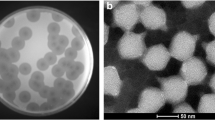
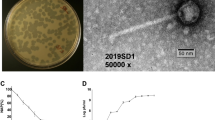
Abstract
The increasing emergence of multi-drug resistant Escherichia coli (E. coli) has become a global concern, primarily due to the limitation of antimicrobial treatment options. Phage therapy has been considered as a promising alternative for treating infections caused by multi-drug resistant E. coli. However, the application of phages as a promising antimicrobial agent is limited by their narrow host range and specificity. In this research, a recombinant T4-like phage, named WGqlae, has been obtained by changing the receptor specificity determinant region of gene 37, using a homologous recombination platform of T4-like phages established by our laboratory previously. The engineered phage WGqlae can lyse four additional hosts, comparing to its parental phages WG01 and QL01. WGqlae showed similar characteristics, including thermo and pH stability, optimal multiplicity of infection and one-step growth curve, to the donor phage QL01. In addition, sequencing results showed that gene 37 of recombinant phage WGqlae had genetically stable even after 20 generations. In planktonic test, phage WGqlae had significant antimicrobial effects on E. coli DE192 and DE205B. The optical density at 600 nm (OD600) of E. coli in phage WGqlae treating group was significantly lower than that of the control group (P < 0.01). Besides, phage WGqlae demonstrated an obvious inhibitory effect on the biofilm formation and the clearance of mature biofilms. Our study suggested that engineered phages may be promising candidates for future phage therapy applications against pathogenic E. coli in planktonic and biofilm forms.







Similar content being viewed by others
References
Adams M (1959) Bacteriophage. Interscience Publishers, New York
Bartual SG, Otero JM, Garcia-Doval C, Llamas-Saiz AL, Kahn R, Fox GC, van Raaij MJ (2010) Structure of the bacteriophage T4 long tail fiber receptor-binding tip. Proc Natl Acad Sci U S A 107:20287–20292
Chan BK, Abedon ST, Loc-Carrillo C (2013) Phage cocktails and the future of phage therapy. Future Microbiol 8:769–783
Chan BK, Turner PE, Kim S, Mojibian HR, Elefteriades JA, Narayan D (2018) Phage treatment of an aortic graft infected with Pseudomonas aeruginosa. Evol Med Public Health 2018:60–66
Chen M, Xu J, Yao H, Lu C, Zhang W (2016) Isolation, genome sequencing and functional analysis of two T7-like coliphages of avian pathogenic Escherichia coli. Gene 582:47–58
Chen M, Zhang L, Abdelgader SA, Yu L, Xu J, Yao H, Lu C, Zhang W (2017) Alterations in gp37 expand the host range of a T4-like phage. Appl Environ Microbiol. https://doi.org/10.1128/AEM.01576-17
Chibani-Chennoufi S, Sidoti J, Bruttin A, Kutter E, Sarker S, Brussow H (2004) in vitro and in vivo bacteriolytic activities of Escherichia coli phages: implications for phage therapy. Antimicrob Agents Chemother 48:2558–2569
Clark JR, March JB (2006) Bacteriophages and biotechnology: vaccines, gene therapy and antibacterials. Trends Biotechnol 24:212–218
Dedrick RM, Guerrero-Bustamante CA, Garlena RA, Russell DA, Ford K, Harris K, Gilmour KC, Soothill J, Jacobs-Sera D, Schooley RT, Hatfull GF, Spencer H (2019) Engineered bacteriophages for treatment of a patient with a disseminated drug-resistant Mycobacterium abscessus. Nat Med 25:730–733
Diarra MS, Malouin F (2014) Antibiotics in Canadian poultry productions and anticipated alternatives. Front Microbiol 5:282
Ebrahimi-Nik H, Bassami MR, Mohri M, Rad M, Khan MI (2018) Bacterial ghost of avian pathogenic E. coli (APEC) serotype O78:K80 as a homologous vaccine against avian colibacillosis. PLoS ONE 13:888
