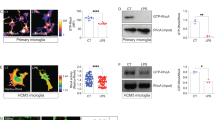
Abstract
Microglia are immunocompetent cells in the central nervous system. Following cerebral ischemia, microglia will be rapidly activated and undergo proliferation, morphological transformation, and changes in gene expression and function. At present, the regulatory mechanisms of microglial activation following ischemia remain largely unclear. In this study, we took advantage of CX3CR1GFP/+ fluorescent mice and a global cerebral ischemia-reperfusion model to investigate the mechanisms of microglial activation following different degrees of global ischemia. Our results showed that the proliferation of microglia was gated by the degree of ischemia. Marked microglial de-ramification and proliferation were observed after 60 min of ischemia but not in transient ischemia (20 min). Immunohistology, qRT-PCR, and Western blotting analysis showed that microglial activation was accompanied with a reduction in Wnt/β-catenin signaling after cerebral ischemia. Downregulation of Wnt/β-catenin signaling using Wnt antagonist XAV939 during 20 min ischemia promoted microglial de-ramification and proliferation. In contrast, enhancing Wnt/β-catenin signaling using Wnt agonist LiCl during 60 min ischemia-reduced microglial de-ramification and proliferation. Importantly, we found that Wnt agonist inhibited inflammation in the ischemic brain and was conducive to animal behavioral recovery. Collectively, these data demonstrated that Wnt/β-catenin signaling played a key role in microglial activation following cerebral ischemia, and regulating microglial activation may be a potential therapeutic strategy for the treatment of ischemic stroke.
Graphical Abstract









Similar content being viewed by others
Data Availability
The data supporting the conclusions of this article are available from the corresponding author upon reasonable request.
Abbreviations
- CNS:
-
central nervous system
- JAK:
-
Janus-activated kinase
- STAT:
-
signal transducer and activator of transcription
- IFN-γ:
-
interferon-γ
- TLR:
-
toll-like receptors
- CSF1R:
-
colony-stimulating factor 1 receptor
- Lef1:
-
lymphoid enhancer-binding factor 1
- Ctnnb1:
-
catenin beta 1
- TCF7L2:
-
transcription factor 7-like 2
- Fzd9:
-
frizzled 9
- TCF1:
-
T cell factor 1
- Arg1:
-
arginase-1
- TGFβ:
-
transforming growth factor beta
- IL-1β:
-
interleukin 1-beta
- IL-4:
-
interleukin 4
- IL-6:
-
interleukin 6
- iNOS:
-
inducible nitric oxide synthase
- BCAL:
-
bilateral common carotid artery ligation
- BCCA:
-
bilateral common carotid arteries
- CBF:
-
cerebral blood flow
- LSI:
-
laser speckle contrast imaging
- FBS:
-
fetal bovine serum
- PBS:
-
phosphate-buffered saline
- OGD/R:
-
oxygen-glucose deprivation and reoxygenation
- DMSO:
-
dimethyl sulfoxide
- PFA:
-
paraformaldehyde
- BrdU:
-
bromodeoxyuridine
- MCAO:
-
middle cerebral occlusion
- RT-qPCR:
-
quantitative real-time polymerase chain reaction
- GFP:
-
green fluorescent protein
- ROI:
-
region of interest
- PU.1:
-
Spi-1 proto-oncogene
- Irf8:
-
interferon regulatory factor 8
References
Askew K, Li K, Olmos-Alonso A, Garcia-Moreno F, Liang Y, Richardson P et al (2017) Coupled proliferation and apoptosis maintain the rapid turnover of microglia in the adult brain. Cell Rep 18(2):391–405
Huang Y, Xu Z, **ong S, Sun F, Qin G, Hu G et al (2018) Repopulated microglia are solely derived from the proliferation of residual microglia after acute depletion. Nat Neurosci 21(4):530–540
Zhan L, Krabbe G, Du F, Jones I, Reichert MC, Telpoukhovskaia M et al (2019) Proximal recolonization by self-renewing microglia re-establishes microglial homeostasis in the adult mouse brain. PLoS Biol 17(2):e3000134
