Abstract
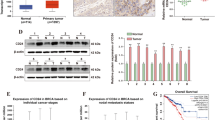
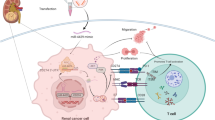
Lysine demethylase 5A (KDM5A) is a histone demethylase frequently involved in cancer progression. This research aimed to explore the function of KDM5A in prostate adenocarcinoma (PRAD) and the molecular mechanism. KDM5A was highly expressed in collected PRAD tissues and acquired PRAD cells. High KDM5A expression was correlated with reduced survival and poor prognosis of patients with PRAD. Knockdown of KDM5A suppressed the proliferation, colony formation, migration, and invasiveness of PRAD cells and reduced angiogenesis ability of endothelial cells. Downstream molecules implicated in KDM5A mediation were predicted using integrated bioinformatic analyses. KDM5A enhanced ETS proto-oncogene 1 (ETS1) expression through demethylation of H3K4me2 at its promoter. ETS1 suppressed the transcription activity of miR-330-3p, and either further ETS1 overexpression or miR-330-3p inhibition blocked the functions of KDM5A knockdown in PRAD. miR-330-3p targeted coatomer protein complex subunit β2 (COPB2) mRNA. Downregulation of miR-330-3p restored the expression of COPB2 and activated the PI3K/AKT pathway in PRAD. The results in vitro were reproduced in vivo where KDM5A downregulation suppressed the growth and metastasis of xenograft tumors in nude mice. In conclusion, this study demonstrated that KDM5A promoted PRAD by suppressing miR-330-3p and activating the COPB2/PI3K/AKT axis in an ETS1-dependent manner.









Similar content being viewed by others
Data availability
The datasets used and analyzed in the current study are available from the corresponding author upon request.
References
Adams BD, Kasinski AL, Slack FJ (2014) Aberrant regulation and function of microRNAs in cancer. Curr Biol 24:R762-776. https://doi.org/10.1016/j.cub.2014.06.043
An C, Li H, Zhang X, Wang J, Qiang Y, Ye X, Li Q, Guan Q, Zhou Y (2019) Silencing of COPB2 inhibits the proliferation of gastric cancer cells and induces apoptosis via suppression of the RTK signaling pathway. Int J Oncol 54:1195–1208. https://doi.org/10.3892/ijo.2019.4717
Bhandari A, Zheng C, Sindan N, Sindan N, Quan R, **a E, Thapa Y, Tamang D, Wang O, Ye X, Huang D (2019) COPB2 is up-regulated in breast cancer and plays a vital role in the metastasis via N-cadherin and Vimentin. J Cell Mol Med 23:5235–5245. https://doi.org/10.1111/jcmm.14398
Blair LP, Cao J, Zou MR, Sayegh J, Yan Q (2011) Epigenetic Regulation by Lysine Demethylase 5 (KDM5) enzymes in cancer. Cancers (basel) 3:1383–1404. https://doi.org/10.3390/cancers3011383
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424. https://doi.org/10.3322/caac.21492
Chen H, Zhou L, Wu X, Li R, Wen J, Sha J, Wen X (2016) The PI3K/AKT pathway in the pathogenesis of prostate cancer. Front Biosci (landmark Ed) 21:1084–1091. https://doi.org/10.2741/4443
Chen T, Yang Z, Liu C, Wang L, Yang J, Chen L, Li W (2019) Circ_0078767 suppresses non-small-cell lung cancer by protecting RASSF1A expression via sponging miR-330-3p. Cell Prolif 52:e12548. https://doi.org/10.1111/cpr.12548
Chen X, Xu M, Xu X, Zeng K, Liu X, Pan B, Li C, Sun L, Qin J, Xu T, He B, Pan Y, Sun H, Wang S (2020) METTL14-mediated N6-methyladenosine modification of SOX4 mRNA inhibits tumor metastasis in colorectal cancer. Mol Cancer 19:106. https://doi.org/10.1186/s12943-020-01220-7
