Abstract
A boy aged 55 months was diagnosed with stage IV Neuroblastoma (NB) of the right adrenal gland 2 years ago. Preoperative chemotherapy was given and he was then treated with retroperitoneal tumor resection and lymph node dissection. After surgery, the children were transferred to the Hemato-Oncology Department for chemotherapy according to the high-risk group NB, with outpatient follow-up every 6 months. In the second postoperative year, abdominal computed tomography (CT) scan revealed a rounded hypodense area in the upper part of the right posterior lobe of the liver, with marked inhomogeneous enhancement in the venous phase after enhancement, which was surgically resected, and postoperative pathology confirmed inflammatory myofibroblastic tumor (IMT) of liver. The patient was not given any special treatment after surgery. In this study, whole transcriptome sequencing was performed on the postoperative specimen of adrenal NB and the specimen of IMT of liver. This unusual case emphasizes the need for close monitoring of second tumor development in NB survivors even in the absence of known predisposing factors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Background
With improved diagnostic and treatment modalities for neuroblastoma (NB), the prognosis for patients is getting better, but the risk of patients develo** a second tumor remains [1]. A so-called second tumor is another tumor that develops near or away from the primary tumor [2]. The appearance of a mass in the organism after the complete cure of a malignant tumor does not necessarily mean the recurrence of the tumor or the development of another malignant tumor [3]. In addition, the development of a second tumor may or may not be related to the treatment of the prior tumor, as genetic risk factors or other external carcinogens may also be involved [4]. The development of a second malignant tumor is one of the most devastating events affecting the survival of these patients [5, 6]. Studies suggest that the younger the cancer survivor, the more deadly the second primary malignancy [7]. Another study reported malignant gastrointestinal neuroectodermal tumour as the second primary malignant tumor after pediatric NB treatment [8]. IMT is a rare inert tumor that has been less frequently reported as a second tumor occurring after treatment of malignancy in pediatric patients, with cases of IMT occurring after treatment of pediatric nephroblastoma reported [9]. Characterized by spindle cell proliferation and inflammatory cell infiltration, IMT is an intermediate state tumor with malignant potential, and therefore should be given adequate attention when it appears as a second tumor [10]. This study reports a case of a child who developed liver inflammatory myofibroblastic tumor (IMT) after NB.
2 Method
This study was reviewed by the ethics committee of the Children’s Hospital of Nan**g Medical University, batch number: NJCH2020137. The study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The legal guardians/next of kin of the participants provided written informed consent for participation in this study. Written informed consent for the publication of any potentially identifiable images or data contained herein was obtained from the legal guardian/next of kin of the minor.
2.1 Case presentation
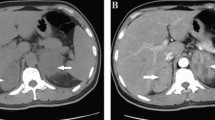
A male child, aged 28 months, was admitted to the hospital for examination in 2018 when he was found to have a frontal parietal mass with fever for 6 days. Chest and whole abdomen CT scan showed soft tissue density mass shadow of the right adrenal gland, with multiple small nodular calcified foci seen internally; multiple nodular calcified foci seen in the retroperitoneum and pelvis, which was considered as NB with possible retroperitoneal pelvic multiple lymph node metastases (Fig. 1A). Cranial CT scan revealed thickening of the cranial plate of the frontal parietal bone on both sides, radiolucent periosteal reaction was seen in the inner plate, and soft tissue mass formation was seen in the surrounding area, metastasis was considered (Fig. 1B). The child underwent cranial tumor biopsy on 3/23/2018, and postoperative pathology showed a small round cell malignancy, and NB metastasis was considered in conjunction with the child’s clinical history and immunohistochemistry results. In this study, we used fluorescence in situ hybridization (FISH) to detect the amplification rate of the MYCN gene, and a MYCN (green signal)/CEP2 (orange signal) ratio > 4 was considered positive, i.e., MYCN amplification; a MYCN/CEP2 ratio ≥ 1 and < 4 was considered gain; and a MYCN/CEP2 ratio < 1 was considered negative. MYCN gene test result was gain (Fig. 2).
The child was diagnosed with NB, stage IV, and was included in the ultra-high-risk group because of cranial, dural, and multiple vertebral metastases, and was subsequently transferred to the Hemato-Oncology Department for chemotherapy. Since 11/04/2018 patient was given 3 courses of chemotherapy in the ultra-high risk group regimen for NB (Course I: cyclophosphamide, topotecan; Course II: topotecan, cyclophosphamide; Course III: pedialyte glycosides, cisplatin). On June 29, 2018, the patient underwent retroperitoneal tumor resection and retroperitoneal lymph node dissection in the Department of General Surgery of Nan**g Children’s Hospital. Postoperative pathology revealed post-chemotherapy changes in NB (differentiated type) with lymph node metastasis (Fig. 3). Transferred to Hemato-Oncology for chemotherapy after surgery (Course 1: CTX + TOPO; Course 2: CTX + TOPO; Course 3: CDDP + VP16; Course 4: CTX + DOXO + VCR + MESNA; Course 5: CDDP + VP16; Course 6: CTX + DOXO + VCR + MESNA; Course 7: CTX + TOPO; Course 8: CDDP + VP16; Course 9: CTX + DOXO + VCR + MESNA; Course 10: CTX + TOPO) and the abdominal CT was reviewed regularly.