Nimmerjahn A, Kirchhoff F, Helmchen F (2005) Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308(5726):1314–1318
Roth TL, Nayak D, Atanasijevic T, Koretsky AP, Latour LL, McGavern DB (2014) Transcranial amelioration of inflammation and cell death after brain injury. Nature 505(7482):223–228
Hanisch UK, Kettenmann H (2007) Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci 10(11):1387–1394
Kettenmann H, Hanisch UK, Noda M, Verkhratsky A (2011) Physiology of microglia. Physiol Rev 91(2):461–553
Zhang S (2019) Microglial activation after ischaemic stroke. Stroke Vasc Neurol 4(2):71–74
Ma Y, Wang J, Wang Y, Yang GY (2017) The biphasic function of microglia in ischemic stroke. Prog Neurobiol 157:247–272
Paolicelli RC, Sierra A, Stevens B, Tremblay ME, Aguzzi A, Ajami B et al (2022) Microglia states and nomenclature: a field at its crossroads. Neuron 110(21):3458–3483
Gao H, Ju F, Ti R, Zhang Y, Zhang S (2022) Differential regulation of microglial activation in response to different degree of ischemia. Front Immunol 13:792638
Li T, Pang S, Yu Y, Wu X, Guo J, Zhang S (2013) Proliferation of parenchymal microglia is the main source of microgliosis after ischaemic stroke. Brain 136(Pt 12):3578–3588
Morrison HW, Filosa JA (2013) A quantitative spatiotemporal analysis of microglia morphology during ischemic stroke and reperfusion. J Neuroinflammation 10:4
Otxoa-de-Amezaga A, Miro-Mur F, Pedragosa J, Gallizioli M, Justicia C, Gaja-Capdevila N et al (2019) Microglial cell loss after ischemic stroke favors brain neutrophil accumulation. Acta Neuropathol 137(2):321–341
Zhou K, Chen J, Wu J, Wu Q, Jia C, Xu YXZ et al (2019) Atractylenolide III ameliorates cerebral ischemic injury and neuroinflammation associated with inhibiting JAK2/STAT3/Drp1-dependent mitochondrial fission in microglia. Phytomedicine 59:152922
Tang Z, Gan Y, Liu Q, Yin JX, Liu Q, Shi J et al (2014) CX3CR1 deficiency suppresses activation and neurotoxicity of microglia/macrophage in experimental ischemic stroke. J Neuroinflammation 11:26
Sawano T, Watanabe F, Ishiguchi M, Doe N, Furuyama T, Inagaki S (2015) Effect of Sema4D on microglial function in middle cerebral artery occlusion mice. Glia 63(12):2249–2259
Perego C, Fumagalli S, De Simoni MG (2011) Temporal pattern of expression and colocalization of microglia/macrophage phenotype markers following brain ischemic injury in mice. J Neuroinflammation 8:174
Ito U, Nagasao J, Kawakami E, Oyanagi K (2007) Fate of disseminated dead neurons in the cortical ischemic penumbra: ultrastructure indicating a novel scavenger mechanism of microglia and astrocytes. Stroke 38(9):2577–2583
Zhang W, Zhao J, Wang R, Jiang M, Ye Q, Smith AD et al (2019) Macrophages reprogram after ischemic stroke and promote efferocytosis and inflammation resolution in the mouse brain. CNS Neurosci Ther 25(12):1329–1342
Carbonell WS, Murase S, Horwitz AF, Mandell JW (2005) Migration of perilesional microglia after focal brain injury and modulation by CC chemokine receptor 5: an in situ time-lapse confocal imaging study. J Neurosci 25(30):7040–7047
Yenari MA, Kauppinen TM, Swanson RA (2010) Microglial activation in stroke: therapeutic targets. Neurotherapeutics 7(4):378–391
Rajan WD, Wojtas B, Gielniewski B, Gieryng A, Zawadzka M, Kaminska B (2019) Dissecting functional phenotypes of microglia and macrophages in the rat brain after transient cerebral ischemia. Glia 67(2):232–245
Song D, Zhang X, Chen J, Liu X, Xue J, Zhang L et al (2019) Wnt canonical pathway activator TWS119 drives microglial anti-inflammatory activation and facilitates neurological recovery following experimental stroke. J Neuroinflammation 16(1):256
