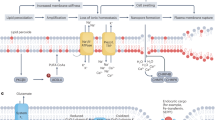
Abstract
Ferroptosis is a regulated form of necrotic cell death reliant on iron-catalyzed lipid peroxidation. Although the precise involvement of mitochondria in ferroptosis remains incompletely elucidated, recent research indicates that mitochondrial oxidative events wield a pivotal influence in this mechanism. This article centers on the most recent discoveries, spotlighting the significance of mitochondrial lipid peroxidation in the occurrence of ferroptosis. Modern investigative tools, such as mitochondria-specific dyes responsive to lipid peroxidation and antioxidants targeting mitochondria, have been employed to delve into this phenomenon. The authors’ recent empirical evidence demonstrates that mitochondrial lipid peroxidation, quantified using the innovative fluorescent ratiometric probe MitoCLox, takes place prior to the onset of ferroptotic cell death. The mitochondria-targeted antioxidant SkQ1 hinders mitochondrial lipid peroxidation and thwarts ferroptosis, all while leaving unaffected the buildup of reactive oxygen species within the cytoplasm, an antecedent to mitochondrial lipid peroxidation. Similarly, the redox agent methylene blue, impeding the genesis of reactive oxygen species in complex I of the electron transport chain, also imparts a comparable protective effect. These findings collectively imply that reactive oxygen species originating from complex I might hold particular significance in fomenting mitochondrial lipid peroxidation, a pivotal trigger of ferroptosis.



Similar content being viewed by others
References
Antonenko YN, Avetisyan AV, Bakeeva LE et al (2008) Mitochondria-targeted plastoquinone derivatives as tools to interrupt execution of the aging program. 1. Cationic plastoquinone derivatives: synthesis and in vitro studies. Biochemistry (Mosc) 73:1273–1287. https://doi.org/10.1134/s0006297908120018
Arai M, Imai H, Koumura T et al (1999) Mitochondrial phospholipid hydroperoxide glutathione peroxidase plays a major role in preventing oxidative injury to cells. J Biol Chem 274:4924–4933. https://doi.org/10.1074/jbc.274.8.4924
Atamna H, Nguyen A, Schultz C et al (2008) Methylene blue delays cellular senescence and enhances key mitochondrial biochemical pathways. FASEB J 22:703–712. https://doi.org/10.1096/fj.07-9610com
Bailey J, Shaw A, Fischer R et al (2017) A novel role for endothelial tetrahydrobiopterin in mitochondrial redox balance. Free Radic Biol Med 104:214–225. https://doi.org/10.1016/j.freeradbiomed.2017.01.012
Barayeu U, Schilling D, Eid M et al (2023) Hydropersulfides inhibit lipid peroxidation and ferroptosis by scavenging radicals. Nat Chem Biol 19:28–37. https://doi.org/10.1038/s41589-022-01145-w
Basit F, van Oppen LM, Schockel L et al (2017) Mitochondrial complex I inhibition triggers a mitophagy-dependent ROS increase leading to necroptosis and ferroptosis in melanoma cells. Cell Death Dis 8:e2716. https://doi.org/10.1038/cddis.2017.133
Belikova NA, Vladimirov YA, Osipov AN et al (2006) Peroxidase activity and structural transitions of cytochrome c bound to cardiolipin-containing membranes. Biochemistry 45:4998–5009. https://doi.org/10.1021/bi0525573
