Abstract
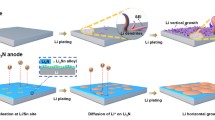
Lithium metal is commonly regarded as the “Holy Grail” anode material for high energy density rechargeable batteries. However, the uncontrollable growth of Li dendrites has posed safety concerns and thus greatly hindered its large-scale application. Here we have modified the surface of a commercial anode current collector, Cu foil, with a thin layer of Zn by a facile electroplating method, in order to regulate the Li nucleation and the following growth processes. Because of the formation of a solid solution buffer layer and Li-Zn alloy phases, the Li nucleation overpotential was dramatically reduced, realizing a uniform Li nucleation and a smooth Li plating morphology. As a result, significantly improved long-term cycling performance with a high Coulombic efficiency was achieved by the lithiophilic Zn coated Cu foil as a current collector. Full cells of Li–LiFePO4 and Li–S using the Li deposited on the Zn modified Cu as the anode, showed increased capacity with low voltage hysteresis and greatly enhanced cycling stability, ascribed to the uniform Li deposition and formation of a stable SEI layer. This work demonstrates the feasibility of employing lithiophilic modified Cu foils as Li metal current collectors for practical applications.

Similar content being viewed by others
References
Armand, M.; Tarascon, J. M. Building better batteries. Nature2008, 451, 652–657.
Tarascon, J. M.; Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature2001, 414, 359–367.
Lu, Y.; Yu, L.; Lou, X. W. Nanostructured conversion-type anode materials for advanced lithium-ion batteries. Chem2018, 4, 972–996.
Xu, W.; Wang, J. L.; Ding, F.; Chen, X. L.; Nasybulin, E.; Zhang, Y. H.; Zhang, J. G. Lithium metal anodes for rechargeable batteries. Energy Environ. Sci. 2014, 7, 513–537.
Bruce, P. G.; Freunberger, S. A.; Hardwick, L. J.; Tarascon, J. M. Li-O2 and Li-S batteries with high energy storage. Nat. Mater. 2012, 11, 19–29.
Yu, S. H.; Feng, X. R.; Zhang, N.; Seok, J.; Abruña, H. D. Understanding conversion-type electrodes for lithium rechargeable batteries. Acc. Chem. Res. 2018, 51, 273–281.
Zhang, N.; Levin, B. D. A.; Yang, Y.; Muller, D. A.; Abruña, H. D. Porous Fe3O4 nanospheres as effective sulfur hosts for Li–S batteries. J. Electrochem. Soc. 2018, 165, A1656–A1661.
Zhang, N.; Yang, Y.; Feng, X. R.; Yu, S. H.; Seok, J.; Muller, D. A.; Abruña, H. D. Sulfur encapsulation by MOF-derived CoS2 embedded in carbon hosts for high-performance Li–S batteries. J. Mater. Chem. A2019, 7, 21128–21139.
Zhang, J. T.; Li, Z.; Chen, Y.; Gao, S. Y.; Lou, X. W. D. Nickel-iron layered double hydroxide hollow polyhedrons as a superior sulfur host for lithium-sulfur batteries. Angew. Chem., Int. Ed.2018, 57, 10944–10948.
Cheng, X. B.; Zhang, R.; Zhao, C. Z.; Zhang, Q. Toward safe lithium metal anode in rechargeable batteries: A review. Chem. Rev. 2017, 117, 10403–10473.
Lin, D. C.; Liu, Y. Y.; Cui, Y. Reviving the lithium metal anode for high-energy batteries. Nat. Nanotechnol. 2017, 12, 194–206.
Peled, E. The electrochemical behavior of alkali and alkaline earth metals in nonaqueous battery systems—the solid electrolyte interphase model. J. Electrochem. Soc. 1979, 126, 2047–2051.
Cheng, X. B.; Zhang, R.; Zhao, C. Z.; Wei, F.; Zhang, J. G.; Zhang, Q. A review of solid electrolyte interphases on lithium metal anode. Adv. Sci. 2016, 3, 1500213.
Liu, J.; Bao, Z. A.; Cui, Y.; Dufek, E. J.; Goodenough, J. B.; Khalifah, P.; Li, Q. Y.; Liaw, B. Y.; Liu, P.; Manthiram, A. et al. Pathways for practical high-energy long-cycling lithium metal batteries. Nat. Energy2019, 4, 180–186.