Liu X, Zhang X, Chen J, Song D, Zhang C, Chen R et al (2022) Chrysophanol facilitates long-term neurological recovery through limiting microglia-mediated neuroinflammation after ischemic stroke in mice. Int Immunopharmacol 112:109220
Kong L, Li W, Chang E, Wang W, Shen N, Xu X et al (2022) mtDNA-STING axis mediates microglial polarization via IRF3/NF-kappaB signaling after ischemic stroke. Front Immunol 13:860977
Rivest S (2009) Regulation of innate immune responses in the brain. Nat Rev Immunol 9(6):429–439
Miron VE, Boyd A, Zhao JW, Yuen TJ, Ruckh JM, Shadrach JL et al (2013) M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nat Neurosci 16(9):1211–1218
Zhang J, He H, Qiao Y, Zhou T, He H, Yi S et al (2020) Priming of microglia with IFN-gamma impairs adult hippocampal neurogenesis and leads to depression-like behaviors and cognitive defects. Glia 68(12):2674–2692
Reusch N, Ravichandran KA, Olabiyi BF, Komorowska-Muller JA, Hansen JN, Ulas T et al (2022) Cannabinoid receptor 2 is necessary to induce toll-like receptor-mediated microglial activation. Glia 70(1):71–88
Squillace S, Salvemini D (2022) Toll-like receptor-mediated neuroinflammation: relevance for cognitive dysfunctions. Trends Pharmacol Sci 43(9):726–739
Elmore MR, Najafi AR, Koike MA, Dagher NN, Spangenberg EE, Rice RA et al (2014) Colony-stimulating factor 1 receptor signaling is necessary for microglia viability, unmasking a microglia progenitor cell in the adult brain. Neuron 82(2):380–397
Rice RA, Spangenberg EE, Yamate-Morgan H, Lee RJ, Arora RP, Hernandez MX et al (2015) Elimination of microglia improves functional outcomes following extensive neuronal loss in the hippocampus. J Neurosci 35(27):9977–9989
Spangenberg EE, Lee RJ, Najafi AR, Rice RA, Elmore MR, Blurton-Jones M et al (2016) Eliminating microglia in Alzheimer’s mice prevents neuronal loss without modulating amyloid-beta pathology. Brain 139(Pt 4):1265–1281
Otero K, Turnbull IR, Poliani PL, Vermi W, Cerutti E, Aoshi T et al (2009) Macrophage colony-stimulating factor induces the proliferation and survival of macrophages via a pathway involving DAP12 and beta-catenin. Nat Immunol 10(7):734–743
Marchetti B, Pluchino S (2013) Wnt your brain be inflamed? Yes, it Wnt! Trends Mol Med 19(3):144–156
Van Steenwinckel J, Schang AL, Krishnan ML, Degos V, Delahaye-Duriez A, Bokobza C et al (2019) Decreased microglial Wnt/beta-catenin signalling drives microglial pro-inflammatory activation in the develo** brain. Brain 142(12):3806–3833
Halleskog C, Mulder J, Dahlstrom J, Mackie K, Hortobagyi T, Tanila H et al (2011) WNT signaling in activated microglia is proinflammatory. Glia 59(1):119–131
Zhu L, Wang L, Ju F, Ran Y, Wang C, Zhang S (2017) Transient global cerebral ischemia induces rapid and sustained reorganization of synaptic structures. J Cereb Blood Flow Metab 37(8):2756–2767
Ju F, Ran Y, Zhu L, Cheng X, Gao H, ** X et al (2018) Increased BBB permeability enhances activation of microglia and exacerbates loss of dendritic spines after transient global cerebral ischemia. Front Cell Neurosci 12:236
Saura J, Tusell JM, Serratosa J (2003) High-yield isolation of murine microglia by mild trypsinization. Glia 44(3):183–189
**a Q, Li X, Zhou H, Zheng L, Shi J (2018) S100A11 protects against neuronal cell apoptosis induced by cerebral ischemia via inhibiting the nuclear translocation of annexin A1. Cell Death Dis 9(6):657
Zhu L, Wang L, Ju F, Khan A, Cheng X, Zhang S (2017) Reversible recovery of neuronal structures depends on the degree of neuronal damage after global cerebral ischemia in mice. Exp Neurol 289:1–8
Li T, Zhao J, **e W, Yuan W, Guo J, Pang S et al (2021) Specific depletion of resident microglia in the early stage of stroke reduces cerebral ischemic damage. J Neuroinflammation 18(1):81
