Abstract
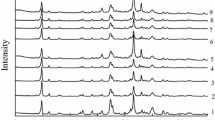
The paper presents original results regarding the effect of long-term treatment of phlogopite concentrate with nitric acid on the phase ratio, chemical composition and catalytic activity of copper–palladium complexes in the reaction of carbon monoxide oxidation with air oxygen. Phlogopite concentrate is polyphasic and contains phases of phlogopite, clinochlore, diopside and tremolite. Samples were modified with 8M HNO3 at room temperature for 1; 24; 48; 72 h (8H-Phl-τ). Samples of 8H-Phl-τ (\(\stackrel{\mathrm{-}}{\text{S}}\)) and Pd(II)–Cu(II)/\(\stackrel{\mathrm{-}}{\text{S}}\) catalysts were characterized by XRD, SEM, FT-IR spectroscopy and pH metric methods. It is shown that the content of the phlogopite phase decreases in a number of these samples, while the content of the clinochlore phase increases. The content of diopside and tremolite phases varies irregularly and remains within the limits for the original sample. Long-term acid modification (72 h) leads to amorphization of the sample and the formation of nanosilica: the silicon content increases to 92 wt. %. The Wacker-type catalyst fixed on nanosilica showed a high degree (97%) of carbon monoxide conversion.





Similar content being viewed by others
Data Availability
The data sets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Bailey SW (1980) Structure of layer silicates. In: Crystal structures of clay minerals and their X-ray identification. Mineralogical Society of Great Britain and Ireland. p. 1–125 https://doi.org/10.1180/mono-5.1
Campopiano A, Olori A, Cannizzaro A, Iannò A, Capone PP (2015) Quantification of tremolite in friable material coming from Calabrian ophiolitic deposits by infrared spectroscopy. J Spectrosc. https://doi.org/10.1155/2015/974902
Chheda TD, Mookherjee M, Mainprice D, Dos Santos AM, Molaison JJ, Chantel J, Bassett WA (2014) Structure and elasticity of phlogopite under compression: geophysical implication. Phys Earth Planet Inter 233:1–12. https://doi.org/10.1016/j.pepi.2014.05.004
CrystalMaker (2022) Crystal structure report. S090-Diopsid. www.crystalmaker.com
Deysel HM, Berluti K, du Plessis BJ, Focke WW (2020) Glass foams from acid-leached phlogopite waste. J Mater Sci 55(19):8050–8060. https://doi.org/10.1007/s10853-020-04589-4
Godymchuk AYu, Ilyin AP (2003) Research of sorption processes on natural minerals and their thermally modified forms. Chem Technol Water 6:621–632
Harkonen MA, Keishi RL (1984) Porosity and Surface Area of Acid – Leached Phlogopite: The effect of leaching conditions and thermal treatment. Colloids Surf 11:323–339. https://doi.org/10.1016/0166-6622(84)80287-5
Hemanthkumar GN, Parthasarathy G, Chakradhar RPS, Omkaram I, Lakshmana RJ, Ratnakaram YC (2009) Electron paramagnetic resonance studies on clinochlore from Longitudinal Valley area, northeastern Taiwan. Phys Chem Miner 36(8):447–453. https://doi.org/10.1007/s00269-009-0291-5
Jang SC, Kim GY, Hong SB, Yang HM, Lee KW, Moon JK, Seo BK, Cho Y, Huh YS, Roh C (2015) Magnetic composites as an effective technology for removal of radioactive cesium. Int J Environ Sci Technol 12:3695–3700. https://doi.org/10.1007/s13762-015-0853-7
Kaviratna H, Pinnavaia TM (1984) Acid Hydrolysis of Octahedral Mg sites in 2:1 layered silicates: an assessment of edge attack and the gallery access mechanisms. Clays Clay Miner 42(6):717–723. https://doi.org/10.1346/CCMN.1994.0420607
Kiose TA, Rakitskaya TL, Ennan AAA, Popruha YuI (2022) Nanocatalysts for carbon monoxide oxidation based on the acid modified polyphase aluminosilicate support and contained palladium(II) and copper(II) salts. Acta Physica Polonica A 141(4):286–292. https://doi.org/10.12693/APhysPolA.141.286
