Abstract
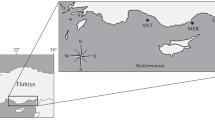
The Spotted sardinella Amblygaster sirm are small pelagic fish that are important protein source to coastal communities in the Indo-West Pacific. In this study, a cytochrome c oxidase subunit 1 (COI) gene of A. sirm from Tanzania was amplified to assess the species’ genetic structure and demographic history. All individuals collected were identified using both morphological examination and genetic barcoding as A. sirm. A total of 19 haplotypes were found in the dataset, with low overall nucleotide (π = 0.13 ± 0.001) and moderate haplotype diversities (h = 0.45 ± 0.07). AMOVA revealed a very low and non-significant genetic differentiation in the dataset (Fst = 0.002, Фst = −0.004, p > 0.05), indicating a lack of population structure. The minimum spanning haplotype network revealed additional evidence for the lack of population structure, which grouped all the sampled haplotypes into one cluster, regardless of their geographical regions. The Tajima’s D, Fu’s Fs tests, and mismatch distribution analyses supported a hypothesis of recent demographic expansion. The lack of population structure identified suggests that the fishery should be treated as a single-stock management unit, consistent with the existing management regime for the species in Mainland Tanzania that currently does not consider genetic structure in managing the fishery. The lack of population structure suggests that populations with low genetic diversity, such as Dar es Salaam, can rebuild by recruiting from other sites if regulations against unsustainable fishing are strictly enforced.





Similar content being viewed by others
References
Alves MJ, Coelho H, Collares-Pereira MJ, Coelho MM (2001) Mitochondrial DNA variation in the highly endangered cyprinid fish Anaecypris hispanica: importance for conservation. Heredity 87:463–473. https://doi.org/10.1046/j.1365-2540.2001.00929.x
Bandelt H, Forster P, Röhl A (1999) Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol 16:37–48. https://doi.org/10.1093/oxfordjournals.molbev.a026036
Bazin E, Glémin S (2006) Population size does not influence mitochondrial genetic diversity in animals. Science 312:570–572. https://doi.org/10.1126/science.1122033
Beerli P, Palczewski M (2010) Unified Framework to evaluate panmixia and migration direction among Multiple Sampling Locations. Genetics 185:313–326. https://doi.org/10.1534/GENETICS.109.112532
Bianchi G (1985) Field guide to the commercial marine and brackish-water species of Tanzania. FAO species identification sheets for fishery purposes. TCP/URT/4406 and FAO (FIRM) Regular Programme FAO, Rome
Chanthran SS, Lim PE, Li Y, Liao TY, Poong SW, Du J, Hussein MA, Sade A, Rumpet R, Loh KH (2020) Genetic diversity and population structure of Terapon jarbua (Forskål, 1775) (Teleostei, Terapontidae) in Malaysian waters. Zookeys 911:139–160. https://doi.org/10.3897/ZOOKEYS.911.39222
Conand F (1991) Biology and phenology of Amblygaster sirm (Clupeidae) in New Caledonia, a sardine of the coral environment. Bull Mar Sci 48:137–149
Cowen R, Gawarkiewicz G, Pineda J (2007) Population connectivity in marine systems an overview. Oceanography 20:14–21
Cowen R, Sponaugle S (2009) Larval dispersal and marine population connectivity. Annu Rev Mar 2009:443–466. https://doi.org/10.1146/annurev.marine.010908.163757
Díaz-Arce N, Grewe P, Krug I, Artetxe I, Ruiz J, Nikolic N, Medieu A, Pernak M, Lansdell M, Aulich J, Clear N (2020) Evidence of connectivity of bigeye tuna (Thunnus obesus) throughout the Indian Ocean inferred from genome-wide genetic markers. IOTC-2020-WPTT22 (AS) https://doi.org/www.hal.archivesouvertes. fr/hal-03206834
Drummond AJ, Suchard MA, **e D, Rambaut A (2012) Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol Biol Evol 29:1969–1973
Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567. https://doi.org/10.1111/J.1755-0998.2010.02847.X
Excoffier L, Schneider S (1999) Why hunter-gatherer populations do not show signs of Pleistocene demographic expansions. Proc Natl Acad Sci U S A 96:10597–10602. https://doi.org/10.1073/PNAS.96.19.10597
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131:479–491. https://doi.org/10.1093/GENETICS/131.2.479
FAO (2018) The state of world fisheries and aquaculture - meeting the sustainable goals. FAO, Rome
