dimethylarginine (ADMA) by cationic amino acid transporter 2 (CAT2), organic cation transporter 2 (OCT2) and multidrug and toxin extrusion protein 1 (MATE1). Amino Acids. 2013;45(4):989–1002.
Jonker JW, Wagenaar E, Van Eijl S, Schinkel AH. Deficiency in the organic cation transporters 1 and 2 (Oct1/Oct2 [Slc22a1/Slc22a2]) in mice abolishes renal secretion of organic cations. Mol Cell Biol. 2003;23(21):7902–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ma, Yr., Qin, Hy., **, Yw. et al. Gender-Related Differences in the Expression of Organic Cation Transporter 2 and its Role in Urinary Excretion of Metformin in Rats. Eur J Drug Metab Pharmacokinet 41, 559–565 (2016). https://doi.org/10.1007/s13318-015-0278-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13318-015-0278-1