Abstract
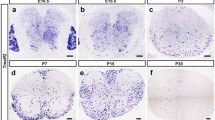
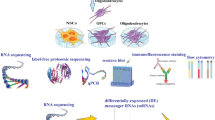
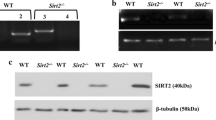
Myelin, a lipid-enriched multi-layer membrane structure, allows for rapid long-distance saltatory conduction of neuronal impulses. Although glycolipids are the predominant types of lipids in the myelin bilayer, the role of glycolipid transfer protein (GLTP), which selectively mediates the transfer of various glycolipids between phospholipid bilayer, in myelin development and maintenance remains unknown at present. In this study, we identified Gltp as the key lipid metabolism gene in myelin-forming oligodendrocytes (OLs) through integrated omics analysis across independent transcriptomic and single-cell sequencing studies. Gene expression analysis revealed that Gltp is selectively expressed in the differentiated OLs. Functional study demonstrated that its expression is essential for the differentiation of OLs, and promotes the outgrowth of OL membrane. Moreover, we found that the expression of Gltp is regulated by OL-lineage transcriptional factors, such as NKX2.2, OLIG2, SOX10, and MYRF. These findings provide important insights into the unrecognized functions of Gltp in OL differentiation and maturation.








Similar content being viewed by others
Data Availability
The datasets supporting the conclusions of this article are included within the article and its supplementary files.
References
Metz RJ, Radin NS (1980) Glucosylceramide uptake protein from spleen cytosol. J Biol Chem 255(10):4463–4467
Abe A, Yamada K, Sasaki T (1982) A protein purified from pig brain accelerates the inter-membranous translocation of mono- and dihexosylceramides, but not the translocation of phospholipids. Biochem Biophys Res Commun 104(4):1386–1393
Yamada K, Sasaki T (1982) Rat liver glycolipid transfer protein. A protein which facilitates the translocation of mono- and dihexosylceramides from donor to acceptor liposomes. J Biochem 92(2):457–64
Li XM et al (2004) Human glycolipid transfer protein: probing conformation using fluorescence spectroscopy. Biochemistry 43(31):10285–10294
West G et al (2008) Identification of a glycosphingolipid transfer protein GLTP1 in Arabidopsis thaliana. FEBS J 275(13):3421–3437
Saupe S et al (1994) Inactivation of the Podospora anserina vegetative incompatibility locus het-c, whose product resembles a glycolipid transfer protein, drastically impairs ascospore production. Proc Natl Acad Sci U S A 91(13):5927–5931
Tuuf J, Mattjus P (2007) Human glycolipid transfer protein–intracellular localization and effects on the sphingolipid synthesis. Biochim Biophys Acta 1771(11):1353–1363
Mattjus P (2009) Glycolipid transfer proteins and membrane interaction. Biochim Biophys Acta 1788(1):267–272
Yamada K, Abe A, Sasaki T (1985) Specificity of the glycolipid transfer protein from pig brain. J Biol Chem 260(8):4615–4621
Ochoa-Lizarralde B et al (2018) Structural analyses of 4-phosphate adaptor protein 2 yield mechanistic insights into sphingolipid recognition by the glycolipid transfer protein family. J Biol Chem 293(43):16709–16723
Yoshihara T et al (2018) Lactosylceramide synthases encoded by B4galt5 and 6 genes are pivotal for neuronal generation and myelin formation in mice. PLoS Genet 14(8):e1007545
Meixner M et al (2011) Myelination in the absence of UDP-galactose:ceramide galactosyl-transferase and fatty acid 2 -hydroxylase. BMC Neurosci 12:22
Kjellberg MA et al (2014) Alternation in the glycolipid transfer protein expression causes changes in the cellular lipidome. PLoS ONE 9(5):e97263
Nakai Y et al (2000) Lysenin-sphingomyelin binding at the surface of oligodendrocyte lineage cells increases during differentiation in vitro. J Neurosci Res 62(4):521–529
Bradl M, Lassmann H (2010) Oligodendrocytes: biology and pathology. Acta Neuropathol 119(1):37–53
Hardy R, Reynolds R (1991) Proliferation and differentiation potential of rat forebrain oligodendroglial progenitors both in vitro and in vivo. Development 111(4):1061–1080
Bergles DE, Richardson WD (2015) Oligodendrocyte development and plasticity. Cold Spring Harb Perspect Biol 8(2):a020453
Nave KA, Werner HB (2014) Myelination of the nervous system: mechanisms and functions. Annu Rev Cell Dev Biol 30:503–533
Norton WT, Poduslo SE (1973) Myelination in rat brain: changes in myelin composition during brain maturation. J Neurochem 21(4):759–773
Coetzee T et al (1996) Myelination in the absence of galactocerebroside and sulfatide: normal structure with abnormal function and regional instability. Cell 86(2):209–219
Saravanan K et al (2004) Specific downregulation and mistargeting of the lipid raft-associated protein MAL in a glycolipid storage disorder. Neurobiol Dis 16(2):396–406
Mastracci TL, Lin CS, Sussel L (2013) Generation of mice encoding a conditional allele of Nkx2.2. Transgenic Res 22(5):965–72
Cai J et al (1999) Expression and regulation of the chicken Nkx-6.2 homeobox gene suggest its possible involvement in the ventral neural patterning and cell fate specification. Dev Dyn 216(4–5):459–68
Zhu Q et al (2011) Dorsally-derived oligodendrocytes in the spinal cord contribute to axonal myelination during development and remyelination following focal demyelination. Glia 59(11):1612–1621
Huang H et al (2018) Interactive repression of MYRF self-cleavage and activity in oligodendrocyte differentiation by TMEM98 protein. J Neurosci 38(46):9829–9839
Sharma R, Sekhon S, Cascella M (2021) White matter lesions. In: StatPearls. StatPearls Publishing, Treasure Island. https://www.ncbi.nlm.nih.gov/books/NBK562167. Accessed 5 Sep 2022
Zhu Q et al (2014) Genetic evidence that Nkx2.2 and Pdgfra are major determinants of the timing of oligodendrocyte differentiation in the develo** CNS. Development 141(3):548–55
Zou X et al (2011) Human glycolipid transfer protein gene (GLTP) expression is regulated by Sp1 and Sp3: involvement of the bioactive sphingolipid ceramide. J Biol Chem 286(2):1301–1311
Henson J, Saffer J, Furneaux H (1992) The transcription factor Sp1 binds to the JC virus promoter and is selectively expressed in glial cells in human brain. Ann Neurol 32(1):72–77
Funding
This work was supported by the Natural Science Foundation of Zhejiang Province (grants LQ23C090003) and the National Natural Science Foundation of China (grants 32170969).
Author information
Authors and Affiliations
Contributions
Z.CF, CZ, and TZ performed the experiments; Z. CF and TZ designed the study; Z. CF and TZ wrote the manuscript; Q. MS, W. XP, and ZD revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All institutional and national guidelines for the care and use of laboratory animals were followed during the experiments. All procedures performed in this study observed the ethical standards of the Second **angya Hospital of Central South University and Hangzhou Normal University.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, C., Chen, Z., Zhang, D. et al. Role of Gltp in Maturation of Oligodendrocytes Under the Regulation of Nkx2.2. Mol Neurobiol 60, 4897–4908 (2023). https://doi.org/10.1007/s12035-023-03383-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03383-y