Cohen, Y. S.; Cohen, Y.; Aurbach, D. Micromorphological studies of lithium electrodes in alkyl carbonate solutions using in situ atomic force microscopy. J. Phys. Chem. B2000, 104, 12282–12291.
Yan, K.; Lu, Z. D.; Lee, H. W.; **ong, F.; Hsu, P. C.; Li, Y. Z.; Zhao, J.; Chu, S.; Cui, Y. Selective deposition and stable encapsulation of lithium through heterogeneous seeded growth. Nat. Energy2016, 1, 16010.
Steiger, J.; Kramer, D.; Mönig, R. Mechanisms of dendritic growth investigated by in situ light microscopy during electrodeposition and dissolution of lithium. J. Power Sources2014, 261, 112–119.
Chen, K. H.; Wood, K. N.; Kazyak, E.; LePage, W. S.; Davis, A. L.; Sanchez, A. J.; Dasgupta, N. P. Dead lithium: Mass transport effects on voltage, capacity, and failure of lithium metal anodes. J. Mater. Chem. A2017, 5, 11671–11681.
Lv, S. S.; Verhallen, T.; Vasileiadis, A.; Ooms, F.; Xu, Y. L.; Li, Z. L.; Li, Z. C.; Wagemaker, M. Operando monitoring the lithium spatial distribution of lithium metal anodes. Nat. Commun. 2018, 9, 2152.
Aurbach, D.; Gofer, Y.; Langzam, J. The correlation between surface chemistry, surface morphology, and cycling efficiency of lithium electrodes in a few polar aprotic systems. J. Electrochem. Soc. 1989, 136, 3198–3205.
Lu, Q. W.; He, Y. B.; Yu, Q. P.; Li, B. H.; Kaneti, Y. V.; Yao, Y. W.; Kang, F. Y.; Yang, Q. H. Dendrite-free, high-rate, long-life lithium metal batteries with a 3D cross-linked network polymer electrolyte. Adv. Mater. 2017, 29, 1604460.
Cheng, X. B.; Yan, C.; Peng, H. J.; Huang, J. Q.; Yang, S. T.; Zhang, Q. Sulfurized solid electrolyte interphases with a rapid Li+ diffusion on dendrite-free Li metal anodes. Energy Storage Mater. 2018, 10, 199–205.
Zheng, Q.; Ma, L.; Khurana, R.; Archer, L. A.; Coates, G. W. Structure-property study of cross-linked hydrocarbon/poly(ethylene oxide) electrolytes with superior conductivity and dendrite resistance. Chem. Sci. 2016, 7, 6832–6838.
Zheng, Q.; Pesko, D. M.; Savoie, B. M.; Timachova, K.; Hasan, A. L.; Smith, M. C.; Miller III, T. F.; Coates, G. W.; Balsara, N. P. Optimizing ion transport in polyether-based electrolytes for lithium batteries. Macromolecules2018, 51, 2847–2858.
Li, J. C.; Ma, C.; Chi, M. F.; Liang, C. D.; Dudney, N. J. Solid electrolyte: The key for high-voltage lithium batteries. Adv. Energy Mater. 2015, 5, 1401408.
Croce, F.; Persi, L.; Ronci, F.; Scrosati, B. Nanocomposite polymer electrolytes and their impact on the lithium battery technology. Solid State Ionics2000, 135, 47–52.
Kim, K. H.; Iriyama, Y.; Yamamoto, K.; Kumazaki, S.; Asaka, T.; Tanabe, K.; Fisher, C. A. J.; Hirayama, T.; Murugan, R.; Ogumi, Z. Characterization of the interface between LiCoO2 and Li7La3Zr2O12 in an all-solid-state rechargeable lithium battery. J. Power Sources2011, 196, 764–767.
Miao, R. R.; Yang, J.; Xu, Z. X.; Wang, J. L.; Nuli, Y.; Sun, L. M. A new ether-based electrolyte for dendrite-free lithium-metal based rechargeable batteries. Sci. Rep. 2016, 6, 21771.
Ding, F.; Xu, W.; Graff, G. L.; Zhang, J.; Sushko, M. L.; Chen, X. L.; Shao, Y. Y.; Engelhard, M. H.; Nie, Z. M.; **ao, J. et al. Dendrite-free lithium deposition via self-healing electrostatic shield mechanism. J. Am. Chem. Soc. 2013, 135, 4450–4456.
