Abstract
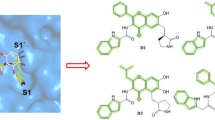
Severe Acute Respiratory Syndrome caused by a coronavirus is a recent viral infection. There is no scientific evidence or clinical trials to indicate that possible therapies have demonstrated results in suspected or confirmed patients. This work aims to perform a virtual screening of 1430 ligands through molecular docking and to evaluate the possible inhibitory capacity of these drugs about the Mpro protease of Covid-19. The selected drugs were registered with the FDA and available in the virtual drug library, widely used by the population. The simulation was performed using the MolAiCalD algorithm, with a Lamarckian genetic model (GA) combined with energy estimation based on rigid and flexible conformation grids. In addition, molecular dynamics studies were also performed to verify the stability of the receptor-ligand complexes formed through analyses of RMSD, RMSF, H–Bond, SASA, and MMGBSA. Compared to the binding energy of the synthetic redocking coupling (−6.8 kcal/mol/RMSD of 1.34 Å), which was considerably higher, it was then decided to analyze the parameters of only three ligands: ergotamine (−9.9 kcal/mol/RMSD of 2.0 Å), dihydroergotamine (−9.8 kcal/mol/RMSD of 1.46 Å) and olysio (−9.5 kcal/mol/RMSD of 1.5 Å). It can be stated that ergotamine showed the best interactions with the Mpro protease of Covid-19 in the in silico study, showing itself as a promising candidate for treating Covid-19.













Similar content being viewed by others
Data availability
Not applicable
References
Guo, Y. R., Cao, Q. D., Hong, Z. S., Tan, Y. Y., Chen, S. D., **, H. J., Sen, T. K., Wang, D. Y., & Yan, Y. (2020). The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak- An update on the status. Military Medical Research, 7, 1–10. https://doi.org/10.1186/s40779-020-00240-0
Martins-Filho, Ricardo P., Gois-Santos, Tavares V., Tavares, C. S. S., de Melo, E. G. M., Nascimento-Júnior, E. M., & do, Santos VS,. (2020). Recommendations for a safety dental care management during SARS-CoV-2 pandemic. Revista Panamericana de Salud Pública, 44, 1–3. https://doi.org/10.26633/rpsp.2020.51
Fehr, A. R., & Perlman, S. (2015). Coronaviruses: An overview of their replication and pathogenesis. Coronaviruses: Methods and protocols (1st ed., pp. 1–23). Springer.
Rothan, H. A., & Byrareddy, S. N. (2020). The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. Journal of Autoimmunity, 109, 1–4. https://doi.org/10.1016/j.jaut.2020.102433
Bogoch, I. I., Watts, A., Thomas-Bachli, A., Huber, C., Kraemer, M. U. G., & Khan, K. (2020). Pneumonia of unknown aetiology in Wuhan, China: Potential for international spread via commercial air travel. Journal of Travel Medicine, 27, 1–3. https://doi.org/10.1093/jtm/taaa008
Lu, H., Stratton, C. W., & Tang, Y. W. (2020). Outbreak of pneumonia of unknown etiology in Wuhan, China: The mystery and the miracle. Journal of Medical Virology, 92, 401–402. https://doi.org/10.1002/jmv.25678
Zhou, T., Liu, Q., Yang, Z., Liao, J., Yang, K., Bai, W., Lu, X., & Zhang, W. (2020). Preliminary prediction of the basic reproduction number of the Wuhan novel coronavirus 2019-nCoV. Journal of Evidence-Based Medicine, 13, 3–7. https://doi.org/10.1111/jebm.12376
Wu, D., Wu, T., Liu, Q., & Yang, Z. (2020). The SARS-CoV-2 outbreak: What we know. International Journal of Infectious Diseases, 94, 44–48. https://doi.org/10.1016/j.ijid.2020.03.004
Zumla, A., Hui, D. S., Azhar, E. I., Memish, Z. A., & Maeurer, M. (2020). Reducing mortality from 2019-nCoV: Host-directed therapies should be an option. The Lancet, 395, 22–28. https://doi.org/10.1016/S0140-6736(20)30305-6
Wang, M., Cao, R., Zhang, L., Yang, X., Liu, J., Xu, M., Shi, Z., Hu, Z., Zhong, W., & **ao, G. (2020). Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Research, 30, 269–271. https://doi.org/10.1038/s41422-020-0282-0
Falavigna, M., Colpani, V., Stein, C., Azevedo, L. C., Bagattini, A. M., & Brito, G. V. (2020). Diretrizes para o tratamento farmacológico da COVID-19. Revista Brasileira de Terapia Intensiva, 32, 166–196.
