Abstract
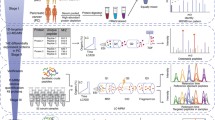
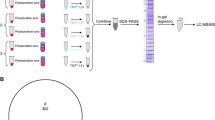
Pancreatic ductal adenocarcinoma (PDAC) is the fourth most frequent cause of cancer mortality in the United States. Because CA 19-9 increases not only in PDAC, but also in benign conditions, there is urgent need for an additional PDAC biomarker. Isotope tags for relative and absolute quantification (iTRAQ) were performed using 6 pairs of PDAC and normal tissues from the same patients, to obtain preliminary PDAC-specific proteins; and verification was performed by multiple reactions monitoring (MRM), using 30 PDAC and 20 normal serum, targeting high-abundant serum proteins without any pre-preparation. As a result, 17 candidate proteins from tissue iTRAQ were verified as potential markers (AUC values > 0.7). Multivariate analysis (MA) demonstrated that a 6-marker panel, consisting of alpha-1 antitrypsin, haptoglobin beta chain, hemopexin, transferrin, zinc alpha-2 glycoprotein, and apolipoprotein A4 from the MRM result, had comparable discriminatory power versus CA 19-9. Our study demonstrated that a combination of iTRAQ on PDAC tissue and verification MRM-MA on individual serum was an efficient method for the development of PDAC multimarkers.
Similar content being viewed by others
References
Jemal, A., R. Siegel, J. Xu, and E. Ward (2010) Cancer statistics. CA Cancer J. Clin. 60: 277–300.
Jung, K. W., S. Park, H. J. Kong, Y. J. Won, Y. K. Boo, H. R. Shin, E. C. Park, and J. S. Lee (2010) Cancer statistics in Korea: Incidence, mortality and survival in 2006–2007. J. Kor. Med. Sci. 25: 1113–1121.
Maitra, A. and R. H. Hruban (2008) Pancreatic cancer. Annu. Rev. Pathol. 3: 157–188.
Ghaneh, P., E. Costello, and J. P. Neoptolemos (2007) Biology and management of pancreatic cancer. Gut. 56: 1134–1152.
Adamek, H. E., J. Albert, H. Breer, M. Weitz, D. Schilling, and J. F. Riemann (2000) Pancreatic cancer detection with magnetic resonance cholangiopancreatography and endoscopic retrograde cholangiopancreatography: A prospective controlled study. Lancet. 356: 190–193.
Porcel, J. M., M. Vives, A. Esquerda, A. Salud, B. Perez, and F. Rodriguez-Panadero (2004) Use of a panel of tumor markers (carcinoembryonic antigen, cancer antigen 125, carbohydrate antigen 15-3, and cytokeratin 19 fragments) in pleural fluid for the differential diagnosis of benign and malignant effusions. Chest. 126: 1757–1763.
Robin, X., N. Turck, A. Hainard, F. Lisacek, J. C. Sanchez, and M. Muller (2009) Bioinformatics for protein biomarker panel classification: What is needed to bring biomarker panels into in vitro diagnostics? Expert. Rev. Proteomics. 6: 675–689.
Ng, I. S., X. S. Zheng, B. Y. Chen, X. Q. Chi, Y. H. Lu, and C. S. Chang (2013) Proteomics approach to decipher novel genes and enzymes characterization of a bioelectricity-generating and dyedecolorizing bacterium Proteus hauseri ZMd44. Biotechnol. Bioproc. Eng. 18: 8–17.
Cheng, J. S., X. M. Lv, and Y. J. Yuan (2012) Investigation of proteomic responses of Streptomyces lydicus to pitching ratios for improving streptolydigin production. Biotechnol. Bioproc. Eng. 17: 997–1007.
Aggarwal, K., L. H. Choe, and K. H. Lee (2005) Quantitative analysis of protein expression using amine-specific isobaric tags in Escherichia coli cells expressing rhsA elements. Proteomics. 5: 2297–2308.
Zhang, Y., A. Wolf-Yadlin, P. L. Ross, D. J. Pappin, J. Rush, D. A. Lauffenburger, and F. M. White (2005) Time-resolved mass spectrometry of tyrosine phosphorylation sites in the epidermal growth factor receptor signaling network reveals dynamic modules. Mol. Cell Proteomics. 4: 1240–1250.
Anderson, L. and C. L. Hunter (2006) Quantitative mass spectrometric multiple reaction monitoring assays for major plasma proteins. Mol. Cell Proteomics. 5: 573–588.
Kiyonami, R., A. Schoen, A. Prakash, S. Peterman, V. Zabrouskov, P. Picotti, R. Aebersold, A. Huhmer, and B. Domon (2011) Increased selectivity, analytical precision, and throughput in targeted proteomics. Mol. Cell Proteomics. 10: 1–11.
Whiteaker, J. R., C. Lin, J. Kennedy, L. Hou, M. Trute, I. Sokal, P. Yan, R. M. Schoenherr, L. Zhao, U. J. Voytovich, K. S. Kelly-Spratt, A. Krasnoselsky, P. R. Gafken, J. M. Hogan, L. A. Jones, P. Wang, L. Amon, L. A. Chodosh, P. S. Nelson, M. W. McIntosh, C. J. Kemp, and A. G. Paulovich (2011) A targeted proteomics-based pipeline for verification of biomarkers in plasma. Nat. Biotechnol. 29: 625–634.
