Abstract
Objective
The present study aimed to investigate the electrophysiological properties of wide dynamic range (WDR) neurons in spinal dorsal horn of rats with neuropathic pain induced by lumber 5 (L5) spinal nerve ligation (SNL) in a large size of samples.
Methods
Adult Sprague-Dawley rats were divided into normal and SNL groups. Electrophysiological technique was used to record the characteristics of WDR neurons in the spinal dorsal horn.
Results
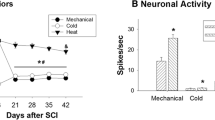
Compared with the WDR neurons in normal rats, the WDR neurons in SNL rats showed an increase in excitability, manifested by an enlargement of the receptive field size, an increase in the proportion of neurons that exhibited spontaneous activities, decreases in the Cresponse threshold and latency, and an increase in the C-response duration. In addition, the numbers of Aβ- and C-fiberevoked discharges were smaller in SNL rats than in normal rats.
Conclusion
The excitability of spinal WDR neurons increased in rats with neuropathic pain induced by L5 SNL. The increase in excitability of WDR neurons may contribute to the development of neuropathic pain.
摘要
目的
探讨腰5 脊神经结扎(spinal nerve ligation, SNL)后, 大鼠脊髓背角的广动力范围(wide dynamic range, WDR)神经元电生理学特性的改变。
方法
将健康雄性Sprague-Dawley 大鼠分为**常组和SNL 组, 利用细胞外电生理学方法记录脊髓背角的WDR 神经元放电。
结果
与**常大鼠相比, SNL 组大鼠WDR 神经元兴奋性增加, 表现为感受野扩大、 有自发放电的神经元比例增加, 以及 C 纤维诱发放电的阈值降低、 潜伏期缩短、 发放时程增加。 此外, SNL 组大鼠WDR 神经元Aβ 和C 纤维诱发放电数目较**常大鼠降低。
结论
大鼠腰5 脊神经结扎后主要引起WDR 神经元的兴奋性增加。 WDR 神经元的兴奋性增加可能参与神经病理痛的发生。
Similar content being viewed by others
References
Latremoliere A, Woolf CJ. Central sensitization: a generator of pain hypersensitivity by central neural plasticity. J Pain 2009, 10: 895–926.
Campbell JN, Meyer RA. Mechanisms of neuropathic pain. Neuron 2006, 52: 77–92.
Chen J. Toward the brain matrix of pain. Neurosci Bull 2009, 25(5): 234–236.
Guan Y, Wacnik PW, Yang F, Carteret AF, Chung CY, Meyer RA, et al. Spinal cord stimulation-induced analgesia: electrical stimulation of dorsal column and dorsal roots attenuates dorsal horn neuronal excitability in neuropathic rats. Anesthesiology 2010, 113: 1392–1405.
Chapman V, Suzuki R, Dickenson AH. Electrophysiological characterization of spinal neuronal response properties in anaesthetized rats after ligation of spinal nerves L5–L6. J Physiol (Lond) 1998, 507: 881–894.
Chu KL, Faltynek CR, Jarvis MF, McGaraughty S. Increased WDR spontaneous activity and receptive field size in rats following a neuropathic or inflammatory injury: implications for mechanical sensitivity. Neurosci Lett 2004, 372: 123–126.
Pertovaara A, Kontinen VK, Kalso EA. Chronic spinal nerve ligation induces changes in response characteristics of nociceptive spinal dorsal horn neurons and in their descending regulation originating in the periaqueductal gray in the rat. Exp Neurol 1997, 147: 428–436.
Suzuki R, Kontinen VK, Matthews E, Williams E, Dickenson AH. Enlargement of the receptive field size to low intensity mechanical stimulation in the rat spinal nerve ligation model of neuropathy. Exp Neurol 2000, 163: 408–413.
Kontinen VK, Dickenson AH. Effects of midazolam in the spinal nerve ligation model of neuropathic pain in rats. Pain 2000, 85: 425–431.
Sokal DM, Chapman V. Effects of spinal administration of muscimol on C- and A-fibre evoked neuronal responses of spinal dorsal horn neurones in control and nerve injured rats. Brain Res 2003, 962: 213–220.
Suzuki R, Rahman W, Hunt SP, Dickenson AH. Descending facilitatory control of mechanically evoked responses is enhanced in deep dorsal horn neurones following peripheral nerve injury. Brain Res 2004, 1019: 68–76.
Zimmermann M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 1983, 16: 109–110.
Geng SJ, Liao FF, Dang WH, Ding X, Liu XD, Cai J, et al. Contribution of the spinal cord BDNF to the development of neuropathic pain by activation of the NR2B-containing NMDA receptors in rats with spinal nerve ligation. Exp Neurol 2010, 222: 256–266.
Jiang YQ, **ng GG, Wang SL, Tu HY, Chi YN, Li J, et al. Axonal accumulation of hyperpolarization-activated cyclic nucleotidegated cation channels contributes to mechanical allodynia after peripheral nerve injury in rat. Pain 2008, 137: 495–506.
