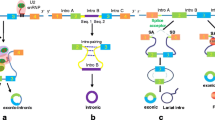
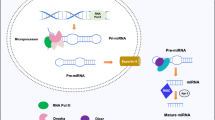
Abstract
Regulation of gene expression by small non-coding RNAs, such as miRNAs and siRNAs, is an inherent part of complex biological processes that allow function and survival of eukaryotic cells. The type III ribonuclease DICER is widely recognized as a key step in the production of miRNAs and siRNAs, although it also has non-canonical functions such as DNA repair and induction of apoptosis. DICER is at the cornerstone of most biological processes; hence, its mRNA and protein levels are subject to multiple layers of regulation. Accordingly, DICER derangement is related to disease and leads to accelerated aging. Interventions that boost DICER function hold great potential as strategies to promote health and longevity. In this review, we will summarize the structural features of DICER, describe its canonical and non-canonical roles, and discuss the most common regulatory mechanisms having an impact on DICER abundance and function. We will also touch upon the current literature demonstrating that DICER deregulation can lead to diseases, thus highlighting the importance of DICER in warranting the beneficial effects of health-promoting interventions.


Similar content being viewed by others
References
Aryal NK, Pant V, Wasylishen AR et al (2019) Constitutive dicer1 phosphorylation accelerates metabolism and aging in vivo. Proc Natl Acad Sci U S A 116:960–969. https://doi.org/10.1073/pnas.1814377116
Bartel DP (2018) Metazoan MicroRNAs. Cell 173:20–51. https://doi.org/10.1016/j.cell.2018.03.006
Bennasser Y, Chable-Bessia C, Triboulet R et al (2011) Competition for XPO5 binding between dicer mRNA, pre-miRNA and viral RNA regulates human dicer levels. Nat Struct Mol Biol 18:323–327. https://doi.org/10.1038/nsmb.1987
Bernstein E, Caudy AA, Hammond SM, Hannon GJ (2001) Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature. https://doi.org/10.1038/35053110
Bernstein E, Kim SY, Carmell MA et al (2003) Dicer is essential for mouse development. Nat Genet 35:215–217. https://doi.org/10.1038/ng1253
Beshore EL, McEwen TJ, Jud MC et al (2011) C. elegans dicer interacts with the P-granule component GLH-1 and both regulate germline RNPs. Dev Biol 350:370–381. https://doi.org/10.1016/j.ydbio.2010.12.005
Boominathan L (2010) The guardians of the genome (p53, TA-p73, and TA-p63) are regulators of tumor suppressor miRNAs network. Cancer Metastasis Rev 29:613–639. https://doi.org/10.1007/s10555-010-9257-9
Boon RA, Iekushi K, Lechner S et al (2013) MicroRNA-34a regulates cardiac ageing and function. Nature 495:107–110. https://doi.org/10.1038/nature11919
Burger K, Gullerova M (2015) Swiss army knives: non-canonical functions of nuclear Drosha and dicer. Nat Rev Mol Cell Biol 16:417–430. https://doi.org/10.1038/nrm3994
Burger K, Schlackow M, Potts M et al (2017) Nuclear phosphorylated dicer processes doublestranded RNA in response to DNA damage. J Cell Biol 216:2373–2389. https://doi.org/10.1083/jcb.201612131
Burley SK, Bhikadiya C, Bi C et al (2021) RCSB Protein Data Bank: powerful new tools for exploring 3D structures of biological macromolecules for basic and applied research and education in fundamental biology, biomedicine, biotechnology, bioengineering and energy sciences. Nucleic Acids Res 49:D437–D451. https://doi.org/10.1093/nar/gkaa1038
Byrd AK, Raney KD (2012) Superfamily 2 helicases. Front Biosci 17:2070–2088
Chakravarthy S, Sternberg SH, Kellenberger CA, Doudna JA (2010) Substrate-specific kinetics of dicer-catalyzed RNA processing. J Mol Biol 404:392–402. https://doi.org/10.1016/j.jmb.2010.09.030
Cheloufi S, Dos Santos CO, Chong MMW, Hannon GJ (2010) A dicer-independent miRNA biogenesis pathway that requires Ago catalysis. Nature 465:584–589. https://doi.org/10.1038/nature09092