Li, W. Y.; Yao, H. B.; Yan, K.; Zheng, G. Y.; Liang, Z.; Chiang, Y. M.; Cui, Y. The synergetic effect of lithium polysulfide and lithium nitrate to prevent lithium dendrite growth. Nat. Commun. 2015, 6, 7436.
Liu, Y. Y.; Lin, D. C.; Li, Y. Z.; Chen, G. X.; Pei, A.; Nix, O.; Li, Y. B.; Cui, Y. Solubility-mediated sustained release enabling nitrate additive in carbonate electrolytes for stable lithium metal anode. Nat. Commun. 2018, 9, 3656.
Mogi, R.; Inaba, M.; Jeong, S. K.; Iriyama, Y.; Abe, T.; Ogumi, Z. Effects of some organic additives on lithium deposition in propylene carbonate. J. Electrochem. Soc. 2002, 149, A1578–A1583.
Yun, Q. B.; He, Y. B.; Lv, W.; Zhao, Y.; Li, B. H.; Kang, F. Y.; Yang, Q. H. Chemical dealloying derived 3D porous current collector for Li metal anodes. Adv. Mater. 2016, 28, 6932–6939.
Lu, L. L.; Ge, J.; Yang, J. N.; Chen, S. M.; Yao, H. B.; Zhou, F.; Yu, S. H. Free-standing copper nanowire network current collector for improving lithium anode performance. Nano Lett. 2016, 16, 4431–4437.
Zhang, Y.; Luo, W.; Wang, C. W.; Li, Y. J.; Chen, C. J.; Song, J. W.; Dai, J. Q.; Hitz, E. M.; Xu, S. M.; Yang, C. P. et al. High-capacity, low-tortuosity, and channel-guided lithium metal anode. Proc. Natl. Acad. Sci. USA2017, 114, 3584–3589.
Ji, X. L.; Liu, D. Y.; Prendiville, D. G.; Zhang, Y. C.; Liu, X. N.; Stucky, G. D. Spatially heterogeneous carbon-fiber papers as surface dendrite-free current collectors for lithium deposition. Nano Today2012, 7, 10–20.
Yang, C. P.; Yin, Y. X.; Zhang, S. F.; Li, N. W.; Guo, Y. G. Accommodating lithium into 3D current collectors with a submicron skeleton towards long-life lithium metal anodes. Nat. Commun. 2015, 6, 8058.
Chen, M.; Zheng, J. H.; Sheng, O. W.; **, C. B.; Yuan, H. D.; Liu, T. F.; Liu, Y. J.; Wang, Y.; Nai, J. W.; Tao, X. Y. Sulfur–nitrogen co-doped porous carbon nanosheets to control lithium growth for a stable lithium metal anode. J. Mater. Chem. A2019, 7, 18267–18274.
Tang, L. S.; Zhang, R.; Zhang, X. Y.; Zhao, N. Q.; Shi, C. S.; Liu, E. Z.; Ma, L. Y.; Luo, J. Y.; He, C. N. ZnO nanoconfined 3D porous carbon composite microspheres to stabilize lithium nucleation/growth for high-performance lithium metal anodes. J. Mater. Chem. A2019, 7, 19442–19452.
Cha, E.; Patel, M. D.; Park, J.; Hwang, J.; Prasad, V.; Cho, K.; Choi, W; 2 D M o S 2 as an efficient protective layer for lithium metal anodes in high-performance Li–S batteries. Nat. Nanotechnol. 2018, 13, 337–344.
Yan, K.; Lee, H. W.; Gao, T.; Zheng, G. Y.; Yao, H. B.; Wang, H. T.; Lu, Z. D.; Zhou, Y.; Liang, Z.; Liu, Z. F. et al. Ultrathin two-dimensional atomic crystals as stable interfacial layer for improvement of lithium metal anode. Nano Lett. 2014, 14, 6016–6022.
**e, J.; Liao, L.; Gong, Y. J.; Li, Y. B.; Shi, F. F.; Pei, A.; Sun, J.; Zhang, R. F.; Kong, B.; Subbaraman, R. et al. Stitching h-BN by atomic layer deposition of LiF as a stable interface for lithium metal anode. Sci. Adv. 2017, 3, eaao3170.
Liu, Y. Y.; Lin, D. C.; Liang, Z.; Zhao, J.; Yan, K.; Cui, Y. Lithium-coated polymeric matrix as a minimum volume-change and dendrite-free lithium metal anode. Nat. Commun. 2016, 7, 10992.
