Abstract
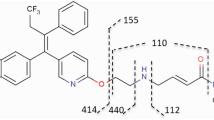
The purpose of this study was to better understand why para-halogen modifications of S-3-(4-halophenoxy)-2-hydroxy-2-methyl-N-(4-nitro-3-trifluoromethylphenyl) propionamide selective androgen receptor modulators (SARMs) had the opposite of expected effects on total clearance, in which electron-withdrawing groups generally protect benzene ring from hydroxylation. We determined the plasma protein binding of this series of halogen substituted SARMs and characterized the qualitative effects of B-ring halogen substitution on in vivo metabolism. In vivo metabolism of S-9, S-10, and S-11 were determined in rats using LC-MSn analysis. Intrinsic clearance was measured by in vitro metabolism using rat liver microsomes. Rat plasma protein binding was measured by equilibrium dialysis and drug concentrations after dialysis were analyzed by LC-MS. The major metabolic pathways of the halogen-substituted SARMs examined were very similar and included three major phase I pathways; (1) hydrolysis of the amide bond, (2) B-ring hydroxylation, and (3) A-ring nitro reduction to an aromatic amine. In plasma protein binding studies, S-1 (F, fu = 0.78 ± 0.17 %) showed the greatest unbound fraction, followed by S-9 (Cl, fu = 0.10 ± 0.04 %), S-10 (Br, fu = 0.03 ± 0.01 %), and S-11 (I, fu = 0.008 ± 0.001 %). The CLint values of S-1, S-9, S-10, and S-11 were 2.4, 2.5, 2.8, and 4.6 μL/min/mg, respectively. These findings suggest that as lipophilicity increased the free fraction was reduced thus compensating for metabolic liability and resulting in the apparent discrepancy between CLint and CL total of halogen-substituted SARMs series.



Similar content being viewed by others
References
Banker, M.J., T.H. Clark, and J.A. Williams. 2003. Development and validation of a 96-well equilibrium dialysis apparatus for measuring plasma protein binding. Journal of Pharmaceutical Sciences 92: 967–974.
Behre, H.M., K. Abshagen, M. Oettel, D. Hubler, and E. Nieschlag. 1999a. Intramuscular injection of testosterone undecanoate for the treatment of male hypogonadism: Phase I studies. European Journal of Endocrinology 140: 414–419.
Behre, H.M., S. von Eckardstein, S. Kliesch, and E. Nieschlag. 1999b. Long-term substitution therapy of hypogonadal men with transscrotal testosterone over 7–10 years. Clin Endocrinol (Oxf) 50: 629–635.
Behre, H.M., C. Wang, D.J. Handelsman, and E. Nieschlag. 2004. Pharmacology of testosterone preparations. In Testosterone, ed. E. Nieschlag, and H.M. Behre, 405–444. Cambridge: Cambridge University Press.
Bhasin, S., R.S. Swerdloff, B. Steiner, M.A. Peterson, T. Meridores, M. Galmirini, M.R. Pandian, R. Goldberg, and N. Berman. 1992. A biodegradable testosterone microcapsule formulation provides uniform eugonadal levels of testosterone for 10–11 weeks in hypogonadal men. Journal of Clinical Endocrinology and Metabolism 74: 75–83.
Boudinot, F.D., and W.J. Jusko. 1984. Fluid shifts and other factors affecting plasma protein binding of prednisolone by equilibrium dialysis. Journal of Pharmaceutical Sciences 73: 774–780.
Boyle, G.W., D. McKillop, P.J. Phillips, J.R. Harding, R. Pickford, and A.D. McCormick. 1993. Metabolism of Casodex in laboratory animals. Xenobiotica 23: 781–798.
Cockshott, I.D., G.F. Plummer, K.J. Cooper, and M.J. Warwick. 1991. The pharmacokinetics of Casodex in laboratory animals. Xenobiotica 21: 1347–1355.
Davies, B., and T. Morris. 1993. Physiological parameters in laboratory animals and humans. Pharmaceutical Research 10: 1093–1095.
Gao, W., J.D. Kearbey, V.A. Nair, K. Chung, A.F. Parlow, D.D. Miller, and J.T. Dalton. 2004. Comparison of the pharmacological effects of a novel selective androgen receptor modulator, the 5alpha-reductase inhibitor finasteride, and the antiandrogen hydroxyflutamide in intact rats: new approach for benign prostate hyperplasia. Endocrinology 145: 5420–5428.