Beretta S, Wood JP, Derham B et al (2006) Partial mitochondrial complex I inhibition induces oxidative damage and perturbs glutamate transport in primary retinal cultures. Relevance to Leber Hereditary Optic Neuropathy (LHON). Neurobiol Dis 24:308–317. https://doi.org/10.1016/j.nbd.2006.07.016
Bersuker K, Hendricks JM, Li Z et al (2019) The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature 575:688–692. https://doi.org/10.1038/s41586-019-1705-2
Bilska-Wilkosz A, Iciek M, Gorny M (2022) Chemistry and biochemistry aspects of the 4-hydroxy-2,3-trans-nonenal. Biomolecules 12:145. https://doi.org/10.3390/biom12010145
Boveris A, Cadenas E, Stoppani AO (1976) Role of ubiquinone in the mitochondrial generation of hydrogen peroxide. Biochem J 156:435–444. https://doi.org/10.1042/bj1560435
Chernyavskij DA, Pletjushkina OY, Kashtanova AV et al (2023) Mitochondrial oxidative stress and mitophagy activation contribute to TNF-dependent impairment of myogenesis. Antioxidants (Basel) 12:602. https://doi.org/10.3390/antiox12030602
Deshwal S, Onishi M, Tatsuta T et al (2023) Mitochondria regulate intracellular coenzyme Q transport and ferroptotic resistance via STARD7. Nat Cell Biol 25:246–257. https://doi.org/10.1038/s41556-022-01071-y
Dixon SJ, Lemberg KM, Lamprecht MR et al (2012) Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149:1060–1072. https://doi.org/10.1016/j.cell.2012.03.042
Doll S, Freitas FP, Shah R et al (2019) FSP1 is a glutathione-independent ferroptosis suppressor. Nature 575:693–698. https://doi.org/10.1038/s41586-019-1707-0
Dolma S, Lessnick SL, Hahn WC, Stockwell BR (2003) Identification of genotype-selective antitumor agents using synthetic lethal chemical screening in engineered human tumor cells. Cancer Cell 3:285–296. https://doi.org/10.1016/s1535-6108(03)00050-3
Drummen GP, van Liebergen LC, Op den Kamp JA, Post JA (2002) C11-BODIPY(581/591), an oxidation-sensitive fluorescent lipid peroxidation probe: (micro)spectroscopic characterization and validation of methodology. Free Radic Biol Med 33:473–490. https://doi.org/10.1016/s0891-5849(02)00848-1
Elguindy MM, Nakamaru-Ogiso E (2015) Apoptosis-inducing factor (AIF) and its family member protein, AMID, are rotenone-sensitive NADH: ubiquinone oxidoreductases (NDH-2). J Biol Chem 290:20815–20826. https://doi.org/10.1074/jbc.M115.641498
Ernster L, Forsmark P, Nordenbrand K (1992) The mode of action of lipid-soluble antioxidants in biological membranes: relationship between the effects of ubiquinol and vitamin E as inhibitors of lipid peroxidation in submitochondrial particles. BioFactors 3:241–248. https://doi.org/10.3177/jnsv.38.special_548
Esterbauer H (1993) Cytotoxicity and genotoxicity of lipid-oxidation products. Am J Clin Nutr 57:779S-785S. https://doi.org/10.1093/ajcn/57.5.779S. (discussion 785S-786S)
Fan BY, Pang YL, Li WX et al (2021) Liproxstatin-1 is an effective inhibitor of oligodendrocyte ferroptosis induced by inhibition of glutathione peroxidase 4. Neural Regen Res 16:561–566. https://doi.org/10.4103/1673-5374.293157
Fang X, Wang H, Han D et al (2019) Ferroptosis as a target for protection against cardiomyopathy. Proc Natl Acad Sci U S A 116:2672–2680. https://doi.org/10.1073/pnas.1821022116
Filipovska A, Kelso GF, Brown SE et al (2005) Synthesis and characterization of a triphenylphosphonium-conjugated peroxidase mimetic. Insights into the interaction of ebselen with mitochondria. J Biol Chem 280:24113–24126. https://doi.org/10.1074/jbc.M501148200