Fratini S, Ragionieri L, Cannicci S (2016) Demographic history and reproductive output correlates with intraspecific genetic variation in seven species of Indo-Pacific mangrove crabs. PLoS One 11:e0158582. https://doi.org/10.1371/JOURNAL.PONE.0158582
Fréon P, Cury P, Shannon L, Roy C (2005) Sustainable exploitation of small pelagic fish stocks challenged by environmental and ecosystem changes: a review. Bull Mar Sci 76:385–462
Fricke R, Mulochau T, Chabanet P et al (2009) Annotated checklist of the fish species (Pisces) of La Réunion, including a red list of threatened and declining species. Stuttg Beitr Naturk A, N Ser 2:1–168
Fu XY (1997) Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection. Genetics 147:915–925
Ganias K, Somarakis S, Nunes (2014) Reproductive potential. In: Ganias K (ed) Biology and Ecology of Sardines and Anchovies. CRC Press: Boca Raton, FL, USA, pp 79–121
Grant WS, Bowen BW (1998) Shallow population histories in deep evolutionary lineages of marine fishes: insights from sardines and anchovies and lessons for conservation. J Hered 89:415–426. https://doi.org/10.1093/JHERED/89.5.415
Hadi S, Andayani N, Muttaqin E et al (2020) Genetic connectivity of the scalloped hammerhead shark Sphyrna lewini across Indonesia and the Western Indian Ocean. PLoS One 15:e0230763. https://doi.org/10.1371/JOURNAL.PONE.0230763
Harpending H (1994) Signature of ancient population growth in a low-resolution mitochondrial DNA mismatch Distribution. Hum Biol 66:591–600
Hoareau TB, Boissin E (2010) Design of phylum-specific hybrid primers for DNA barcoding: addressing the need for efficient COI amplification in the Echinodermata. Mol Ecol Resour 10:960–967. https://doi.org/10.1111/j.1755-0998.2010.02848.x
Holm S (1979) A simple sequentially rejective multiple test procedure. Scand J Stat 6:65–70 https://doi.org/http://www.jstor.org/stable/4615733
Hunnam K (2021) The biology and ecology of tropical marine sardines and herrings in Indo-West Pacific fisheries: a review. Rev Fish Biol Fish 31:449–484. https://doi.org/10.1007/S11160-021-09649-9
Huyghe F, Kochzius M (2017) Highly restricted gene flow between disjunct populations of the skunk clownfish (Amphiprion akallopisos) in the Indian Ocean. Mar Ecol 38:e12357. https://doi.org/10.1111/MAEC.12357
Huyghe F, Kochzius M (2018) Sea surface currents and geographic isolation shape the genetic population structure of a coral reef fish in the Indian Ocean. PLoS One 13:e0193825. https://doi.org/10.1371/JOURNAL.PONE.0193825
Isaacs M (2016) The humble sardine (small pelagics): fish as food or fodder. Agric Food Secur 5:1–14. https://doi.org/10.1186/s40066-016-0073-5
Ivanova P, Dzhembekova N, Atanassov I, Rusanov K, Raykov V, Zlateva I, Yankova M, Raev Y, Nikolov G (2021) Genetic diversity and morphological characterisation of three turbot (Scophthalmus maximus L., 1758) populations along the Bulgarian Black Sea coast. Nat Conserv 43:123–146. https://doi.org/10.3897/natureconservation.43.64195
Jamaludin NA, Jamaluddin JA, Rahim MA, Akib NA, Ratmuangkhwang S, Arshaad WM, Nor SA (2022) Mitochondrial marker implies fishery separate management units for spotted sardinella, Amblygaster sirm (Walbaum, 1792) populations in the South China Sea and the Andaman Sea. PeerJ 10:e13706. https://doi.org/10.7717/peerj.13706
Johnson MG, Mgaya YD, Shaghude YW (2021) Analysis of the genetic stock structure and phylogenetic relationship of narrow-barred Spanish mackerel Scomberomorus commerson (Lacépède, 1800) along the northern Tanzanian coastal waters using mitochondrial DNA. Reg Stud Mar Sci 46:101862. https://doi.org/10.1016/J.RSMA.2021.101862
Kerr LA, Hintzen NT, Cadrin SX, Clausen LW, Dickey-Collas M, Goethel DR, Hatfield EM, Kritzer JP, Nash RD (2017) Lessons learned from practical approaches to reconcile mismatches between biological population structure and stock units of marine fish. ICES J Mar Sci 74:1708–1722. https://doi.org/10.1093/icesjms/fsw188
Lage C, Kuhn K, Kornfield I (2004) Genetic differentiation among Atlantic cod (Gadus morhua) from Browns Bank, Georges Bank, and Nantucket Shoals. Fish Bull 102:289–298
Letourneur Y, Chabanet P, Durville P et al (2004) An updated checklist of the marine fish fauna of Reunion Island, south-western Indian Ocean. Cybium 28:199–216. https://doi.org/10.26028/cybium/2004-283-002
McClanahan T (1988) Seasonality in East Africa’s coastal waters. Mar Ecol Prog Ser 44:191–199. https://doi.org/10.3354/meps044191