Weber BZC, Agca S, Domaniku A, Bilgic SN, Arabaci DH, Kir S (2022) Inhibition of epidermal growth factor receptor suppresses parathyroid hormone-related protein expression in tumours and ameliorates cancer-associated cachexia. J Cachexia Sarcopenia Muscle 13(3):1582–1594
Srivastava P, Cronin CG, Scranton VL, Jacobson KA, Liang BT, Verma R (2020) Neuroprotective and neuro-rehabilitative effects of acute purinergic receptor P2X4 (P2X4R) blockade after ischemic stroke. Exp Neurol 329:113308
Heneka MT, Kummer MP, Latz E (2014) Innate immune activation in neurodegenerative disease. Nat Rev Immunol 14(7):463–477
Wolf SA, Boddeke HW, Kettenmann H (2017) Microglia in physiology and disease. Annu Rev Physiol 79:619–643
Ji YB, Gao Q, Tan XX, Huang XW, Ma YZ, Fang C et al (2021) Lithium alleviates blood-brain barrier breakdown after cerebral ischemia and reperfusion by upregulating endothelial Wnt/beta-catenin signaling in mice. Neuropharmacology 186:108474
Cheng J, Shen W, ** L, Pan J, Zhou Y, Pan G et al (2020) Treadmill exercise promotes neurogenesis and myelin repair via upregulating Wnt/betacatenin signaling pathways in the juvenile brain following focal cerebral ischemia/reperfusion. Int J Mol Med 45(5):1447–1463
Colonna M, Butovsky O (2017) Microglia function in the central nervous system during health and neurodegeneration. Annu Rev Immunol 35:441–468
Lee TK, Kim H, Song M, Lee JC, Park JH, Ahn JH et al (2019) Time-course pattern of neuronal loss and gliosis in gerbil hippocampi following mild, severe, or lethal transient global cerebral ischemia. Neural Regen Res 14(8):1394–1403
Park CW, Lee JC, Ahn JH, Lee DH, Cho GS, Yan BC et al (2013) Neuronal damage using fluoro-Jade B histofluorescence and gliosis in the gerbil septum submitted to various durations of cerebral ischemia. Cell Mol Neurobiol 33(7):991–1001
Cotrina ML, Lou N, Tome-Garcia J, Goldman J, Nedergaard M (2017) Direct comparison of microglial dynamics and inflammatory profile in photothrombotic and arterial occlusion evoked stroke. Neuroscience 343:483–494
Wahul AB, Joshi PC, Kumar A, Chakravarty S (2018) Transient global cerebral ischemia differentially affects cortex, striatum and hippocampus in bilateral common carotid arterial occlusion (BCCAo) mouse model. J Chem Neuroanat 92:1–15
Kierdorf K, Erny D, Goldmann T, Sander V, Schulz C, Perdiguero EG et al (2013) Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nat Neurosci 16(3):273–280
Olmos-Alonso A, Schetters ST, Sri S, Askew K, Mancuso R, Vargas-Caballero M et al (2016) Pharmacological targeting of CSF1R inhibits microglial proliferation and prevents the progression of Alzheimer's-like pathology. Brain 139(Pt 3):891–907
Burguillos MA, Svensson M, Schulte T, Boza-Serrano A, Garcia-Quintanilla A, Kavanagh E et al (2015) Microglia-secreted galectin-3 acts as a toll-like receptor 4 ligand and contributes to microglial activation. Cell Rep 10(9):1626–1638
Rahimian R, Beland LC, Sato S, Kriz J (2021) Microglia-derived galectin-3 in neuroinflammation; a bittersweet ligand? Med Res Rev 41(4):2582–2589
Zheng H, Jia L, Liu CC, Rong Z, Zhong L, Yang L et al (2017) TREM2 promotes microglial survival by activating Wnt/beta-catenin pathway. J Neurosci 37(7):1772–1784
Young KF, Gardner R, Sariana V, Whitman SA, Bartlett MJ, Falk T et al (2021) Can quantifying morphology and TMEM119 expression distinguish between microglia and infiltrating macrophages after ischemic stroke and reperfusion in male and female mice? J Neuroinflammation 18(1):58
Zhang L, Wang Y, **ao F, Wang S, **ng G, Li Y et al (2014) CKIP-1 regulates macrophage proliferation by inhibiting TRAF6-mediated Akt activation. Cell Res 24(6):742–761