Handelsman, D.J., A.J. Conway, and L.M. Boylan. 1990. Pharmacokinetics and pharmacodynamics of testosterone pellets in man. Journal of Clinical Endocrinology and Metabolism 71: 216–222.
He, Y., D. Yin, M. Perera, L. Kirkovsky, N. Stourman, W. Li, J.T. Dalton, and D.D. Miller. 2002. Novel nonsteroidal ligands with high binding affinity and potent functional activity for the androgen receptor. European Journal of Medicinal Chemistry 37: 619–634.
Jones, T.K., C. Pathirana, M.E. Goldman, L.G. Hamann, L.J. Farmer, T. Ianiro, M.G. Johnson, S.L. Bender, D.E. Mais, and R.B. Stein. 1996. Discovery of novel intracellular receptor modulating drugs. Journal of Steroid Biochemistry and Molecular Biology 56: 61–66.
Kearbey, J.D., D. Wu, W. Gao, D.D. Miller, and J.T. Dalton. 2004. Pharmacokinetics of S-3-(4-acetylamino-phenoxy)-2-hydroxy-2-methyl-N-(4-nitro-3-trifluoromethyl-phenyl)-propionamide in rats, a non-steroidal selective androgen receptor modulator. Xenobiotica 34: 273–280.
Kim, J., D. Wu, D.J. Hwang, D.D. Miller, and J.T. Dalton. 2005. The para substituent of S-3-(phenoxy)-2-hydroxy-2-methyl-N-(4-nitro-3-trifluoromethyl-phenyl)-prop ionamides is a major structural determinant of in vivo disposition and activity of selective androgen receptor modulators. Journal of Pharmacology and Experimental Therapeutics 315: 230–239.
Marhefka, C.A., W. Gao, K. Chung, J. Kim, Y. He, D. Yin, C. Bohl, J.T. Dalton, and D.D. Miller. 2004. Design, synthesis, and biological characterization of metabolically stable selective androgen receptor modulators. Journal of Medicinal Chemistry 47: 993–998.
Mohler, M.L., C.E. Bohl, A. Jones, C.C. Coss, R. Narayanan, Y. He, D.J. Hwang, J.T. Dalton, and D.D. Miller. 2009. Nonsteroidal selective androgen receptor modulators (SARMs): Dissociating the anabolic and androgenic activities of the androgen receptor for therapeutic benefit. Journal of Medicinal Chemistry 52: 3597–3617.
Negro-Vilar, A. 1999. Selective androgen receptor modulators (SARMs): a novel approach to androgen therapy for the new millennium. Journal of Clinical Endocrinology and Metabolism 84: 3459–3462.
Perera, M.A., D. Yin, D. Wu, K.K. Chan, D.D. Miller, and J. Dalton. 2006. In vivo metabolism and final disposition of a novel nonsteroidal androgen in rats and dogs. Drug Metabolism and Disposition 34: 1713–1721.
Smith, H.J., and H.C. Williams. 2006. Smith and Williams’ introduction to the principles of drug design and action. Boca Raton: CRC Press.
Wu, D., Z. Wu, J. Yang, V.A. Nair, D.D. Miller, and J.T. Dalton. 2006. Pharmacokinetics and metabolism of a selective androgen receptor modulator in rats: implication of molecular properties and intensive metabolic profile to investigate ideal pharmacokinetic characteristics of a propanamide in preclinical study. Drug Metabolism and Disposition 34: 483–494.
Yin, D., W. Gao, J.D. Kearbey, H. Xu, K. Chung, Y. He, C.A. Marhefka, K.A. Veverka, D.D. Miller, and J.T. Dalton. 2003a. Pharmacodynamics of selective androgen receptor modulators. Journal of Pharmacology and Experimental Therapeutics 304: 1334–1340.
Yin, D., H. Xu, Y. He, L.I. Kirkovsky, D.D. Miller, and J.T. Dalton. 2003b. Pharmacology, pharmacokinetics, and metabolism of acetothiolutamide, a novel nonsteroidal agonist for the androgen receptor. Journal of Pharmacology and Experimental Therapeutics 304: 1323–1333.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, J., Coss, C.C. & Dalton, J.T. Effect of para halogen modification of S-3-(phenoxy)-2-hydroxy-2-methyl-N-(4-nitro-3-trifluoromethyl-phenyl)-propionamides on metabolism and clearance. Arch. Pharm. Res. 37, 1464–1476 (2014). https://doi.org/10.1007/s12272-013-0258-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-013-0258-4