Kim, K., S. J. Kim, H. G. Yu, J. Yu, K. S. Park, I. J. Jang, and Y. Kim (2010) Verification of biomarkers for diabetic retinopathy by multiple reaction monitoring. J. Proteome Res. 9: 689–699.
Chen, R., E. C. Yi, S. Donohoe, S. Pan, J. Eng, K. Cooke, D. A. Crispin, Z. Lane, D. R. Goodlett, M. P. Bronner, R. Aebersold, and T. A. Brentnall (2005) Pancreatic cancer proteome: The proteins that underlie invasion, metastasis, and immunologic escape. Gastroenterol. 129: 1187–1197.
Lu, Z., L. Hu, S. Evers, J. Chen, and Y. Shen (2004) Differential expression profiling of human pancreatic adenocarcinoma and healthy pancreatic tissue. Proteomics. 4: 3975–3988.
Ohuchida, K., K. Mizumoto, N. Ishikawa, K. Fujii, H. Konomi, E. Nagai, K. Yamaguchi, M. Tsuneyoshi, and M. Tanaka (2005) The role of S100A6 in pancreatic cancer development and its clinical implication as a diagnostic marker and therapeutic target. Clin. Cancer Res. 11: 7785–7793.
**, J., Y. W. Kwon, J. S. Paek, H. J. Cho, J. Yu, J. Y. Lee, I. S. Chu, I. H. Park, Y. B. Park, H. S. Kim, and Y. Kim (2011) Analysis of differential proteomes of induced pluripotent stem cells by protein-based reprogramming of fibroblasts. J. Proteome Res. 10: 977–989.
Han, D., S. Moon, H. Kim, S. E. Choi, S. J. Lee, K. S. Park, H. Jun, Y. Kang, and Y. Kim (2011) Detection of differential proteomes associated with the development of type 2 diabetes in the Zucker rat model using the iTRAQ technique. J. Proteome Res. 10: 564–577.
**, J., Y. H. Ku, Y. Kim, K. Kim, J. Y. Lee, Y. M. Cho, H. K. Lee, and K. S. Park (2012) Differential proteome profiling using iTRAQ in microalbuminuric and normoalbuminuric type 2 diabetic patients. Exp. Diabetes Res. 2012: 168602.
Matsubara, J., M. Ono, K. Honda, A. Negishi, H. Ueno, T. Okusaka, J. Furuse, K. Furuta, E. Sugiyama, Y. Saito, N. Kaniwa, J. Sawada, A. Shoji, T. Sakuma, T. Chiba, N. Saijo, S. Hirohashi, and T. Yamada (2010) Survival prediction for pancreatic cancer patients receiving gemcitabine treatment. Mol. Cell Proteomics. 9: 695–704.
Kang, S. M., H. J. Sung, J. M. Ahn, J. Y. Park, S. Y. Lee, C. S. Park, and J. Y. Cho (2011) The Haptoglobin beta chain as a supportive biomarker for human lung cancers. Mol. Biosyst. 7: 1167–1175.
Thompson, S., B. M. Cantwell, C. Cornell, and G. A. Turner (1991) Abnormally-fucosylated haptoglobin: A cancer marker for tumour burden but not gross liver metastasis. Br. J. Cancer. 64: 386–390.
Thompson, S., E. Dargan, and G. A. Turner (1992) Increased fucosylation and other carbohydrate changes in haptoglobin in ovarian cancer. Cancer Lett. 66: 43–48.
Okuyama, N., Y. Ide, M. Nakano, T. Nakagawa, K. Yamanaka, K. Moriwaki, K. Murata, H. Ohigashi, S. Yokoyama, H. Eguchi, O. Ishikawa, T. Ito, M. Kato, A. Kasahara, S. Kawano, J. Gu, N. Taniguchi, and E. Miyoshi (2006) Fucosylated haptoglobin is a novel marker for pancreatic cancer: A detailed analysis of the oligosaccharide structure and a possible mechanism for fucosylation. Int. J. Cancer. 118: 2803–2808.
Wang, Z., C. Wang, X. Huang, Y. Shen, J. Shen, and K. Ying (2012) Differential proteome profiling of pleural effusions from lung cancer and benign inflammatory disease patients. Biochim. Biophys. Acta 1824: 692–700.
Jou, Y. J., C. D. Lin, C. H. Lai, C. H. Chen, J. Y. Kao, S. Y. Chen, M. H. Tsai, S. H. Huang, and C. W. Lin (2010) Proteomic identification of salivary transferrin as a biomarker for early detection of oral cancer. Anal. Chim. Acta 681: 41–48.
Dubois, V., L. Delort, F. Mishellany, T. Jarde, H. Billard, C. Lequeux, O. Damour, F. Penault-Llorca, M. P. Vasson, and F. Caldefie-Chezet (2010) Zinc-alpha2-glycoprotein: A new biomarker of breast cancer? Anticancer Res. 30: 2919–2925.
Dieplinger, H., D. P. Ankerst, A. Burges, M. Lenhard, A. Lingenhel, L. Fineder, H. Buchner, and P. Stieber (2009) Afamin and apolipoprotein A-IV: Novel protein markers for ovarian cancer. Cancer Epidemiol. Biomarkers Prev. 18: 1127–1133.
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yu, J., Kim, K., Kang, M. et al. Development of candidate biomarkers for pancreatic ductal adenocarcinoma using multiple reaction monitoring. Biotechnol Bioproc E 18, 1038–1047 (2013). https://doi.org/10.1007/s12257-013-0421-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-013-0421-2