Kim SH, Chung JM. An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 1992, 50: 355–363.
Sun Q, **ng GG, Tu HY, Han JS, Wan Y. Inhibition of hyperpolarization-activated current by ZD7288 suppresses ectopic discharges of injured dorsal root ganglion neurons in a rat model of neuropathic pain. Brain Res 2005, 1032: 63–69.
**ng GG, Liu FY, Qu XX, Han JS, Wan Y. Long-term synaptic plasticity in the spinal dorsal horn and its modulation by electroacupuncture in rats with neuropathic pain. Exp Neurol 2007, 208: 323–332.
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 1994, 53: 55–63.
Dixon WJ. Efficient analysis of experimental observations. Annu Rev Pharmacol Toxicol 1980, 20: 441–462.
Liu FY, **ng GG, Qu XX, Xu IS, Han JS, Wan Y. Roles of 5-hydroxytryptamine (5-HT) receptor subtypes in the inhibitory effects of 5-HT on C-fiber responses of spinal wide dynamic range neurons in rats. J Pharmacol Exp Ther 2007, 321: 1046–1053.
Qu XX, Cai J, Li MJ, Chi YN, Liao FF, Liu FY, et al. Role of the spinal cord NR2B-containing NMDA receptors in the development of neuropathic pain. Exp Neurol 2009, 215: 298–307.
Liu FY, Qu XX, Ding X, Cai J, Jiang H, Wan Y, et al. Decrease in the descending inhibitory 5-HT system in rats with spinal nerve ligation. Brain Res 2010, 1330: 45–60.
Kim HK, Kim JH, Gao X, Zhou JL, Lee I, Chung K, et al. Analgesic effect of vitamin E is mediated by reducing central sensitization in neuropathic pain. Pain 2006, 122: 53–62.
Cumberbatch MJ, Carlson E, Wyatt A, Boyce S, Hill RG, Rupniak NM. Reversal of behavioural and electrophysiological correlates of experimental peripheral neuropathy by the NK1 receptor antagonist GR205171 in rats. Neuropharmacology 1998, 37: 1535–1543.
Hu JW, Sessle BJ, Raboisson P, Dallel R, Woda A. Stimulation of craniofacial muscle afferents induces prolonged facilitatory effects in trigeminal nociceptive brain-stem neurones. Pain 1992, 48: 53–60.
Tabo E, **ks SL, Eisele JH Jr, Carstens E. Behavioral manifestations of neuropathic pain and mechanical allodynia, and changes in spinal dorsal horn neurons, following L4-L6 dorsal root constriction in rats. Pain 1999, 80: 503–520.
Schoffnegger D, Ruscheweyh R, Sandkühler J. Spread of excitation across modality borders in spinal dorsal horn of neuropathic rats. Pain 2008, 135: 300–310.
Besse D, Lombard MC, Besson JM. The distribution of mu and delta opioid binding sites belonging to a single cervical dorsal root in the superficial dorsal horn of the rat spinal cord: A quantitative autoradiographic study. Eur J Neurosci 1991, 3: 1343–1352.
Shehab SA. Acute and chronic sectioning of fifth lumbar spinal nerve has equivalent effects on the primary afferents of sciatic nerve in rat spinal cord. J Comp Neurol 2009, 517: 481–492.
D’Mello R, Dickenson AH. Spinal cord mechanisms of pain. Br J Anaesth 2008, 101: 8–16.
Woolf CJ, Shortland P, Coggeshall RE. Peripheral nerve injury triggers central sprouting of myelinated afferents. Nature 1992, 355: 75–78.
Leem JW, Park ES, Paik KS. Electrophysiological evidence for the antinociceptive effect of transcutaneous electrical stimulation on mechanically evoked responsiveness of dorsal horn neurons in neuropathic rats. Neurosci Lett 1995, 192: 197–200.
Sun Q, Tu H, **ng GG, Han JS, Wan Y. Ectopic discharges from injured nerve fibers are highly correlated with tactile allodynia only in early, but not late, stage in rats with spinal nerve ligation. Exp Neurol 2005, 191: 128–136.
Wu G, Ringkamp M, Murinson BB, Pogatzki EM, Hartke TV, Weerahandi HM, et al. Degeneration of myelinated efferent fibers induces spontaneous activity in uninjured C-fiber afferents. J Neurosci 2002, 22: 7746–7753.
Costigan M, Scholz J, Woolf CJ. Neuropathic pain: a maladap tive response of the nervous system to damage. Annu Rev Neurosci 2009, 32: 1–32.
Sotgiu ML, Biella G. Contribution of central sensitization to the pain-related abnormal activity in neuropathic rats. Somatosens Mot Res 2000, 17: 32–38.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, FY., Qu, XX., Cai, J. et al. Electrophysiological properties of spinal wide dynamic range neurons in neuropathic pain rats following spinal nerve ligation. Neurosci. Bull. 27, 1–8 (2011). https://doi.org/10.1007/s12264-011-1039-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-011-1039-z