Chen JF, Murchison EP, Tang R et al (2008) Targeted deletion of dicer in the heart leads to dilated cardiomyopathy and heart failure. Proc Natl Acad Sci U S A 105:2111–2116. https://doi.org/10.1073/pnas.0710228105
Chendrimada TP, Gregory RI, Kumaraswamy E et al (2005) TRBP recruits the dicer complex to Ago2 for microRNA processing and gene silencing. Nature 436:740–744. https://doi.org/10.1038/nature03868
Chitale S, Richly H (2017) DICER and ZRF1 contribute to chromatin decondensation during nucleotide excision repair. Nucleic Acids Res 45:5901–5912. https://doi.org/10.1093/nar/gkx261
Chitale S, Richly H (2018) DIC ER- and MMS ET-catalyzed H4K20me2 recruits the nucleotide excision repair factor XPA to DNA damage sites. J Cell Biol 217:527–540. https://doi.org/10.1083/jcb.201704028
Drake M, Furuta T, Suen KM et al (2014) A requirement for ERK-dependent dicer phosphorylation in coordinating oocyte-to-embryo transition in C.elegans. Dev Cell 31:614–628. https://doi.org/10.1016/j.devcel.2014.11.004
Ebert MS, Sharp PA (2012) Roles for MicroRNAs in conferring robustness to biological processes. Cell 149:515–524. https://doi.org/10.1016/j.cell.2012.04.005
Eichhorn SW, Guo H, McGeary SE et al (2014) MRNA Destabilization Is the dominant effect of mammalian microRNAs by the time substantial repression ensues. Mol Cell 56:104–115. https://doi.org/10.1016/j.molcel.2014.08.028
Emde A, Eitan C, Liou L-L et al (2015) Dysregulated mi RNA biogenesis downstream of cellular stress and ALS -causing mutations: a new mechanism for ALS. EMBO J 34:2633–2651. https://doi.org/10.15252/embj.201490493
Feinberg-Gorenshtein G, Guedj A, Shichrur K et al (2013) miR-192 directly binds and regulates Dicer1 expression in neuroblastoma. PLoS One 8:e78713. https://doi.org/10.1371/journal.pone.0078713
Flemr M, Malik R, Franke V et al (2013) A retrotransposon-driven dicer isoform directs endogenous small interfering RNA production in mouse oocytes. Cell 155:807–816. https://doi.org/10.1016/j.cell.2013.10.001
Forman JJ, Legesse-Miller A, Coller HA (2008) A search for conserved sequences in coding regions reveals that the let-7 microRNA targets dicer within its coding sequence. Proc Natl Acad Sci U S A 105:14879–14884. https://doi.org/10.1073/pnas.0803230105
Foulkes WD, Priest JR, Duchaine TF (2014) DICER1: Mutations, microRNAs and mechanisms. Nat Rev Cancer 14:662–672
Gibbings D, Mostowy S, Jay F et al (2012) Selective autophagy degrades DICER and AGO2 and regulates miRNA activity. Nat Cell Biol 14:1314–1321. https://doi.org/10.1038/ncb2611
Grelier G, Voirin N, Ay AS et al (2009) Prognostic value of dicer expression in human breast cancers and association with the mesenchymal phenotype. Br J Cancer 101:673–683. https://doi.org/10.1038/sj.bjc.6605193
Gross TJ, Powers LS, Boudreau RL et al (2014) A Microrna processing defect in smokers’ macrophages is linked to sumoylation of the endonuclease DICER. J Biol Chem 289:12823–12834. https://doi.org/10.1074/jbc.M114.565473
Gu S, ** L, Zhang Y et al (2012) The loop position of shRNAs and pre-miRNAs is critical for the accuracy of dicer processing in vivo. Cell 151:900–911. https://doi.org/10.1016/j.cell.2012.09.042
Guerra BA, Brandão BB, Pinto SS et al (2019) Dietary sulfur amino acid restriction upregulates DICER to confer beneficial effects. Mol Metab 29:124–135. https://doi.org/10.1016/j.molmet.2019.08.017
Ha M, Kim VN (2014) Regulation of microRNA biogenesis. Nat Rev Mol Cell Biol 15:509–524. https://doi.org/10.1038/nrm3838
Hamaya Y, Kuriyama S, Takai T, et al (2012) A distinct expression pattern of the long 3′-untranslated region dicer mRNA and its implications for posttranscriptional regulation in colorectal cancer. Clin Transl Gastroenterol 3. https://doi.org/10.1038/ctg.2012.12
Hill DA, Ivanovich J, Priest JR et al (2009) DICER1 mutations in familial pleuropulmonary blastoma. Science 325(80):965. https://doi.org/10.1126/science.1174334