The child underwent a routine postoperative thoracic and abdominal CT review in June 2020, which revealed an abnormal occupancy in the upper right posterior lobe of the liver. Further examination of the whole abdomen using CT scan with contrast showed that the right lobe of the liver was visible as a classically rounded low-density area with an extent of about 22 × 24 mm, and enhancement reveals inhomogeneous enhancement, which is evident in the venous phase, within which enhanced nodules are also seen.; NB liver metastasis is considered (Fig. 4). Subsequently, hepatic anomalous occupancy resection was performed in our General Surgery Department, and the occupancy was anatomically resected using indocyanine green fluorescence navigated hepatic occupancy resection, and postoperative pathology showed IMT (Fig. 5A). Ventana D5F3 immunohistochemical staining showed positive anaplastic lymphoma kinas (ALK), consistent with the diagnosis of IMT (Fig. 5B). Whole transcriptome sequencing of the adrenal NB specimen and the IMT of liver revealed a large number of differentially expressed genes (Fig. 6). A VIT chemotherapy regimen (Vincristine, Irinotecan and Temozolomide) was used postoperatively with outpatient follow-up.
3 Discussion
Comprehensive treatment of NB is associated with multiple complications, including cognitive deficits, cardiotoxicity, and chronic kidney disease [1, 11, 12]. In addition, many individual cases have reported a second tumor after treatment [13, 14]. IMT of the Liver is a tumor-like lesion characterized by fibrous tissue, capillary hyperplasia, and local tissue inflammatory cell infiltration following necrosis of liver tissue caused by various inflammatory factors. The condition as a second tumor is reported for the first time [15].
When two tumors appear in adjacent locations at different times, multiple etiologies may be involved [16]. The second tumor may represent an extension or metastasis of the primary tumor [17]. Irradiation and drug therapy used as adjuvant modalities during the preoperative and postoperative period may have contributed to the development of the new tumor [18]. In addition, a patient’s genetic susceptibility to cancer, other environmental factors, or unknown causes may also lead to the development of a second tumor [19, 20]. Low-dose irradiation is known to induce a second tumor [21]. In general, the following criteria should be followed for the diagnosis of radiation tumors [22]: the new lesion appears in the radiated area; it is histologically distinct from the first tumor; it develops after a latent period that reasonably allows for the development of a secondary tumor, and there are no other conditions that predispose to tumor development. And the patient's medical records showed no other genetic or environmental factors predisposing him to cancer. Based on the fact that the patient received prior chemotherapy, the authors of this article do not exclude the possibility that chemotherapy may have acted as a causative factor in the development of hepatic IMT. However, it is not entirely clear whether this is the case.
In addition, in this study, whole transcriptome next-generation sequencing of adrenal NB specimens and IMT of liver specimens were performed. Whole transcriptome sequencing showed complete discordance in gene expression between adrenal NB specimens and liver IMT specimens, this transcriptomic level analysis again validated the non-homologous nature of the patient's two successive tumors. Genetic studies have similarly shown that rearrangement of the ALK gene may contribute to tumorigenesis, suggesting that IMT is likely to occur as a tumor entity and is not a reactive progression due to a particular disease [9]. Our immunohistochemistry results also showed strong positivity for ALK, which can be used to differentiate from NB. It is more likely that radiotherapy and medication used as adjuvant modalities during the preoperative and postoperative period contributed to the development of IMT of liver after adrenal NB surgery. Overall, the association between the two tumors awaits further study.
Data availability
The datasets for this article are not publicly available due to concerns regarding participant/patient anonymity. Requests to access the datasets should be directed to the corresponding author.
References
Castle JT, Levy BE, Rodeberg DA. Abdominal tumors: wilms, neuroblastoma, rhabdomyosarcoma, and hepatoblastoma [J]. Surg Clin North Am. 2022;102(5):715–37.
Baird DC, Meyers GJ, Hu JS. Testicular cancer: diagnosis and treatment [J]. Am Fam Phys. 2018;97(4):261–8.
Breslow NE, Lange JM, Friedman DL, et al. Secondary malignant neoplasms after Wilms tumor: an international collaborative study [J]. Int J Cancer. 2010;127(3):657–66.
Guan X, Wei R, Yang R, et al. Association of radiotherapy for rectal cancer and second gynecological malignant neoplasms [J]. JAMA Netw Open. 2021;4(1):e2031661.
Attarbaschi A, Carraro E, Ronceray L, et al. Second malignant neoplasms after treatment of non-Hodgkin’s lymphoma-a retrospective multinational study of 189 children and adolescents [J]. Leukemia. 2021;35(2):534–49.