Luo, J.; Fang, C. C.; Wu, N. L. High polarity poly(vinylidene difluoride) thin coating for dendrite-free and high-performance lithium metal anodes. Adv. Energy Mater. 2018, 8, 1701482.
Zhang, C.; Lv, W.; Zhou, G. M.; Huang, Z. J.; Zhang, Y. B.; Lyu, R.; Wu, H. L.; Yun, Q. B.; Kang, F. Y.; Yang, Q. H. Vertically aligned lithiophilic CuO nanosheets on a Cu collector to stabilize lithium deposition for lithium metal batteries. Adv. Energy Mater. 2018, 8, 1703404.
Zhang, N.; Yu, S. H.; Abruña, H. D. Uniform lithium deposition on N-doped carbon-coated current collectors. Chem. Commun. 2019, 55, 10124–10127.
Burns, J. C.; Kassam, A.; Sinha, N. N.; Downie, L. E.; Solnickova, L.; Way, B. M.; Dahn, J. R. Predicting and extending the lifetime of Li-ion batteries. J. Electrochem. Soc. 2013, 160, A1451–A1456.
Adams, B. D.; Zheng, J. M.; Ren, X. D.; Xu, W.; Zhang, J. G. Accurate determination of Coulombic efficiency for lithium metal anodes and lithium metal batteries. Adv. Energy Mater. 2018, 8, 1702097.
Wood, K. N.; Kazyak, E.; Chadwick, A. F.; Chen, K. H.; Zhang, J. G.; Thornton, K.; Dasgupta, N. P. Dendrites and pits: Untangling the complex behavior of lithium metal anodes through operando video microscopy. ACS Cent. Sci. 2016, 2, 790–801.
Wang, X.; Zeng, W.; Hong, L.; Xu, W. W.; Yang, H. K.; Wang, F.; Duan, H. G.; Tang, M.; Jiang, H. Q. Stress-driven lithium dendrite growth mechanism and dendrite mitigation by electroplating on soft substrates. Nat. Energy2018, 3, 227–235.
Liu, W.; Li, W. Y.; Zhuo, D.; Zheng, G. Y.; Lu, Z. D.; Liu, K.; Cui, Y. Core-shell nanoparticle coating as an interfacial layer for dendrite-free lithium metal anodes. ACS Cent. Sci. 2017, 3, 135–140.
Zhang, X. Q.; Cheng, X. B.; Chen, X.; Yan, C.; Zhang, Q. Fluoroethylene carbonate additives to render uniform Li deposits in lithium metal batteries. Adv. Funct. Mater. 2017, 27, 1605989.
Mueller, F.; Geiger, D.; Kaiser, U.; Passerini, S.; Bresser, D. Elucidating the impact of cobalt do** on the lithium storage mechanism in conversion/alloying-type zinc oxide anodes. ChemElectroChem2016, 3, 1311–1319.
Varzi, A.; Mattarozzi, L.; Cattarin, S.; Guerriero, P.; Passerini, S. 3D porous Cu-Zn alloys as alternative anode materials for Li-Ion batteries with superior low t performance. Adv. Energy Mater. 2018, 8, 1701706.
**, C. B.; Sheng, O. W.; Luo, J. M.; Yuan, H. D.; Fang, C.; Zhang, W. K.; Huang, H.; Gan, Y. P.; **a, Y.; Liang, C. et al. 3D lithium metal embedded within lithiophilic porous matrix for stable lithium metal batteries. Nano Energy2017, 37, 177–186.
Zhang, R.; Chen, X. R.; Chen, X.; Cheng, X. B.; Zhang, X. Q.; Yan, C.; Zhang, Q. Lithiophilic sites in doped graphene guide uniform lithium nucleation for dendrite-free lithium metal anodes. Angew. Chem., Int. Ed.2017, 56, 7764–7768.
Okamoto, H. Li-Zn (lithium-Zinc). J. Phase Equilib. Diff.2012, 33, 345–345.
Acknowledgements
This work made use of the Cornell Center for Materials Research (CCMR) Shared Facilities with funding from the NSF MRSEC program [DMR-1719875].
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Zhang, N., Yu, SH. & Abruña, H.D. Regulating lithium nucleation and growth by zinc modified current collectors. Nano Res. 13, 45–51 (2020). https://doi.org/10.1007/s12274-019-2567-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-019-2567-7