Freigang S (2016) The regulation of inflammation by oxidized phospholipids. Eur J Immunol 46:1818–1825. https://doi.org/10.1002/eji.201545676
Friedmann Angeli JP, Schneider M, Proneth B et al (2014) Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol 16:1180–1191. https://doi.org/10.1038/ncb3064
Fukushima T, Yamada K, Hojo N et al (1994) Mechanism of cytotoxicity of paraquat. III. The effects of acute paraquat exposure on the electron transport system in rat mitochondria. Exp Toxicol Pathol 46:437–441. https://doi.org/10.1016/S0940-2993(11)80056-4
Gao M, Monian P, Quadri N et al (2015) Glutaminolysis and transferrin regulate ferroptosis. Mol Cell 59:298–308. https://doi.org/10.1016/j.molcel.2015.06.011
Gao M, Yi J, Zhu J et al (2019) Role of mitochondria in ferroptosis. Mol Cell 73:354-363 e353. https://doi.org/10.1016/j.molcel.2018.10.042
Gaschler MM, Hu F, Feng H et al (2018) Determination of the subcellular localization and mechanism of action of ferrostatins in suppressing ferroptosis. ACS Chem Biol 13:1013–1020. https://doi.org/10.1021/acschembio.8b00199
Glinn M, Ernster L, Lee CP (1991) Initiation of lipid peroxidation in submitochondrial particles: effect of respiratory inhibitors. Arch Biochem Biophys 290:57–65. https://doi.org/10.1016/0003-9861(91)90591-6
Gureev AP, Shaforostova EA, Laver DA et al (2019) Methylene blue elicits non-genotoxic H(2)O(2) production and protects brain mitochondria from rotenone toxicity. J Appl Biomed 17:107–114. https://doi.org/10.32725/jab.2019.008
Gureev AP, Sadovnikova IS, Popov VN (2022) Molecular mechanisms of the neuroprotective effect of methylene blue. Biochemistry (Mosc) 87:940–956. https://doi.org/10.1134/S0006297922090073
Hess ML, Manson NH (1984) Molecular oxygen: friend and foe. The role of the oxygen free radical system in the calcium paradox, the oxygen paradox and ischemia/reperfusion injury. J Mol Cell Cardiol 16:969–985. https://doi.org/10.1016/s0022-2828(84)80011-5
Homma T, Kobayashi S, Sato H, Fujii J (2021) Superoxide produced by mitochondrial complex III plays a pivotal role in the execution of ferroptosis induced by cysteine starvation. Arch Biochem Biophys 700:108775. https://doi.org/10.1016/j.abb.2021.108775
Hou W, **e Y, Song X et al (2016) Autophagy promotes ferroptosis by degradation of ferritin. Autophagy 12:1425–1428. https://doi.org/10.1080/15548627.2016.1187366
Hruza LL, Pentland AP (1993) Mechanisms of UV-induced inflammation. J Invest Dermatol 100:35S-41S. https://doi.org/10.1111/1523-1747.ep12355240
Huang Z, Jiang J, Tyurin VA et al (2008) Cardiolipin deficiency leads to decreased cardiolipin peroxidation and increased resistance of cells to apoptosis. Free Radic Biol Med 44:1935–1944. https://doi.org/10.1016/j.freeradbiomed.2008.02.016
Ide T, Tsutsui H, Kinugawa S et al (1999) Mitochondrial electron transport complex I is a potential source of oxygen free radicals in the failing myocardium. Circ Res 85:357–363. https://doi.org/10.1161/01.res.85.4.357
Jang S, Chapa-Dubocq XR, Tyurina YY et al (2021) Elucidating the contribution of mitochondrial glutathione to ferroptosis in cardiomyocytes. Redox Biol 45:102021. https://doi.org/10.1016/j.redox.2021.102021