Endersen L, Buttimer C, Nevin E, Coffey A, Neve H, Oliveira H, Lavigne R, O’Mahony J (2017) Investigating the biocontrol and anti-biofilm potential of a three phage cocktail against Cronobacter sakazakii in different brands of infant formula. Int J Food Microbiol 253:1–11
Haq IU, Chaudhry WN, Akhtar MN, Andleeb S, Qadri I (2012) Bacteriophages and their implications on future biotechnology: a review. Virol J 9:9
Hashemolhosseini S, Montag D, Kramer L, Henning U (1994) Determinants of receptor specificity of coliphages of the T4 family. A chaperone alters the host range. J Mol Biol 241:524–533
Hermoso JA, Garcia JL, Garcia P (2007) Taking aim on bacterial pathogens: from phage therapy to enzybiotics. Curr Opin Microbiol 10:461–472
Hughes G, Webber MA (2017) Novel approaches to the treatment of bacterial biofilm infections. Br J Pharmacol 174:2237–2246
Ibrahim RA, Cryer TL, Lafi SQ, Basha EA, Good L, Tarazi YH (2019) Identification of Escherichia coli from broiler chickens in Jordan, their antimicrobial resistance, gene characterization and the associated risk factors. BMC Vet Res 15:159
Jenkins C (2018) Enteroaggregative Escherichia coli. Curr Top Microbiol Immunol 416:27–50
Johnson RP, Gyles CL, Huff WE, Ojha S, Huff GR, Rath NC, Donoghue AM (2008) Bacteriophages for prophylaxis and therapy in cattle, poultry and pigs. Anim Health Res Rev 9:201–215
Kaistha SD, Umrao PD (2016) Bacteriophage for mitigation of multiple drug resistant biofilm forming pathogens. Recent Pat Biotechnol 10:184–194
Kaper JB, Nataro JP, Mobley HL (2004) Pathogenic Escherichia coli. Nat Rev Microbiol 2:123–140
Kellenberger E, Bolle A, Boydelatour E, Epstein RH, Franklin NC, Jerne NK, Reale Scafati A, Sechaud J (1965) Functions and properties related to the tail fibers of bacteriophage T4. Virology 26:419–440
Kelly D, McAuliffe O, Ross RP, Coffey A (2012) Prevention of Staphylococcus aureus biofilm formation and reduction in established biofilm density using a combination of phage K and modified derivatives. Lett Appl Microbiol 54:286–291
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Li Y, Chen L, Wu X, Huo S (2015) Molecular characterization of multidrug-resistant avian pathogenic Escherichia coli isolated from septicemic broilers. Poult Sci 94:601–611
Liu C, Diao Y, Wang D, Chen H, Tang Y, Diao Y (2019) Duck viral infection escalated the incidence of avian pathogenic Escherichia coli in China. Transbound Emerg Dis 66:929–938
Lu Z, Breidt F Jr, Fleming HP, Altermann E, Klaenhammer TR (2003) Isolation and characterization of a Lactobacillus plantarum bacteriophage, phiJL-1, from a cucumber fermentation. Int J Food Microbiol 84:225–235
Lutful Kabir SM (2010) Avian colibacillosis and salmonellosis: a closer look at epidemiology, pathogenesis, diagnosis, control and public health concerns. Int J Environ Res Public Health 7:89–114
Mahichi F, Synnott AJ, Yamamichi K, Osada T, Tanji Y (2009) Site-specific recombination of T2 phage using IP008 long tail fiber genes provides a targeted method for expanding host range while retaining lytic activity. FEMS Microbiol Lett 295:211–217
Moulin-Schouleur M, Reperant M, Laurent S, Bree A, Mignon-Grasteau S, Germon P, Rasschaert D, Schouler C (2007) Extraintestinal pathogenic Escherichia coli strains of avian and human origin: link between phylogenetic relationships and common virulence patterns. J Clin Microbiol 45:3366–3376
Nesta B, Pizza M (2018) Vaccines against Escherichia coli. Curr Top Microbiol Immunol 416:213–242