Chou NH, Lo YH, Wang KC, Kang CH, Tsai CY, Tsai KW (2018) MiR-193a-5p and -3p play a distinct role in gastric cancer: miR-193a-3p suppresses gastric cancer cell growth by targeting ETS1 and CCND1. Anticancer Res 38:3309–3318. https://doi.org/10.21873/anticanres.12596
Cui J, Quan M, **e D, Gao Y, Guha S, Fallon MB, Chen J, **e K (2020) A novel KDM5A/MPC-1 signaling pathway promotes pancreatic cancer progression via redirecting mitochondrial pyruvate metabolism. Oncogene 39:1140–1151. https://doi.org/10.1038/s41388-019-1051-8
Culp MB, Soerjomataram I, Efstathiou JA, Bray F, Jemal A (2020) Recent global patterns in prostate cancer incidence and mortality rates. Eur Urol 77:38–52. https://doi.org/10.1016/j.eururo.2019.08.005
Dai B, Huang H, Guan F, Zhu G, **ao Z, Mao B, Su H, Hu Z (2018) Histone demethylase KDM5A inhibits glioma cells migration and invasion by down regulating ZEB1. Biomed Pharmacother 99:72–80. https://doi.org/10.1016/j.biopha.2018.01.020
Dittmer J (2015) The role of the transcription factor Ets1 in carcinoma. Semin Cancer Biol 35:20–38. https://doi.org/10.1016/j.semcancer.2015.09.010
Du C, Lv C, Feng Y, Yu S (2020) Activation of the KDM5A/miRNA-495/YTHDF2/m6A-MOB3B axis facilitates prostate cancer progression. J Exp Clin Cancer Res 39:223. https://doi.org/10.1186/s13046-020-01735-3
Foroozan M, Roudi R, Abolhasani M, Gheytanchi E, Mehrazma M (2017) Clinical significance of endothelial cell marker CD34 and mast cell marker CD117 in prostate adenocarcinoma. Pathol Res Pract 213:612–618. https://doi.org/10.1016/j.prp.2017.04.027
Ghafouri-Fard S, Shoorei H, Taheri M (2020) Role of microRNAs in the development, prognosis and therapeutic response of patients with prostate cancer. Gene 759:144995. https://doi.org/10.1016/j.gene.2020.144995
Harris TA, Yamakuchi M, Kondo M, Oettgen P, Lowenstein CJ (2010) Ets-1 and Ets-2 regulate the expression of microRNA-126 in endothelial cells. Arterioscler Thromb Vasc Biol 30:1990–1997. https://doi.org/10.1161/ATVBAHA.110.211706
Hong Z, Wu G, **ang ZD, Xu CD, Huang SS, Li C, Shi L, Wu DL (2019) KDM5C is transcriptionally regulated by BRD4 and promotes castration-resistance prostate cancer cell proliferation by repressing PTEN. Biomed Pharmacother 114:108793. https://doi.org/10.1016/j.biopha.2019.108793
Huang Y, Sun H, Ma X, Zeng Y, Pan Y, Yu D, Liu Z, **ang Y (2020) HLA-F-AS1/miR-330-3p/PFN1 axis promotes colorectal cancer progression. Life Sci 254:117180. https://doi.org/10.1016/j.lfs.2019.117180
Itoh T, Takeda S, Akao Y (2010) MicroRNA-208 modulates BMP-2-stimulated mouse preosteoblast differentiation by directly targeting V-ets erythroblastosis virus E26 oncogene homolog 1. J Biol Chem 285:27745–27752. https://doi.org/10.1074/jbc.M110.105080
Janevska S, Guldener U, Sulyok M, Tudzynski B, Studt L (2018) Set1 and Kdm5 are antagonists for H3K4 methylation and regulators of the major conidiation-specific transcription factor gene ABA1 in Fusarium fujikuroi. Environ Microbiol 20:3343–3362. https://doi.org/10.1111/1462-2920.14339
** Z, Jia B, Tan L, Liu Y (2019) miR-330-3p suppresses liver cancer cell migration by targeting MAP2K1. Oncol Lett 18:314–320. https://doi.org/10.3892/ol.2019.10280
Kang AD, Cosenza SC, Bonagura M, Manair M, Reddy MV, Reddy EP (2013) ON01210.Na (Ex-RAD(R)) mitigates radiation damage through activation of the AKT pathway. PLoS ONE. https://doi.org/10.1371/journal.pone.0058355