Mendez M, Rosenbaum HC, Subramaniam A, Yackulic C, Bordino P (2010) Isolation by environmental distance in mobile marine species: molecular ecology of franciscana dolphins at their southern range. Wiley Online Libr 19:2212–2228. https://doi.org/10.1111/j.1365-294X.2010.04647.x
Milton D, Blaber S, Rawlinson N (1994) Reproductive biology and egg production of three species of Clupeidae from Kiribati, tropical central Pacific. Fish Bull 92:102–121
Muhando CA, Mohammed MS (2002) Coral reef benthos and fisheries in Tanzania before and after the 1998 bleaching and mortality event. West Indian Ocean J Mar Sci 1:43–52
Mwakosya CA, Mgaya YD, Jiddawi NS (2018) Genetic connectivity of Fenneropenaeus indicus (H. Milne Edwards 1837) among three prawn fishing grounds of Tanzanian coastal waters. Reg Stud Mar Sci 24:107–112. https://doi.org/10.1016/j.rsma.2018.08.001
Nyandwi N (2013) The effects of monsoons on the East African Coastal Current through the Zanzibar Channel, Tanzania. J Ocean Technol 8:63–74
Obura D, Bandeira S, Bodin N, Braulik G (2019) Chapter 4 - the northern Mozambique channel. In: Sheppard C (ed) World seas: an environmental evaluation, 2nd edn. Academic Press, pp 75–99
Postaire B, Gélin P, Bruggemann JH, Magalon H (2017) One species for one island? Unexpected diversity and weak connectivity in a widely distributed tropical hydrozoan. Heredity 118:385–394. https://doi.org/10.1038/hdy.2016.126
Pradeep HD, Shirke SS, Kar AB (2014) Age, growth and mortality of Amblygaster sirm (Walbaum, 1792) from Andaman waters. J Andaman Sci Assoc 19:201–208
Rahimi P, Rezvani Gilkolaie S, Ghavam Mostafavi P, Jamili S, Rahnema M (2016) Population genetic structure of the white sardine, Sardinella albella, in the Persian Gulf and Sea of Oman by analysis of mitochondrial control region. Iran J Fish Sci 15:995–1008
Rambaut A, Drummond AJ, **e D, Baele G, Suchard MA (2018) Posterior summarization in Bayesian phylogenetics using Tracer 1.7. Syst Biol 67:901–904. https://doi.org/10.1093/sysbio/syy032
Reiss H, Hoarau G, Dickey-Collas M, Wolff WJ (2009) Genetic population structure of marine fish: mismatch between biological and fisheries management units. Fish Fish 10:361–395. https://doi.org/10.1111/j.1467-2979.2008.00324.x
Rogers AR, Harpending H (1992) Population growth makes waves in the distribution of pairwise genetic differences. Mol Biol Evol 9:552–569. https://doi.org/10.1093/OXFORDJOURNALS.MOLBEV.A040727
Rumisha C, Bugota VJ, Mtonga CJ (2023) Evidence of extensive genetic connectivity and recent demographic expansion of the queen mackerel Scomberomorus plurilineatus in Tanzanian coastal waters. Mar Biodivers 53:9. https://doi.org/10.1007/s12526-022-01315-y
Rumisha C, Huyghe F, Rapanoel D, Mascaux N, Kochzius M (2017) Genetic diversity and connectivity in the East African giant mud crab Scylla serrata: Implications for fisheries management. PLoS One 12:e0186817
Rumisha C, Kochzius M (2022) Genetic evidence for a single stock of giant tiger prawns Penaeus monodon in demarcated prawn fishing zones of Tanzania. Fish Manag Ecol 30:36–43. https://doi.org/10.1111/FME.12598
Rumisha C, Mdegela RH, Gwakisa PS, Kochzius M (2018) Genetic diversity and gene flow among the giant mud crabs (Scylla serrata) in anthropogenic-polluted mangroves of mainland Tanzania: implications for conservation. Fish Res 205:96–104. https://doi.org/10.1016/j.fishres.2018.04.015
Russell B, Houston W (1989) Offshore fishes of the Arafura Sea. Beagle Rec North Territ Museum Arts Sci 6:69–84
Russo CAM, Selvatti AP (2018) Bootstrap and rogue identification tests for phylogenetic analyses. Mol Biol Evol 35:2327–2333
Sahyoun R, Guidetti P, Di Franco A, Planes S (2016) Patterns of fish connectivity between a marine protected area and surrounding fished areas. PLoS One 11:e0167441. https://doi.org/10.1371/JOURNAL.PONE.0167441
Saleh MF, Arshaad WM, Hassan RB, Masaya K, Latun AR, Fatah NN, Jaafar K (2020) Towards the sustainable management of purse seine fisheries in Southeast Asia. In: Fish for the People. SEAFDEC, Bangkok, pp 20–26
Semba M, Lumpkin R, Kimirei I, Shaghude Y, Nyandwi N (2019) Seasonal and spatial variation of surface current in the Pemba Channel, Tanzania. PLoS One 14:e0210303. https://doi.org/10.1371/journal.pone.0210303