Hinkal GW, Grelier G, Puisieux A, Moyret-Lalle C (2011) Complexity in the regulation of dicer expression: dicer variant proteins are differentially expressed in epithelial and mesenchymal breast cancer cells and decreased during EMT. Br J Cancer 104:387–388. https://doi.org/10.1038/sj.bjc.6606022
Hutvágner G, McLachlan J, Pasquinelli AE et al (2001) A cellular function for the RNA-interference enzyme dicer in the maturation of the let-7 small temporal RNA. Science 293(80):834–838. https://doi.org/10.1126/science.1062961
Irvin-Wilson CV, Chaudhuri G (2005) Alternative initiation and splicing in dicer gene expression in human breast cells. Breast Cancer Res 7:1–7. https://doi.org/10.1186/bcr1043
Jafarnejad SM, Ardekani GS, Ghaffari M et al (2013) Sox4-mediated dicer expression is critical for suppression of melanoma cell invasion. Oncogene 32:2131–2139. https://doi.org/10.1038/onc.2012.239
Kurzynska-Kokorniak A, Koralewska N, Pokornowska M et al (2015) The many faces of dicer: the complexity of the mechanisms regulating dicer gene expression and enzyme activities. Nucleic Acids Res 43:4365–4380. https://doi.org/10.1093/nar/gkv328
Kurzynska-Kokorniak A, Pokornowska M, Koralewska N et al (2016) Revealing a new activity of the human dicer DUF283 domain in vitro. Sci Rep 6:1–13. https://doi.org/10.1038/srep23989
Lee HY, Zhou K, Smith AM et al (2013) Differential roles of human dicer-binding proteins TRBP and PACT in small RNA processing. Nucleic Acids Res 41:6568–6576. https://doi.org/10.1093/nar/gkt361
Lee Y, Hur I, Park SY et al (2006) The role of PACT in the RNA silencing pathway. EMBO J 25:522–532. https://doi.org/10.1038/sj.emboj.7600942
Levy C, Khaled M, Robinson KC et al (2010) Lineage-specific transcriptional regulation of DICER by MITF in melanocytes. Cell 141:994–1005. https://doi.org/10.1016/J.CELL.2010.05.004
Li S, Xu Z, Sheng J (2018) tRNA-derived small RNA: a novel regulatory small non-coding RNA. Genes (basel) 9:246. https://doi.org/10.3390/genes9050246
Liu Q, Rand TA, Kalidas S et al (2003) R2D2, a bridge between the initiation and effector steps of the Drosophila RNAi pathway. Science 301(80):1921–1925. https://doi.org/10.1126/science.1088710
Liu Z, Wang J, Cheng H et al (2018) Cryo-EM structure of human dicer and its complexes with a pre-miRNA substrate. Cell 173:1191-1203.e12. https://doi.org/10.1016/j.cell.2018.03.080
Luo QJ, Zhang J, Li P et al (2021) RNA structure probing reveals the structural basis of dicer binding and cleavage. Nat Commun 12:1–12. https://doi.org/10.1038/s41467-021-23607-w
Ma E, MacRae IJ, Kirsch JF, Doudna JA (2008) Autoinhibition of human dicer by its internal helicase domain. J Mol Biol 380:237–243. https://doi.org/10.1016/j.jmb.2008.05.005
Ma E, Zhou K, Kidwell MA, Doudna JA (2012) Coordinated activities of human dicer domains in regulatory RNA processing. J Mol Biol 422:466–476. https://doi.org/10.1016/j.jmb.2012.06.009
MacRae IJ, Zhou K, Doudna JA (2007) Structural determinants of RNA recognition and cleavage by dicer. Nat Struct Mol Biol 14:934–940. https://doi.org/10.1038/nsmb1293
MacRae IJ, Zhou K, Li F et al (2006) Structural basis for double-stranded RNA processing by dicer. Science 311(80):195–198. https://doi.org/10.1126/science.1121638
Martello G, Rosato A, Ferrari F et al (2010) A microRNA targeting dicer for metastasis control. Cell 141:1195–1207. https://doi.org/10.1016/j.cell.2010.05.017
Masliah G, Barraud P, Allain FHTH-T (2013) RNA recognition by double-stranded RNA binding domains: A matter of shape and sequence. Cell Mol Life Sci 70:1875–1895. https://doi.org/10.1007/s00018-012-1119-x
Matskevich AA, Moelung K (2008) Stimuli-dependent cleavage of dicer during apoptosis. Biochem J 412:527–534. https://doi.org/10.1042/BJ20071461