Jenner P, Olanow CW (1998) Understanding cell death in Parkinson’s disease. Ann Neurol 44:S72-84. https://doi.org/10.1002/ana.410440712
Jiang X, Stockwell BR, Conrad M (2021) Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol 22:266–282. https://doi.org/10.1038/s41580-020-00324-8
Kagan VE, Bayir HA, Belikova NA et al (2009) Cytochrome c/cardiolipin relations in mitochondria: a kiss of death. Free Radic Biol Med 46:1439–1453. https://doi.org/10.1016/j.freeradbiomed.2009.03.004
Kagan VE, Mao G, Qu F et al (2017) Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat Chem Biol 13:81–90. https://doi.org/10.1038/nchembio.2238
Kagan VE, Tyurina YY, Vlasova II et al (2020) Redox epiphospholipidome in programmed cell death signaling: catalytic mechanisms and regulation. Front Endocrinol (Lausanne) 11:628079. https://doi.org/10.3389/fendo.2020.628079
Kamalden TA, Ji D, Osborne NN (2012) Rotenone-induced death of RGC-5 cells is caspase independent, involves the JNK and p38 pathways and is attenuated by specific green tea flavonoids. Neurochem Res 37:1091–1101. https://doi.org/10.1007/s11064-012-0713-5
Kelner MJ, Alexander NM (1985) Methylene blue directly oxidizes glutathione without the intermediate formation of hydrogen peroxide. J Biol Chem 260:15168–15171. https://doi.org/10.1016/S0021-9258(18)95717-0
Kelso GF, Porteous CM, Coulter CV et al (2001) Selective targeting of a redox-active ubiquinone to mitochondria within cells: antioxidant and antiapoptotic properties. J Biol Chem 276:4588–4596. https://doi.org/10.1074/jbc.M009093200
Kraft VAN, Bezjian CT, Pfeiffer S et al (2020) GTP cyclohydrolase 1/tetrahydrobiopterin counteract ferroptosis through lipid remodeling. ACS Cent Sci 6:41–53. https://doi.org/10.1021/acscentsci.9b01063
Krainz T, Gaschler MM, Lim C et al (2016) A mitochondrial-targeted nitroxide is a potent inhibitor of ferroptosis. ACS Cent Sci 2:653–659. https://doi.org/10.1021/acscentsci.6b00199
Kuschner CE, Choi J, Yin T et al (2018) Comparing phospholipid profiles of mitochondria and whole tissue: higher PUFA content in mitochondria is driven by increased phosphatidylcholine unsaturation. J Chromatogr B Analyt Technol Biomed Life Sci 1093–1094:147–157. https://doi.org/10.1016/j.jchromb.2018.07.006
Lipper CH, Stofleth JT, Bai F et al (2019) Redox-dependent gating of VDAC by mitoNEET. Proc Natl Acad Sci U S A 116:19924–19929. https://doi.org/10.1073/pnas.1908271116
Liu J, Bandyopadhyay I, Zheng L et al (2020) Antiferroptotic activity of phenothiazine analogues: a novel therapeutic strategy for oxidative stress related disease. ACS Med Chem Lett 11:2165–2173. https://doi.org/10.1021/acsmedchemlett.0c00293
Luo X, Pitkanen S, Kassovska-Bratinova S et al (1997) Excessive formation of hydroxyl radicals and aldehydic lipid peroxidation products in cultured skin fibroblasts from patients with complex I deficiency. J Clin Invest 99:2877–2882. https://doi.org/10.1172/JCI119481
Lyamzaev KG, Pustovidko AV, Simonyan RA et al (2011) Novel mitochondria-targeted antioxidants: plastoquinone conjugated with cationic plant alkaloids berberine and palmatine. Pharm Res 28:2883–2895. https://doi.org/10.1007/s11095-011-0504-8
Lyamzaev KG, Sumbatyan NV, Nesterenko AM et al (2019) MitoCLox: a novel mitochondria-targeted fluorescent probe for tracing lipid peroxidation. Oxid Med Cell Longev 2019:9710208. https://doi.org/10.1155/2019/9710208