Kong M, Vogt T, Lee Y (2018) High-pressure synchrotron X-ray diffraction study of tremolite and actinolite in various fluids. Curr Appl Phys 18(11):1218–1224. https://doi.org/10.1016/j.cap.2018.05.018
Kuwahara Y, Aoki Y (1995) Dissolution process of phlogopite in acid solutions. Clays Clay Miner 43(1):39–50. https://doi.org/10.1346/CCMN.1995.0430105
Lipshutz BH, Gholinejad M, Oftadeh E, Shojafar M, Sansano JM (2019) Synergistic effects of ppm levels of palladium on natural clinochlore: a new reagent for reductions of nitroarenes. ChemSusChem 12(18):4240–4248. https://doi.org/10.1002/cssc.201901535
Luo Z, Yang J, Ma H, Liu M, Ma X (2015) Recovery of magnesium and potassium from biotite by sulfuric acid leaching and alkali precipitation with ammonia. Hydrometallurgy 157:188–193. https://doi.org/10.1016/j.hydromet.2015.08.018
Nazar A, Rakitskaya T, Kiose T (2022) Influence of acid modification of natural phlogopite on catalytic activity of supported Pd(II)-Cu(II) complexes in the reaction of oxidation of carbon monoxide by atmospheric oxygen. Chem J Moldova 17(1):47–55. https://doi.org/10.19261/cjm.2022.927
Park ED, Lee JS (2000) Effect of surface treatment of the support on CO oxidation over carbon-supported Wacker type catalysts. J Catal 193:5–15. https://doi.org/10.1006/jcat.2000.2879
Polshettiwar V, Len A, Fihri A (2009) Silica-supported palladium: Sustainable catalysts for cross-coupling reactions. Coord Chem Rev 253(21–22):2599–2626. https://doi.org/10.1016/j.ccr.2009.06.001
Radkevich VZ, Sen’ko TL, Khaminets SG, Wilson K, Egiazarov YuG (2008) Catalytic systems based on carbon supports for the low-temperature oxidation of carbon monoxide. Kinet Catal 49:545–551. https://doi.org/10.1134/s0023158408040149
Radkevich VZ, Wilson K, Khaminets SG, Sen’ko TL (2014) Effect of preparation conditions on the formation of the active phase of carbon fiber catalytic systems for the low-temperature oxidation of carbon monoxide. Kinet Catal 55:252–267. https://doi.org/10.1134/s0023158414020086
Rakitskaya TL, Kiose TA, Zryutina AM, Gladyshevskii RE, Truba AS, Vasylechko VO, Demchenko PYu, Gryschouk GV, Volkova VYa (2013) Solid-state catalysts based on bentonites and Pd(II)-Cu(II) complexes for low temperature carbon monoxide oxidation. Solid State Phenom 200:299–304. https://doi.org/10.4028/www.scientific.net/SSP.200.299
Rakitskaya TL, Kiose TA, Golubchik KO, Ennan AA, Volkova VY (2017) Acid-modified clinoptilolite as a support for palladium-copper complexes catalyzing carbon monoxide oxidation with air oxygen. Chem Cent J 11:1–10. https://doi.org/10.1186/s13065-017-0256-6
Rakitskaya T, Dzhyga G, Kiose T, Volkova V (2020a) Natural nanobentonites as supports in palladium(II)–copper(II) catalysts for carbon monoxide oxidation with air oxygen. Nanostructure Surf Appl 247:141–157. https://doi.org/10.1007/978-3-030-52268-1_11
Rakitskaya TL, Kiose TA, Ennan AA (2020b) Conceptual foundations for the development of low-temperature catalysts for the carbon monoxide oxidation with atmospheric oxygen. ONU Herald. Chemistry 25(4):6–23. https://doi.org/10.18524/2304-0947.2020.4(76).216920
Rakitskaya TL, Kiose TA, Truba AS, Ennan AA (2022) Effect of water on activity and protective properties of catalysts used in respiratory protective equipment. Handb Res Water Sci Soc 2:469–499. https://doi.org/10.4018/978-1-7998-7356-3.ch021
Rodriguez-Carvajal J, Roisnel T (1998) “FullProf. 98 and WinPLOTR: new windows 95/NT applications for diffraction. Comm Powder Diffr Int Union Crystallogr Newsl 20:35–36