Gomez-Nicola D, Fransen NL, Suzzi S, Perry VH (2013) Regulation of microglial proliferation during chronic neurodegeneration. J Neurosci 33(6):2481–2493
Zhou Y, Zhang L, Hao Y, Yang L, Fan S, **ao Z (2022) FKN/CX3CR1 axis facilitates migraine-Like behaviour by activating thalamic-cortical network microglia in status epilepticus model rats. J Headache Pain 23(1):42
Song J, Han K, Wang Y, Qu R, Liu Y, Wang S et al (2022) Microglial activation and oxidative stress in PM(2.5)-induced neurodegenerative disorders. Antioxidants (Basel) 11(8):1482
Kim RE, Shin CY, Han SH, Kwon KJ (2020) Astaxanthin suppresses PM2.5-induced neuroinflammation by regulating Akt phosphorylation in BV-2 microglial cells. Int J Mol Sci 21(19):7227
Zhao R, Hu W, Tsai J, Li W, Gan WB (2017) Microglia limit the expansion of beta-amyloid plaques in a mouse model of Alzheimer’s disease. Mol Neurodegener 12(1):47
Willis EF, MacDonald KPA, Nguyen QH, Garrido AL, Gillespie ER, Harley SBR et al (2020) Repopulating microglia promote brain repair in an IL-6-dependent manner. Cell 180(5):833–46 e16
Jolivel V, Bicker F, Biname F, Ploen R, Keller S, Gollan R et al (2015) Perivascular microglia promote blood vessel disintegration in the ischemic penumbra. Acta Neuropathol 129(2):279–295
da Fonseca AC, Matias D, Garcia C, Amaral R, Geraldo LH, Freitas C et al (2014) The impact of microglial activation on blood-brain barrier in brain diseases. Front Cell Neurosci 8:362
Morimoto M, Nakano T, Egashira S, Irie K, Matsuyama K, Wada M et al (2022) Haptoglobin regulates macrophage/microglia-induced inflammation and prevents ischemic brain damage via binding to HMGB1. J Am Heart Assoc 11(6):e024424
Zhang L, Wei W, Ai X, Kilic E, Hermann DM, Venkataramani V et al (2021) Extracellular vesicles from hypoxia-preconditioned microglia promote angiogenesis and repress apoptosis in stroke mice via the TGF-beta/Smad2/3 pathway. Cell Death Dis 12(11):1068
Szalay G, Martinecz B, Lenart N, Kornyei Z, Orsolits B, Judak L et al (2016) Microglia protect against brain injury and their selective elimination dysregulates neuronal network activity after stroke. Nat Commun 7:11499
Chen B, Zhang M, Ji M, Zhang D, Chen B, Gong W et al (2022) The neuroprotective mechanism of lithium after ischaemic stroke. Commun Biol 5(1):105
Acknowledgements
The authors thank Lei Wang, Liang Peng, and Li** Guan at the Core Facility of School of Life Sciences, Lanzhou University, for excellent technical assistance.
Funding
This study was supported by the National Natural Science Foundation of China (No. 82271402), the Natural Science Foundation of Gansu Province (No. 22JR5RA437), and the Excellent Graduate Student “Innovation Star” Program of Education Department of Gansu Province (No. 2021CXZX-027).
Author information
Authors and Affiliations
Contributions
JRL and SXZ designed the research plan; JRL, XYZ, and YYX performed the experiments; JRL, XYZ, and YYX analyzed the data; JRL and SXZ wrote the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All experimental procedures in this study complied with the institutional guidelines from the Ethics Committee of Lanzhou University, China.
Consent to Publication
All authors have read and approved the submitted manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 2757 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, J., Zhang, X., Xu, Y. et al. Regulation of Microglial Activation by Wnt/β-Catenin Signaling After Global Cerebral Ischemia in Mice. Mol Neurobiol 61, 308–325 (2024). https://doi.org/10.1007/s12035-023-03557-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03557-8