Mahdi, M. A., Yousefi, S. R., Jasim, L. S., & Salavati-Niasari, M. (2022). Green synthesis of DyBa2Fe3O7.988/DyFeO3 nanocomposites using almond extract with dual eco-friendly applications: Photocatalytic and antibacterial activities. International Journal of Hydrogen Energy, 47, 14319–14330. https://doi.org/10.1016/j.ijhydene.2022.02.175
Yousefi, S. R., Ghanbari, M., Amiri, O., Marzhoseyni, Z., Mehdizadeh, P., Hajizadeh-Oghaz, M., & Salavati-Niasari, M. (2021). Dy2BaCuO5/Ba4DyCu3O9.09 S-scheme heterojunction nanocomposite with enhanced photocatalytic and antibacterial activities. Journal of the American Ceramic Society, 104, 2952–2965. https://doi.org/10.1111/jace.17696
Yousefi, S. R., Sobhani, A., Alshamsi, H. A., & Salavati-Niasari, M. (2021). Green sonochemical synthesis of BaDy2NiO5/Dy2O3 and BaDy2NiO5/NiO nanocomposites in the presence of core almond as a cap** agent and their application as photocatalysts for the removal of organic dyes in water. RSC Advances, 11, 11500–11512. https://doi.org/10.1039/d0ra10288a
Ledford, H. (2020). Dozens of coronavirus drugs are in development: What happens next? Nature, 581, 247–248. https://doi.org/10.1038/d41586-020-01367-9
Menezes, C. R., Sanches, C., & Chequer, F. M. D. (2020). Efetividade e toxicidade da cloroquina e da hidroxicloroquina associada (ou não) à azitromicina para tratamento da COVID-19. O que sabemos até o momento? Journal of Health & Biological Sciences, 8, 1–9. https://doi.org/10.12662/2317-3076jhbs.v8i1.3206.p1-9.2020
Dourado, P., Porto, M., Dal Pizzol, T., Ramos, L., Serrate, S., Luiza, V., Leão, N., Rocha, M., Oliveira, M., Dâmaso, A., Da, T., Dal, S., Ii, P., Ramos, R., Serrate, S., Iv, M., Luiza, V. L., Urruth, N., Tavares, L., … Viii, B. (2016). Prevalence of self-medication in Brazil and associated factors. Revista de Saude Publica, 50, 1s–11s. https://doi.org/10.1590/S1518-8787.2016050006117
de Correia, B., & C, Trindade JK, Almeida AB,. (2019). Fatores Correlacionados À Automedicação Entre Os Jovens E Adultos- Uma Revisão Integrativa Da Literatura. Rev Iniciação Científica e Extensão, 2, 57–61.
Hahn, K. L. (2011). Old drugs are new again. Pharmacy Times, 77, 159–166.
Aronson, J. K. (2007). Old drugs: New uses. British Journal of Clinical Pharmacology, 64, 563–565. https://doi.org/10.1111/j.1365-2125.2007.03058.x
Chong, C. R., & Sullivan, D. J. (2007). New uses for old drugs. Nature, 448, 645–646. https://doi.org/10.1038/448645a
Sliwoski, G., Kothiwale, S., Meiler, J., & Lowe, E. W. (2014). Computational methods in drug discovery. Pharmacological Reviews, 66, 334–395.