Kar A, Gutierrez-Hartmann A (2013) Molecular mechanisms of ETS transcription factor-mediated tumorigenesis. Crit Rev Biochem Mol Biol 48:522–543. https://doi.org/10.3109/10409238.2013.838202
Kato M, Dang V, Wang M, Park JT, Deshpande S, Kadam S, Mardiros A, Zhan Y, Oettgen P, Putta S, Yuan H, Lanting L, Natarajan R (2013) TGF-beta induces acetylation of chromatin and of Ets-1 to alleviate repression of miR-192 in diabetic nephropathy. Sci Signal. https://doi.org/10.1126/scisignal.2003389
Kern HB, Niemeyer BF, Parrish JK, Kerr CA, Yaghi NK, Prescott JD, Gutierrez-Hartmann A, Jedlicka P (2012) Control of MicroRNA-21 expression in colorectal cancer cells by oncogenic epidermal growth factor/Ras signaling and Ets transcription factors. DNA Cell Biol 31:1403–1411. https://doi.org/10.1089/dna.2011.1469
Kfir-Elirachman K, Ortenberg R, Vizel B, Besser MJ, Barshack I, Schachter J, Nemlich Y, Markel G (2018) Regulation of CEACAM1 protein expression by the transcription factor ETS-1 in BRAF-mutant human metastatic melanoma cells. Neoplasia 20:401–409. https://doi.org/10.1016/j.neo.2018.01.012
Khan MI, Hamid A, Rath S, Ateeq B, Khan Q, Siddiqui IA, Adhami VM, Choudhry H, Zamzami MA, Mukhtar H (2019) AKT inhibition modulates H3K4 demethylase levels in PTEN-null prostate cancer. Mol Cancer Ther 18:356–363. https://doi.org/10.1158/1535-7163.MCT-18-0141
Kirtana R, Manna S, Patra SK (2020) Molecular mechanisms of KDM5A in cellular functions: Facets during development and disease. Exp Cell Res 396:112314. https://doi.org/10.1016/j.yexcr.2020.112314
Komura K, Yoshikawa Y, Shimamura T, Chakraborty G, Gerke TA, Hinohara K, Chadalavada K, Jeong SH, Armenia J, Du SY, Mazzu YZ, Taniguchi K, Ibuki N, Meyer CA, Nanjangud GJ, Inamoto T, Lee GM, Mucci LA, Azuma H, Sweeney CJ, Kantoff PW (2018) ATR inhibition controls aggressive prostate tumors deficient in Y-linked histone demethylase KDM5D. J Clin Invest 128:2979–2995. https://doi.org/10.1172/JCI96769
Kouzarides T (2007) Chromatin modifications and their function. Cell 128:693–705. https://doi.org/10.1016/j.cell.2007.02.005
Lee CG, Kwon HK, Sahoo A, Hwang W, So JS, Hwang JS, Chae CS, Kim GC, Kim JE, So HS, Hwang ES, Grenningloh R, Ho IC, Im SH (2012) Interaction of Ets-1 with HDAC1 represses IL-10 expression in Th1 cells. J Immunol 188:2244–2253. https://doi.org/10.4049/jimmunol.1101614
Li N, Dhar SS, Chen TY, Kan PY, Wei Y, Kim JH, Chan CH, Lin HK, Hung MC, Lee MG (2016) JARID1D is a suppressor and prognostic marker of prostate cancer invasion and metastasis. Cancer Res 76:831–843. https://doi.org/10.1158/0008-5472.CAN-15-0906
Li G, Kanagasabai T, Lu W, Zou MR, Zhang SM, Celada SI, Izban MG, Liu Q, Lu T, Ballard BR, Zhou X, Adunyah SE, Matusik RJ, Yan Q, Chen Z (2020a) KDM5B is essential for the hyperactivation of PI3K/AKT signaling in prostate tumorigenesis. Cancer Res 80:4633–4643. https://doi.org/10.1158/0008-5472.CAN-20-0505
Li Q, Wang W, Zhang M, Sun W, Shi W, Li F (2020b) Circular RNA circ-0016068 promotes the growth, migration, and invasion of prostate cancer cells by regulating the miR-330-3p/BMI-1 axis as a competing endogenous RNA. Front Cell Dev Biol 8:827. https://doi.org/10.3389/fcell.2020.00827
Li H, Ma Z, Lei Y (2021) The expression of kappa-opioid receptor promotes the migration of breast cancer cells in vitro. BMC Anesthesiol 21:210. https://doi.org/10.1186/s12871-021-01429-z