Silva SE, Silva IC, Madeira C, Sallema R, Paulo OS, Paula J (2013) Genetic and morphological variation in two littorinid gastropods: evidence for recent population expansions along the East African coast. Biol J Linn Soc 108:494–508. https://doi.org/10.1111/J.1095-8312.2012.02041.X
Sululu JS, Kamukuru AT, Sekadende BC, Mahongo SB, Igulu MM (2020) Reproductive biology of the anchovy (Stolephorus commersonnii, Lacepède, 1803) and spotted sardine (Amblygaster sirm, Walbaum, 1792) from Tanga Region, Tanzania. West Indian Ocean J Mar Sci:81–94. https://doi.org/10.4314/wiojms.si2020.1.8
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123:585–595. https://doi.org/10.1093/genetics/123.3.585
Tamura K, Stecher G, Kumar S (2021) MEGA11: Molecular evolutionary genetics analysis version 11. Mol Biol Evol 38:3022–3027. https://doi.org/10.1093/molbev/msab120
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680. https://doi.org/10.1093/NAR/22.22.4673
Van Nieuwenhove AHM, Ratsimbazafy HA, Kochzius M (2019) Cryptic diversity and limited connectivity in octopuses: recommendations for fisheries management. PLoS One 14:e0214748. https://doi.org/10.1371/journal.pone.0214748
Villesen P (2007) FaBox: an online toolbox for fasta sequences. Mol Ecol Notes 7:965–968. https://doi.org/10.1111/J.1471-8286.2007.01821.X
Whitehead PJP (1988) FAO species catalogue: an annotated and illustrated catalogue of the herrings, sardines, pilchards, sprats, shads, anchovies and wolf-herrings. Pt 2 – Engraulididae. FAO Fisheries Synopsis, Rome
Zemeckis DR, Martins D, Kerr LA, Cadrin SX (2014) Stock identification of Atlantic cod (Gadus morhua) in US waters: an interdisciplinary approach. ICES J Mar Sci 71:1490–1506. https://doi.org/10.4135/9781412953924.n678
Zhang DX, Hewitt GM (1996) Nuclear integrations: challenges for mitochondrial DNA markers. Trends Ecol Evol 11:247–251. https://doi.org/10.1016/0169-5347(96)10031-8
Zink RM, Barrowclough GF (2008) Mitochondrial DNA under siege in avian phylogeography. Mol Ecol 17:2107–2121. https://doi.org/10.1111/J.1365-294X.2008.03737.X
Acknowledgements
The authors would like to thank the Western Indian Ocean Marine Sciences Association (WIOMSA) through the MARG I 2019 program for financial support to conduct this study. The authors also would like to thank the Tanzania Fisheries Research Institute (TAFIRI) and the Institute of Marine Sciences (IMS) for hosting and providing logistical support. Thanks to the College of Natural and Applied Sciences (CoNAS) of the Sokoine University of Agriculture (SUA) for providing space for molecular laboratory analysis. The authors appreciate the assistance of Asha Said and Alex Zuberi at the Department of Biosciences, SUA, during sample analysis in the lab. Also, the authors would like to thank all anonymous reviewers whose comments and advice helped improve this manuscript.
Funding
This study was funded by the Western Indian Ocean Marine Sciences Association (WIOMSA) through the MARG I 2019 program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
All applicable international guidelines for animal testing and the use of animals were followed by the authors.
Sampling and field studies
All necessary permits for sampling and observational field studies have been obtained by the authors from the competent authorities and are mentioned in the acknowledgements. The study is compliant with CBD and Nagoya protocols.
Data availability
The dataset of all COI sequences are available in the GenBank repository with the accession numbers ON631654 - ON631731.
Authors’ contribution
The study was conceptualized, planned, and carried out by Fabiani G and Chauka JL. Fabiani G and Rumisha C analyzed laboratory molecular samples. Fabiani G and Mtonga JC conducted a statistical analysis of the data. Mtonga JC and Fabiani G wrote the manuscript. Muhando AC, Chauka JL, and Rumisha C revised the manuscript. The final paper was read and approved by all authors.
Additional information
Communicated by R. Thiel
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fabiani, G., Mtonga, C.J., Chauka, L.J. et al. Mitochondrial DNA revealed a single stock structure of the Spotted sardinella Amblygaster sirm (Walbaum, 1792) (Teleostei; Clupeidae) in Tanzanian coastal waters. Mar. Biodivers. 53, 40 (2023). https://doi.org/10.1007/s12526-023-01351-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12526-023-01351-2