Fabius AWM, van Hoefen WM, van Leeuwen FE, et al. Subsequent malignant neoplasms in retinoblastoma survivors [J]. Cancers. 2021;13(6):1200.
Keegan THM, Bleyer A, Rosenberg AS, et al. Second primary malignant neoplasms and survival in adolescent and young adult cancer survivors [J]. JAMA Oncol. 2017;3(11):1554–7.
Ottaviano M, Maddalena C, D’Armiento M, et al. Systemic treatment of malignant gastrointestinal neuroectodermal tumour after childhood neuroblastoma: chemotherapy in malignant gastrointestinal neuroectodermal tumour [J]. Anticancer Drugs. 2019;30(9):959–63.
Li YP, Han WW, He LJ, et al. Inflammatory myofibroblastic tumor after treatment of wilms tumor in a 6-year-old boy: a case report and literature review [J]. Urology. 2021;149:e25–8.
Webb TR, Slavish J, George RE, et al. Anaplastic lymphoma kinase: role in cancer pathogenesis and small-molecule inhibitor development for therapy [J]. Expert Rev Anticancer Ther. 2009;9(3):331–56.
Cairo SB, Urias AR, Murphy JT. Pediatric abdominal malignancies and intravascular extension: contemporary single-center experience [J]. J Surg Res. 2022;280:396–403.
Uttinger KL, Riedmeier M, Reibetanz J, et al. Adrenalectomies in children and adolescents in Germany—a diagnose related groups based analysis from 2009–2017 [J]. Front Endocrinol. 2022;13:914449.
Martin A, Schneiderman J, Helenowski IB, et al. Secondary malignant neoplasms after high-dose chemotherapy and autologous stem cell rescue for high-risk neuroblastoma [J]. Pediatr Blood Cancer. 2014;61(8):1350–6.
Applebaum MA, Vaksman Z, Lee SM, et al. Neuroblastoma survivors are at increased risk for second malignancies: a report from the international neuroblastoma risk group project [J]. Eur J Cancer. 2017;72:177–85.
Filips A, Maurer MH, Montani M, et al. Inflammatory myofibroblastic tumor of the liver: a case report and review of literature [J]. World J Hepatol. 2020;12(4):170–83.
Applebaum MA, Henderson TO, Lee SM, et al. Second malignancies in patients with neuroblastoma: the effects of risk-based therapy [J]. Pediatr Blood Cancer. 2015;62(1):128–33.
Westerveld ASR, van Dalen EC, Asogwa OA, et al. Neuroblastoma survivors at risk for develo** subsequent neoplasms: a systematic review [J]. Cancer Treat Rev. 2022;104:102355.
Zhen H, Guan H, Ma J, et al. Risk of develo** second malignant neoplasms in patients with neuroblastoma: a population study of the US SEER database [J]. Radiat Oncol. 2021;16(1):228.
Lee JS, Dubois SG, Coccia PF, et al. Increased risk of second malignant neoplasms in adolescents and young adults with cancer [J]. Cancer. 2016;122(1):116–23.
Yalcin B, Kremer LC, van Dalen EC. High-dose chemotherapy and autologous haematopoietic stem cell rescue for children with high-risk neuroblastoma [J]. Cochrane Database Syst Rev. 2010;12(5):CD006301.
Hawkins M, Bhatia S, Henderson TO, et al. Subsequent primary neoplasms: risks, risk factors, surveillance, and future research [J]. Pediatr Clin North Am. 2020;67(6):1135–54.
Park KJ, Kang SH, Lee HG, et al. Olfactory neuroblastoma following treatment for pituitary adenoma [J]. J Neurooncol. 2008;90(2):237–41.
Acknowledgements
We thank the patient and her parents for their participation in this research.
Funding
The study was based on the Pediatric Solid Tumor Cohort of Nan**g Children’s Hospital.
Author information
Authors and Affiliations
Contributions
Qiyang Shen and **ngyu Liu conducted the research. Qiyang Shen, Tao Li and Jianfeng Zhou are responsible for tumor resection and pathology specimen collection. **ngyu Liu and Lijie Zhang analyzed data. Qiyang Shen and **ngyu Liu drafted the manuscript. Tao Li and Jianfeng Zhou reviewed the draft. All authors contribute to the article and approve the submitted version.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study involving human participants was reviewed and approved by the Ethics Committee. The legal guardians/next of kin of the participants provided written informed consent for participation in this study. Written informed consent for the publication of any potentially identifiable images or data contained herein was obtained from the legal guardian/next of kin of the minor.
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shen, Q., Liu, X., Zhang, L. et al. Inflammatory myofibroblastic tumor of the liver after adrenal neuroblastoma surgery: a case report. Discov Onc 15, 174 (2024). https://doi.org/10.1007/s12672-024-01039-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12672-024-01039-4