Lyamzaev KG, Panteleeva AA, Karpukhina AA et al (2020) Novel fluorescent mitochondria-targeted probe MitoCLox reports lipid peroxidation in response to oxidative stress in vivo. Oxid Med Cell Longev 2020:3631272. https://doi.org/10.1155/2020/3631272
Lyamzaev KG, Panteleeva AA, Simonyan RA et al (2023) Mitochondrial lipid peroxidation is responsible for ferroptosis. Cells 12(4):611. https://doi.org/10.3390/cells12040611
Mao C, Liu X, Zhang Y et al (2021) DHODH-mediated ferroptosis defence is a targetable vulnerability in cancer. Nature 593:586–590. https://doi.org/10.1038/s41586-021-03539-7
Mbemba F, Houbion A, Raes M, Remacle J (1985) Subcellular localization and modification with ageing of glutathione, glutathione peroxidase and glutathione reductase activities in human fibroblasts. Biochim Biophys Acta 838:211–220. https://doi.org/10.1016/0304-4165(85)90081-9
Muller FL, Liu Y, Van Remmen H (2004) Complex III releases superoxide to both sides of the inner mitochondrial membrane. J Biol Chem 279:49064–49073. https://doi.org/10.1074/jbc.M407715200
Nesterenko AM, Kholina EG, Lyamzaev KG et al (2019) Molecular dynamics modeling of the interaction of cationic fluorescent lipid peroxidation-sensitive probes with the mitochondrial membrane. Dokl Biochem Biophys 486:220–223. https://doi.org/10.1134/S1607672919030153
Oh SJ, Ikeda M, Ide T et al (2022) Mitochondrial event as an ultimate step in ferroptosis. Cell Death Discov 8:414. https://doi.org/10.1038/s41420-022-01199-8
Orr AL, Vargas L, Turk CN et al (2015) Suppressors of superoxide production from mitochondrial complex III. Nat Chem Biol 11:834–836. https://doi.org/10.1038/nchembio.1910
Panov AV, Dikalov SI (2020) Cardiolipin, perhydroxyl radicals, and lipid peroxidation in mitochondrial dysfunctions and aging. Oxid Med Cell Longev 2020:1323028. https://doi.org/10.1155/2020/1323028
Pap EH, Drummen GP, Winter VJ et al (1999) Ratio-fluorescence microscopy of lipid oxidation in living cells using C11-BODIPY(581/591). FEBS Lett 453:278–282. https://doi.org/10.1016/s0014-5793(99)00696-1
Paul BT, Manz DH, Torti FM, Torti SV (2017) Mitochondria and iron: current questions. Expert Rev Hematol 10:65–79. https://doi.org/10.1080/17474086.2016.1268047
Prime TA, Forkink M, Logan A et al (2012) A ratiometric fluorescent probe for assessing mitochondrial phospholipid peroxidation within living cells. Free Radic Biol Med 53:544–553. https://doi.org/10.1016/j.freeradbiomed.2012.05.033
Ramsay RR, Singer TP (1992) Relation of superoxide generation and lipid peroxidation to the inhibition of NADH-Q oxidoreductase by rotenone, piericidin A, and MPP+. Biochem Biophys Res Commun 189:47–52. https://doi.org/10.1016/0006-291x(92)91523-s
Reis A (2017) Oxidative Phospholipidomics in health and disease: achievements, challenges and hopes. Free Radic Biol Med 111:25–37. https://doi.org/10.1016/j.freeradbiomed.2017.01.014
Sakharov DV, Elstak ED, Chernyak B, Wirtz KW (2005) Prolonged lipid oxidation after photodynamic treatment. Study with oxidation-sensitive probe C11-BODIPY581/591. FEBS Lett 579:1255–1260. https://doi.org/10.1016/j.febslet.2005.01.024
Seo BB, Marella M, Yagi T, Matsuno-Yagi A (2006) The single subunit NADH dehydrogenase reduces generation of reactive oxygen species from complex I. FEBS Lett 580:6105–6108. https://doi.org/10.1016/j.febslet.2006.10.008