Mori MA, Raghavan P, Thomou T et al (2012) Role of microRNA processing in adipose tissue in stress defense and longevity. Cell Metab 16:336–347. https://doi.org/10.1016/j.cmet.2012.07.017
Mori MA, Thomou T, Boucher J et al (2014) Altered miRNA processing disrupts brown/white adipocyte determination and associates with lipodystrophy. J Clin Invest 124:3339–3351. https://doi.org/10.1172/JCI73468
Nakagawa A, Shi Y, Kage-Nakadai E et al (2010) Caspase-dependent conversion of dicer ribonuclease into a death-promoting deoxyribonuclease. Science 328(80):327–334. https://doi.org/10.1126/science.1182374
Nowotny M, Yang W (2006) Stepwise analyses of metal ions in RNase H catalysis from substrate destabilization to product release. EMBO J 25:1924–1933. https://doi.org/10.1038/sj.emboj.7601076
Oliverio M, Schmidt E, Mauer J et al (2016) Dicer1–miR-328–Bace1 signalling controls brown adipose tissue differentiation and function. Nat Cell Biol 18(3):328–336. https://doi.org/10.1038/ncb3316
Ota H, Sakurai M, Gupta R et al (2013) ADAR1 forms a complex with dicer to promote MicroRNA processing and RNA-induced gene silencing. Cell 153:575–589. https://doi.org/10.1016/j.cell.2013.03.024
Park JE, Heo I, Tian Y et al (2011) Dicer recognizes the 5′ end of RNA for efficient and accurate processing. Nature 475:201–205. https://doi.org/10.1038/nature10198
Paroo Z, Ye X, Chen S, Liu Q (2009) Phosphorylation of the human microRNA-generating complex mediates MAPK/Erk signaling. Cell 139:112–122. https://doi.org/10.1016/j.cell.2009.06.044
Paturi S, Deshmukh MV (2021) A glimpse of “dicer biology” through the structural and functional perspective. Front Mol Biosci 8:304. https://doi.org/10.3389/fmolb.2021.643657
Pinto S, Sato VN, De-Souza EA et al (2018) Enoxacin extends lifespan of C. elegans by inhibiting miR-34-5p and promoting mitohormesis. Redox Biol 18:84–92. https://doi.org/10.1016/j.redox.2018.06.006
Poirier EZ, Buck MD, Chakravarty P et al (2021) An isoform of dicer protects mammalian stem cells against multiple RNA viruses. Science 373(80):231–236. https://doi.org/10.1126/science.abg2264
Pong SK, Gullerova M (2018) Noncanonical functions of microRNA pathway enzymes – Drosha, DGCR8. Dicer and Ago Proteins FEBS Lett 592:2973–2986. https://doi.org/10.1002/1873-3468.13196
Potenza N, Papa U, Scaruffi P et al (2010) A novel splice variant of the human dicer gene is expressed in neuroblastoma cells. FEBS Lett 584:3452–3457. https://doi.org/10.1016/j.febslet.2010.06.045
Reis FCG, Branquinho JLO, Brandão BB et al (2016) Fat-specific dicer deficiency accelerates aging and mitigates several effects of dietary restriction in mice. Aging (Albany NY) 8:1201–1222. https://doi.org/10.18632/aging.100970
Rigbolt KTG, Prokhorova TA, Akimov V et al (2011) System-wide temporal characterization of the proteome and phosphoproteome of human embryonic stem cell differentiation. Sci Signal 4:rs3–rs3. https://doi.org/10.1126/scisignal.2001570
Rocha AL, de Lima TI, de Souza GP et al (2020) Enoxacin induces oxidative metabolism and mitigates obesity by regulating adipose tissue miRNA expression. Sci Adv 6:eabc6250. https://doi.org/10.1126/SCIADV.ABC6250
Ruby JG, Jan CH, Bartel DP (2007) Intronic microRNA precursors that bypass Drosha processing. Nature 448:83–86. https://doi.org/10.1038/nature05983
Rybak-Wolf A, Jens M, Murakawa Y et al (2014) A variety of dicer substrates in human and C. elegans. Cell 159:1153–1167. https://doi.org/10.1016/j.cell.2014.10.040
Schultz KAP, Stewart DR, Kamihara J, et al (2020) DICER1 Tumor Predisposition. GeneReviews®
Scott MS, Avolio F, Ono M et al (2009) Human miRNA precursors with box H/ACA snoRNA features. PLoS Comput Biol 5:e1000507. https://doi.org/10.1371/journal.pcbi.1000507
Sehnal D, Bittrich S, Deshpande M et al (2021) Mol∗Viewer: modern web app for 3D visualization and analysis of large biomolecular structures. Nucleic Acids Res 49:W431–W437. https://doi.org/10.1093/nar/gkab314