Ross GJ (1967) Kinetics of acid dissolution of an orthochlorite mineral. Can J Chem. https://doi.org/10.1139/v67-491
Rufe E (2000) Assessing the reactive surface area of phlogopite during acid dissolution: an atomic force microscopy, X-ray photoelectron spectroscopy, and low energy electron diffraction study. Thesis, Blacksburg, Virginia
Schroeder PA (2002) Infrared spectroscopy in clay science. In: Rule A, Guggenheim S (eds) CMS workshop lectures, Teaching Clay Science, The Clay Mineral Society, Aurora, CO, vol 11. p 181–206. https://doi.org/10.1346/CMS-WLS-11.11
Shen Y, Lu G, Guo Y, Wang Y, Guo Y, Gong X (2011a) Study on the catalytic reaction mechanism of low temperature oxidation of CO over Pd-Cu-Clx/Al2O3 catalyst. Catal Today 175:558–567. https://doi.org/10.1016/j.cattod.2011.03.042
Shen YX, Guo Y, Wang L, Wang YQ, Guo YL, Gong XQ, Lu GZ (2011b) The stability and deactivation of Pd-Cu-Clx/Al2O3 catalyst for low temperature CO oxidation: an effect of moisture. Catal Sci Technol 1:1202–1207. https://doi.org/10.1039/C1CY00146A
Spinnler GE, Self PG, Iijima S, Buseck PR (1984) Stacking disorder in clinochlore chlorite. Am Miner 69(3–4):252–263
Tamura K, Kogure T, Watanabe Y, Nagai C, Yamada H (2014) Uptake of cesium and strontium ions by artificially altered phlogopite. Environ Sci Technol 48:5808–5815. https://doi.org/10.1021/es4052654
Vassileva RD, Bonev IK (2008) Mineralogy of diopside-phlogopite marbles from the Modi-Khola valley, the Central Nepal Himalaya. Geochem Mineral Petrol 46:69–84
Venter I (2015) Mesoporous silica recovery from phlogopite by aqua regia leaching. Ph.D. Thesis, University of Pretoria
Ventura DG, Caprilli E, Bellatreccia F, De Benedetti AA, Mottana A (2014) Asbestiform tremolite within the Holocene late pyroclastic deposits of Colli Albani volcano (Latium, Italy): occurrence and crystal chemistry. Rendiconti Lincei 25(2):229–236. https://doi.org/10.1007/s12210-013-0283-8
Villieras F, Yvon J, Cases JM, De Donato P, Lhote F, Baeza R (1994) Development of microporosity in clinochlore upon heating. Clays Clay Miner 42(6):679–688. https://doi.org/10.1346/CCMN.1994.0420604
Wang L, Zhou Y, Liu Q, Guo Y, Lu G (2010) Effect of surface properties of activated carbon on CO oxidation over supported Wacker-type catalysts. Catal Today 153:184–188. https://doi.org/10.1016/j.cattod.2010.02.067
Wang Y, Shi J, Wu R, Li X, Zhao Y (2016) Room-temperature CO oxidation over calcined Pd–Cu/palygorskite catalysts. Appl Clay Sci 119:126–131. https://doi.org/10.1016/j.clay.2015.08.034
Wu H, Qiang S, Fan Q, Zhao X, Liu P, Li P, Liang J, Wu W (2018) Exploring the relationship between Th(IV) adsorption and the structure alteration of phlogopite. Appl Clay Sci 152:295–302. https://doi.org/10.1016/j.clay.2017.11.026
Yamamoto T, Fujimura T, Shimada T, Takagi S (2014) Preparation of modified mica as an effective adsorbent to remove Cs+ from water. Chem Lett 43:860–861. https://doi.org/10.1246/cl.140089
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not for-profit sectors.
Author information
Authors and Affiliations
Contributions
All authors contributed to the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical standard
This article does not contain any studies with humans or animals.
Informed consent
On behalf of other authors, informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rakitskaya, T., Nazar, A., Kiose, T. et al. Catalyst containing natural nanosilica, palladium(II) and copper(II) salts in oxidation of carbon monoxide with oxygen. Appl Nanosci 13, 6777–6786 (2023). https://doi.org/10.1007/s13204-023-02772-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-023-02772-y