Shioji K, Oyama Y, Okuma K, Nakagawa H (2010) Synthesis and properties of fluorescence probe for detection of peroxides in mitochondria. Bioorg Med Chem Lett 20:3911–3915. https://doi.org/10.1016/j.bmcl.2010.05.017
Skulachev VP, Antonenko YN, Cherepanov DA et al (2010) Prevention of cardiolipin oxidation and fatty acid cycling as two antioxidant mechanisms of cationic derivatives of plastoquinone (SkQs). Biochim Biophys Acta 1797:878–889. https://doi.org/10.1016/j.bbabio.2010.03.015
Soula M, Weber RA, Zilka O et al (2020) Metabolic determinants of cancer cell sensitivity to canonical ferroptosis inducers. Nat Chem Biol 16:1351–1360. https://doi.org/10.1038/s41589-020-0613-y
Takayanagi R, Takeshige K, Minakami S (1980) NADH- and NADPH-dependent lipid peroxidation in bovine heart submitochondrial particles. Dependence on the rate of electron flow in the respiratory chain and an antioxidant role of ubiquinol. Biochem J 192:853–860. https://doi.org/10.1042/bj1920853
Takebe G, Yarimizu J, Saito Y et al (2002) A comparative study on the hydroperoxide and thiol specificity of the glutathione peroxidase family and selenoprotein P. J Biol Chem 277:41254–41258. https://doi.org/10.1074/jbc.M202773200
Tan S, Sagara Y, Liu Y et al (1998) The regulation of reactive oxygen species production during programmed cell death. J Cell Biol 141:1423–1432. https://doi.org/10.1083/jcb.141.6.1423
Tan S, Schubert D, Maher P (2001) Oxytosis: a novel form of programmed cell death. Curr Top Med Chem 1:497–506. https://doi.org/10.2174/1568026013394741
Tretter L, Horvath G, Holgyesi A et al (2014) Enhanced hydrogen peroxide generation accompanies the beneficial bioenergetic effects of methylene blue in isolated brain mitochondria. Free Radic Biol Med 77:317–330. https://doi.org/10.1016/j.freeradbiomed.2014.09.024
Tyurina YY, St Croix CM, Watkins SC et al (2019) Redox (phospho)lipidomics of signaling in inflammation and programmed cell death. J Leukoc Biol 106:57–81. https://doi.org/10.1002/JLB.3MIR0119-004RR
Vile GF, Winterbourn CC (1987) Iron binding to microsomes and liposomes in relation to lipid peroxidation. FEBS Lett 215:151–154. https://doi.org/10.1016/0014-5793(87)80131-x
Vinogradov AD, Grivennikova VG (2016) Oxidation of NADH and ROS production by respiratory complex I. Biochim Biophys Acta 1857:863–871. https://doi.org/10.1016/j.bbabio.2015.11.004
Vladimirov YA, Olenev VI, Suslova TB, Cheremisina ZP (1980) Lipid peroxidation in mitochondrial membrane. Adv Lipid Res 17:173–249. https://doi.org/10.1016/b978-0-12-024917-6.50011-2
Wipf P, **ao J, Jiang J et al (2005) Mitochondrial targeting of selective electron scavengers: synthesis and biological analysis of hemigramicidin-TEMPO conjugates. J Am Chem Soc 127:12460–12461. https://doi.org/10.1021/ja053679l
Wu S, Mao C, Kondiparthi L et al (2022a) A ferroptosis defense mechanism mediated by glycerol-3-phosphate dehydrogenase 2 in mitochondria. Proc Natl Acad Sci U S A 119:e2121987119. https://doi.org/10.1073/pnas.2121987119
Wu Z, Khodade VS, Chauvin JR et al (2022b) Hydropersulfides inhibit lipid peroxidation and protect cells from ferroptosis. J Am Chem Soc 144:15825–15837. https://doi.org/10.1021/jacs.2c06804