Shabalina SA, Koonin EV (2008) Origins and evolution of eukaryotic RNA interference. Trends Ecol Evol 23:578–587. https://doi.org/10.1016/j.tree.2008.06.005
Singh S, Bevan SC, Patil K et al (2005) Extensive variation in the 5′-UTR of dicer mRNAs influences translational efficiency. Biochem Biophys Res Commun 335:643–650. https://doi.org/10.1016/j.bbrc.2005.07.138
Sinha NK, Iwasa J, Shen PS, Bass BL (2018) Dicer uses distinct modules for recognizing dsRNA termini. Science 359(80):329–334. https://doi.org/10.1126/science.aaq0921
Song JJ, Liu J, Tolia NH et al (2003) The crystal structure of the Argonaute2 PAZ domain reveals an RNA binding motif in RNAi effector complexes. Nat Struct Biol 10:1026–1032. https://doi.org/10.1038/nsb1016
Song MS, Rossi JJ (2017) Molecular mechanisms of dicer: endonuclease and enzymatic activity. Biochem J 474:1603–1618. https://doi.org/10.1042/BCJ20160759
Stavast CJ, Erkeland SJ (2019) The non-canonical aspects of MicroRNAs: many roads to gene regulation. Cells 8:1465. https://doi.org/10.3390/cells8111465
Su X, Chakravarti D, Cho MS et al (2010) TAp63 suppresses metastasis through coordinate regulation of dicer and miRNAs. Nature 467:986–990. https://doi.org/10.1038/nature09459
Swahari V, Nakamura A, Baran-Gale J et al (2016) Essential function of dicer in resolving DNA damage in the rapidly dividing cells of the develo** and malignant cerebellum. Cell Rep 14:216–224. https://doi.org/10.1016/j.celrep.2015.12.037
Theotoki EI, Pantazopoulou VI, Georgiou S et al (2020) Dicing the disease with dicer: the implications of dicer ribonuclease in human pathologies. Int J Mol Sci 21:7223. https://doi.org/10.3390/ijms21197223
Tian Y, Simanshu DK, Ma JB et al (2014) A phosphate-binding pocket within the platform-PAZ-connector helix cassette of human dicer. Mol Cell 53:606–616. https://doi.org/10.1016/j.molcel.2014.01.003
Van Den Beucken T, Koch E, Chu K et al (2014) Hypoxia promotes stem cell phenotypes and poor prognosis through epigenetic regulation of DICER. Nat Commun 5:1–13. https://doi.org/10.1038/ncomms6203
Welker NC, Maity TS, Ye X et al (2011) Dicer’s helicase domain discriminates dsRNA termini to promote an altered reaction mode. Mol Cell 41:589–599. https://doi.org/10.1016/j.molcel.2011.02.005
Wiesen JL, Tomasi TB (2009) Dicer is regulated by cellular stresses and interferons. Mol Immunol 46:1222–1228. https://doi.org/10.1016/j.molimm.2008.11.012
Wojnicka M, Szczepanska A, Kurzynska-Kokorniak A (2020) Unknown areas of activity of human ribonuclease dicer: a putative deoxyribonuclease activity. Molecules 25:1414. https://doi.org/10.3390/molecules25061414
Zhang H, Kolb FA, Brondani V et al (2002) Human dicer preferentially cleaves dsRNAs at their termini without a requirement for ATP. EMBO J 21:5875–5885. https://doi.org/10.1093/emboj/cdf582
Zhang H, Kolb FA, Jaskiewicz L et al (2004) Single processing center models for human dicer and bacterial RNase III. Cell 118:57–68. https://doi.org/10.1016/j.cell.2004.06.017
Funding
Authors were supported by grants of FAPESP (2019/01587–1 to C.A.V-J and 2019/25958–9 to M.A.M), CNPq (310287/2018–9 to M.A.M.) and CAPES (“This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance Code 001” to G.T-S and M.D.I).
Author information
Authors and Affiliations
Contributions
CAVS, GTS, MDI, and MAM wrote the paper. CAVS, GTS, and MDI contributed equally.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Carlos A. Vergani-Junior, Guilherme Tonon-da-Silva and Mehmet Dinçer Inan have contributed equally.
Rights and permissions
About this article
Cite this article
Vergani-Junior, C.A., Tonon-da-Silva, G., Inan, M.D. et al. DICER: structure, function, and regulation. Biophys Rev 13, 1081–1090 (2021). https://doi.org/10.1007/s12551-021-00902-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-021-00902-w