Liu Y, Liu K, Yin L, Yu Y, Qi J, Shen WH, Zhu J, Zhang Y, Dong A (2019) H3K4me2 functions as a repressive epigenetic mark in plants. Epigenetics Chromatin 12:40. https://doi.org/10.1186/s13072-019-0285-6
Luo Y, Liu F, Yan C, Qu W, Zhu L, Guo Z, Zhou F, Zhang W (2020) Long non-coding RNA CASC19 sponges microRNA-532 and promotes oncogenicity of clear cell renal cell carcinoma by increasing ETS1 expression. Cancer Manag Res 12:2195–2207. https://doi.org/10.2147/CMAR.S242472
Ma B, Ma J, Yang Y, He X, Pan X, Wang Z, Qian Y (2020) Effects of miR-330-3p on invasion, migration and EMT of gastric cancer cells by targeting PRRX1-mediated Wnt/beta-catenin signaling pathway. Onco Targets Ther 13:3411–3423. https://doi.org/10.2147/OTT.S238665
Mi Y, Yu M, Zhang L, Sun C, Wei B, Ding W, Zhu Y, Tang J, **a G, Zhu L (2016) COPB2 is upregulated in prostate cancer and regulates PC-3 cell proliferation, cell cycle, and apoptosis. Arch Med Res 47:411–418. https://doi.org/10.1016/j.arcmed.2016.09.005
Nagarajan P, Chin SS, Wang D, Liu S, Sinha S, Garrett-Sinha LA (2010) Ets1 blocks terminal differentiation of keratinocytes and induces expression of matrix metalloproteases and innate immune mediators. J Cell Sci 123:3566–3575. https://doi.org/10.1242/jcs.062240
Nazir SU, Kumar R, Singh A, Khan A, Tanwar P, Tripathi R, Mehrotra R, Hussain S (2019) Breast cancer invasion and progression by MMP-9 through Ets-1 transcription factor. Gene 711:143952. https://doi.org/10.1016/j.gene.2019.143952
Oser MG, Sabet AH, Gao W, Chakraborty AA, Schinzel AC, Jennings RB, Fonseca R, Bonal DM, Booker MA, Flaifel A, Novak JS, Christensen CL, Zhang H, Herbert ZT, Tolstorukov MY, Buss EJ, Wong KK, Bronson RT, Nguyen QD, Signoretti S, Kaelin WG Jr (2019) The KDM5A/RBP2 histone demethylase represses NOTCH signaling to sustain neuroendocrine differentiation and promote small cell lung cancer tumorigenesis. Genes Dev 33:1718–1738. https://doi.org/10.1101/gad.328336.119
Petronikolou N, Longbotham JE, Fujimori DG (2020) Extended recognition of the histone H3 tail by histone demethylase KDM5A. Biochemistry 59:647–651. https://doi.org/10.1021/acs.biochem.9b01036
Ren F, Shrestha C, Shi H, Sun F, Zhang M, Cao Y, Li G (2020) Targeting of KDM5A by miR-421 in human ovarian cancer suppresses the progression of ovarian cancer cells. Onco Targets Ther 13:9419–9428. https://doi.org/10.2147/OTT.S266211
Rodgers JJ, McClure R, Epis MR, Cohen RJ, Leedman PJ, Harvey JM, Australian Prostate Cancer B, Thomas MA, Bentel JM (2019) ETS1 induces transforming growth factor beta signaling and promotes epithelial-to-mesenchymal transition in prostate cancer cells. J Cell Biochem 120:848–860. https://doi.org/10.1002/jcb.27446
Sechler M, Parrish JK, Birks DK, Jedlicka P (2017) The histone demethylase KDM3A, and its downstream target MCAM, promote Ewing Sarcoma cell migration and metastasis. Oncogene 36:4150–4160. https://doi.org/10.1038/onc.2017.44
Shen MM, Abate-Shen C (2010) Molecular genetics of prostate cancer: new prospects for old challenges. Genes Dev 24:1967–2000. https://doi.org/10.1101/gad.1965810
Shen L, Yi S, Huang L, Li S, Bai F, Lei S, Breitzig M, Czachor A, Sun H, Zheng Q, Wang F (2019) miR-330-3p promotes lung cancer cells invasion, migration, and metastasis by directly targeting hSOD2b. Biotechnol Appl Biochem 66:21–32. https://doi.org/10.1002/bab.1691