Pajunen M, Kiljunen S, Skurnik M (2000) Bacteriophage phiYeO3-12, specific for Yersinia enterocolitica serotype O:3, is related to coliphages T3 and T7. J Bacteriol 182:5114–5120
Rios AC, Moutinho CG, Pinto FC, Del Fiol FS, Jozala A, Chaud MV, Vila MM, Teixeira JA, Balcao VM (2016) Alternatives to overcoming bacterial resistances: state-of-the-art. Microbiol Res 191:51–80
Schooley RT, Biswas B, Gill JJ, Hernandez-Morales A, Lancaster J, Lessor L, Barr JJ, Reed SL, Rohwer F, Benler S, Segall AM, Taplitz R, Smith DM, Kerr K, Kumaraswamy M, Nizet V, Lin L, McCauley MD, Strathdee SA, Benson CA, Pope RK, Leroux BM, Picel AC, Mateczun AJ, Cilwa KE, Regeimbal JM, Estrella LA, Wolfe DM, Henry MS, Quinones J, Salka S, Bishop-Lilly KA, Young R, Hamilton T (2017) Development and use of personalized bacteriophage-based therapeutic cocktails to treat a patient with a disseminated resistant Acinetobacter baumannii infection. Antimicrob Agents Chemother. https://doi.org/10.1128/AAC.00954-17
Stepanovic S, Vukovic D, Dakic I, Savic B, Svabic-Vlahovic M (2000) A modified microtiter-plate test for quantification of staphylococcal biofilm formation. J Microbiol Methods 40:175–179
Stepanovic S, Cirkovic I, Ranin L, Svabic-Vlahovic M (2004) Biofilm formation by Salmonella spp. and Listeria monocytogenes on plastic surface. Lett Appl Microbiol 38:428–432
Suresh G, Das RK, Kaur Brar S, Rouissi T, Avalos Ramirez A, Chorfi Y, Godbout S (2018) Alternatives to antibiotics in poultry feed: molecular perspectives. Crit Rev Microbiol 44:318–335
Tetart F, Repoila F, Monod C, Krisch HM (1996) Bacteriophage T4 host range is expanded by duplications of a small domain of the tail fiber adhesin. J Mol Biol 258:726–731
Tetart F, Desplats C, Krisch HM (1998) Genome plasticity in the distal tail fiber locus of the T-even bacteriophage: recombination between conserved motifs swaps adhesin specificity. J Mol Biol 282:543–556
Wais AC, Goldberg EB (1969) Growth and transformation of phage T4 in Escherichia coli B-4, Salmonella, Aerobacter, Proteus, and Serratia. Virology 39:153–161
Yoichi M, Abe M, Miyanaga K, Unno H, Tanji Y (2005) Alteration of tail fiber protein gp38 enables T2 phage to infect Escherichia coli O157:H7. J Biotechnol 115:101–107
Yu L, Wang S, Guo Z, Liu H, Sun D, Yan G, Hu D, Du C, Feng X, Han W, Gu J, Sun C, Lei L (2018) A guard-killer phage cocktail effectively lyses the host and inhibits the development of phage-resistant strains of Escherichia coli. Appl Microbiol Biotechnol 102:971–983
Acknowledgements
This study was supported by Grants from the National Natural Science Foundation of China (U1803109), Key research and development plan of Jiangsu province (BE2019304), National Key R&D Program of China (2018YFC1602500), the Central University Basic Scientific Research Fund-Animal pathogenic bacteria (KYZ201846) and Jiangsu modern agriculture (waterfowl) industrial technology system disease prevention and control innovation team (JATS[2018]222).
Author information
Authors and Affiliations
Contributions
WZ and HD conceived of the study. ML, DS, YL, YX, MC performed experiment, computational analysis. WZ and ML wrote the paper. ML and LC revised the final manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Animal and Human Rights Statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, M., Shi, D., Li, Y. et al. Recombination of T4-like Phages and Its Activity against Pathogenic Escherichia coli in Planktonic and Biofilm Forms. Virol. Sin. 35, 651–661 (2020). https://doi.org/10.1007/s12250-020-00233-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12250-020-00233-2