Xun Z, Rivera-Sanchez S, Ayala-Pena S et al (2012) Targeting of XJB-5-131 to mitochondria suppresses oxidative DNA damage and motor decline in a mouse model of Huntington’s disease. Cell Rep 2:1137–1142. https://doi.org/10.1016/j.celrep.2012.10.001
Yamada K, Fukushima T (1993) Mechanism of cytotoxicity of paraquat. II. Organ specificity of paraquat-stimulated lipid peroxidation in the inner membrane of mitochondria. Exp Toxicol Pathol 45:375–380. https://doi.org/10.1016/S0940-2993(11)80433-1
Yamanaka K, Saito Y, Sakiyama J et al (2012) A novel fluorescent probe with high sensitivity and selective detection of lipid hydroperoxides in cells. RSC Adv 2:7894–7900. https://doi.org/10.1039/C2RA20816D
Yang WS, Stockwell BR (2008) Synthetic lethal screening identifies compounds activating iron-dependent, nonapoptotic cell death in oncogenic-RAS-harboring cancer cells. Chem Biol 15:234–245. https://doi.org/10.1016/j.chembiol.2008.02.010
Yang WS, SriRamaratnam R, Welsch ME et al (2014) Regulation of ferroptotic cancer cell death by GPX4. Cell 156:317–331. https://doi.org/10.1016/j.cell.2013.12.010
Yang WS, Kim KJ, Gaschler MM et al (2016) Peroxidation of polyunsaturated fatty acids by lipoxygenases drives ferroptosis. Proc Natl Acad Sci U S A 113:E4966-4975. https://doi.org/10.1073/pnas.1603244113
Yuan H, Li X, Zhang X et al (2016) CISD1 inhibits ferroptosis by protection against mitochondrial lipid peroxidation. Biochem Biophys Res Commun 478:838–844. https://doi.org/10.1016/j.bbrc.2016.08.034
Zager RA (1996) Mitochondrial free radical production induces lipid peroxidation during myohemoglobinuria. Kidney Int 49:741–751. https://doi.org/10.1038/ki.1996.103
Zeitler L, Fiore A, Meyer C et al (2021) Anti-ferroptotic mechanism of IL4i1-mediated amino acid metabolism. Elife 10:e64806. https://doi.org/10.7554/eLife.64806
Zhang JG, Tirmenstein MA, Nicholls-Grzemski FA, Fariss MW (2001) Mitochondrial electron transport inhibitors cause lipid peroxidation-dependent and -independent cell death: protective role of antioxidants. Arch Biochem Biophys 393:87–96. https://doi.org/10.1006/abbi.2001.2486
Zhang X, Rojas JC, Gonzalez-Lima F (2006) Methylene blue prevents neurodegeneration caused by rotenone in the retina. Neurotox Res 9:47–57. https://doi.org/10.1007/BF03033307
Zilka O, Shah R, Li B et al (2017) On the mechanism of cytoprotection by ferrostatin-1 and liproxstatin-1 and the role of lipid peroxidation in ferroptotic Cell Death. ACS Cent Sci 3:232–243. https://doi.org/10.1021/acscentsci.7b00028
Zinovkin RA, Zamyatnin AA (2019) Mitochondria-targeted drugs. Curr. Mol Pharmacol 12:202–214. https://doi.org/10.2174/1874467212666181127151059
Funding
The work was supported by the grant of the Russian Science Foundation No. 23–14-00061.
Author information
Authors and Affiliations
Contributions
BVC conceptualized and outlined the review. Material preparation, data collection, and analysis were performed by BVC and KGL. BVC and KGL wrote the first draft of the manuscript and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lyamzaev, K.G., Panteleeva, A.A., Simonyan, R.A. et al. The critical role of mitochondrial lipid peroxidation in ferroptosis: insights from recent studies. Biophys Rev 15, 875–885 (2023). https://doi.org/10.1007/s12551-023-01126-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-023-01126-w