Shokri G, Doudi S, Fathi-Roudsari M, Kouhkan F, Sanati MH (2018) Targeting histone demethylases KDM5A and KDM5B in AML cancer cells: A comparative view. Leuk Res 68:105–111. https://doi.org/10.1016/j.leukres.2018.02.003
Singh SK, Apata T, Gordetsky JB, Singh R (2019) Docetaxel combined with thymoquinone induces apoptosis in prostate cancer cells via inhibition of the PI3K/AKT signaling pathway. Cancers (basel). https://doi.org/10.3390/cancers11091390
Sobral LM, Hicks HM, Parrish JK, McCann TS, Hsieh J, Goodspeed A, Costello JC, Black JC, Jedlicka P (2020) KDM3A/Ets1 epigenetic axis contributes to PAX3/FOXO1-driven and independent disease-promoting gene expression in fusion-positive Rhabdomyosarcoma. Mol Oncol 14:2471–2486. https://doi.org/10.1002/1878-0261.12769
Taylor MA, Wappett M, Delpuech O, Brown H, Chresta CM (2016) Enhanced MAPK signaling drives ETS1-mediated induction of miR-29b leading to downregulation of TET1 and changes in epigenetic modifications in a subset of lung SCC. Oncogene 35:4345–4357. https://doi.org/10.1038/onc.2015.499
Toren P, Zoubeidi A (2014) Targeting the PI3K/Akt pathway in prostate cancer: challenges and opportunities (review). Int J Oncol 45:1793–1801. https://doi.org/10.3892/ijo.2014.2601
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108. https://doi.org/10.3322/caac.21262
Vieira FQ, Costa-Pinheiro P, Ramalho-Carvalho J, Pereira A, Menezes FD, Antunes L, Carneiro I, Oliveira J, Henrique R, Jeronimo C (2014) Deregulated expression of selected histone methylases and demethylases in prostate carcinoma. Endocr Relat Cancer 21:51–61. https://doi.org/10.1530/ERC-13-0375
Voichek Y, Mittelman K, Gordon Y, Bar-Ziv R, Lifshitz Smit D, Shenhav R, Barkai N (2018) Epigenetic control of expression homeostasis during replication is stabilized by the replication checkpoint. Mol Cell. https://doi.org/10.1016/j.molcel.2018.05.015
Wang Z, Qu H, Gong W, Liu A (2018) Up-regulation and tumor-promoting role of SPHK1 were attenuated by miR-330-3p in gastric cancer. IUBMB Life 70:1164–1176. https://doi.org/10.1002/iub.1934
Wang X, Shi J, Niu Z, Wang J, Zhang W (2020) MiR-216a-3p regulates the proliferation, apoptosis, migration, and invasion of lung cancer cells via targeting COPB2. Biosci Biotechnol Biochem 84:2014–2027. https://doi.org/10.1080/09168451.2020.1783197
Ward RD, Purysko AS (2020) Magnetic resonance imaging of prostate adenocarcinoma: detection and staging. Top Magn Reson Imaging 29:17–30. https://doi.org/10.1097/RMR.0000000000000226
**ang Y, Zhu Z, Han G, Ye X, Xu B, Peng Z, Ma Y, Yu Y, Lin H, Chen AP, Chen CD (2007) JARID1B is a histone H3 lysine 4 demethylase up-regulated in prostate cancer. Proc Natl Acad Sci U S A 104:19226–19231. https://doi.org/10.1073/pnas.0700735104
**ong X, Shi Q, Yang X, Wang W, Tao J (2019) LINC00052 functions as a tumor suppressor through negatively modulating miR-330-3p in pancreatic cancer. J Cell Physiol 234:15619–15626. https://doi.org/10.1002/jcp.28209
Xu S, Ge J, Zhang Z, Zhou W (2017) MiR-129 inhibits cell proliferation and metastasis by targeting ETS1 via PI3K/AKT/mTOR pathway in prostate cancer. Biomed Pharmacother 96:634–641. https://doi.org/10.1016/j.biopha.2017.10.037
Yalim-Camci I, Balcik-Ercin P, Cetin M, Odabas G, Tokay N, Sayan AE, Yagci T (2019) ETS1 is coexpressed with ZEB2 and mediates ZEB2-induced epithelial-mesenchymal transition in human tumors. Mol Carcinog 58:1068–1081. https://doi.org/10.1002/mc.22994
Yan Y, Huang H (2019) Interplay Among PI3K/AKT, PTEN/FOXO and AR Signaling in Prostate Cancer. Adv Exp Med Biol 1210:319–331. https://doi.org/10.1007/978-3-030-32656-2_14
Yang GJ, Wang W, Mok SWF, Wu C, Law BYK, Miao XM, Wu KJ, Zhong HJ, Wong CY, Wong VKW, Ma DL, Leung CH (2018) Selective Inhibition of Lysine-Specific Demethylase 5A (KDM5A) using a Rhodium(III) complex for triple-negative breast cancer therapy. Angew Chem Int Ed Engl 57:13091–13095. https://doi.org/10.1002/anie.201807305
Yang GJ, Ko CN, Zhong HJ, Leung CH, Ma DL (2019a) Structure-based discovery of a selective KDM5A inhibitor that exhibits anti-cancer activity via inducing cell cycle arrest and senescence in breast cancer cell lines. Cancers (basel). https://doi.org/10.3390/cancers11010092
Yang YB, Tan H, Wang Q (2019b) MiRNA-300 suppresses proliferation, migration and invasion of non-small cell lung cancer via targeting ETS1. Eur Rev Med Pharmacol Sci 23:10827–10834. https://doi.org/10.26355/eurrev_201912_19786
Acknowledgements
This work was supported by National Natural Science Foundation (No. 81802576, 81902565, 81372316), Wuxi City Medical Young Talent (No. QNRC043), Wuxi Commission of Health and Family Planning (No. T202102, T202024, J202012, Z202011, CXTDPY2021003), the Science and Technology Development Fund of Wuxi (No. WX18IIAN024, N20202021), Jiangnan University Wuxi School of Medicine (No. 1286010242190070), Wuxi “Taihu Talent Program”-High-end Talent in Medical and Healthentalent plan of Taihu Lake in Wuxi (Double Hundred Medical Youth Professionals Program) from Health Committee of Wuxi (No. BJ2020061), clinical trial of Affiliated Hospital of Jiangnan University (No. LCYJ202227). The authors are thankful for the guidance of Professor Yongquan Chen.
Author information
Authors and Affiliations
Contributions
YYM, LFZ, CYS and YYF conceived the design of the research and carried out most of the experiments. JS, JW, DJY, XWQ, HYW, SW, GWX and LJZ obtained, analyzed and interpreted the data. YYM, LFZ, CYS and YYF wrote and revised the manuscript. SW and GWX prepared the fgures and provides financial support for all experiments. All authors read and approved the fnal manuscript. We confirmed that no third parties were used in either the writing of the paper or the generation of data.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
This research was ratified by the Ethics Committee of Affiliated Hospital of Jiangnan University and performed in line with the Declaration of Helsinki. Written informed consent was obtained from each eligible respondent. All animal experiments were ratified by the Ethics Committee of Affiliated Hospital of Jiangnan University and conducted in line with the Guide for the Care and Use of Laboratory Animals issued by the National Institutes of Health (NIH, Bethesda, Maryland, USA). Significant efforts were made to minimize the suffering of animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mi, Y., Zhang, L., Sun, C. et al. Lysine demethylase 5A promotes prostate adenocarcinoma progression by suppressing microRNA-330-3p expression and activating the COPB2/PI3K/AKT axis in an ETS1-dependent manner. J. Cell Commun. Signal. 16, 579–599 (2022). https://doi.org/10.1007/s12079-022-00671-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-022-00671-5