Aparoy, P., Kumar Reddy, K., & Reddanna, P. (2012). Structure and ligand based drug design strategies in the development of novel 5- LOX inhibitors. Current Medicinal Chemistry, 19, 3763–3778. https://doi.org/10.2174/092986712801661112
Guido, R. V. C., & Andricopulo, A. D. (2008). Modelagem Molecular de Fármacos. Revista Processos Químicos, 2, 24–36. https://doi.org/10.19142/rpq.v2i4.66
Coutinho, J. P. (2013). Busca De Novos Fármacos Para O Tratamento Da Malária Humana Através De Diferentes Abordagens. Journal of Chemical Information and Modeling. https://doi.org/10.1017/CBO9781107415324.004
Trott, O., & Olson, A. J. (2010). AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. Journal of Computational Chemistry, 31, 455–461. https://doi.org/10.1002/jcc.21334
Bai, Q., Tan, S., Xu, T., Liu, H., Huang, J., & Yao, X. (2021). MolAICal: A soft tool for 3D drug design of protein targets by artificial intelligence and classical algorithm. Briefings in Bioinformatics, 22, 1–12. https://doi.org/10.1093/bib/bbaa161
Vieira, T. F., & Sousa, S. F. (2019). Comparing AutoDock and Vina in ligand/decoy discrimination for virtual screening. Applied Sciences, 9, 1–18. https://doi.org/10.3390/app9214538
Foundation TAS. (2012). Apache Cordova
Dias, L. C., Dessoy, M. A., Guido, R. V. C., Oliva, G., & Andricopulo, A. D. (2013). Doenças tropicais negligenciadas: Uma nova era de desafios e oportunidades. Quimica Nova, 36, 1552–1556. https://doi.org/10.1590/S0100-40422013001000011
Chen, N., Zhou, M., Dong, X., Qu, J., Gong, F., Han, Y., Qiu, Y., Wang, J., Liu, Y., Wei, Y., **a, J., Yu, T., Zhang, X., & Zhang, L. (2020). Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. The Lancet, 395, 507–513. https://doi.org/10.1016/S0140-6736(20)30211-7
Cui, J., Li, F., & Shi, Z. L. (2019). Origin and evolution of pathogenic coronaviruses. Nature Reviews Microbiology, 17, 181–192. https://doi.org/10.1038/s41579-018-0118-9
**, Z., Du, X., Xu, Y., Deng, Y., Liu, M., Zhao, Y., Zhang, B., Li, X., Zhang, L., Peng, C., Duan, Y., Yu, J., Wang, L., Yang, K., Liu, F., Jiang, R., Yang, X., You, T., Liu, X., … Yang, H. (2020). Structure of Mpro from SARS-CoV-2 and discovery of its inhibitors. Nature, 582, 289–293. https://doi.org/10.1038/s41586-020-2223-y
Ferreira, J. C., Fadl, S., Villanueva, A. J., & Rabeh, W. M. (2021). Catalytic Dyad residues His41 and Cys145 impact the catalytic activity and overall conformational fold of the main SARS-CoV-2 Protease 3-chymotrypsin-like protease. Frontiers in Chemistry, 9, 1–11. https://doi.org/10.3389/fchem.2021.692168
de Almeida, J. O., de Oliveira, V. R. T., Avelar, J. L. D. S., Moita, B. S., & Lima, L. M. (2020). COVID-19: Physiopathology and targets for therapeutic intervention [COVID-19: Fisiopatologia e Alvos para Intervenção Terapêutica]. Revista Virtual De Quimica, 12, 1464–1497.
Rester, U. (2008). From virtuality to reality—virtual screening in lead discovery and lead optimization: A medicinal chemistry perspective. Current Opinion in Drug Discovery & development, 11, 559–568.
Ashutosh, T., & Vytas, A. B. (2018). Molecular docking: From lock and key to combination lock. Journal of Molecular Medicine and Clinical Applications, 2, 1–9. https://doi.org/10.16966/2575-0305.106
Benet, L. Z., Hosey, C. M., Ursu, O., & Oprea, T. I. (2016). BDDCS, the rule of 5 and drugability. Advanced Drug Delivery Reviews. https://doi.org/10.1016/j.addr.2016.05.007
Beigel, J. H., Tomashek, K. M., Dodd, L. E., Mehta, A. K., Zingman, B. S., Kalil, A. C., Hohmann, E., Chu, H. Y., Luetkemeyer, A., Kline, S., Lopez de Castilla, D., Finberg, R. W., Dierberg, K., Tapson, V., Hsieh, L., Patterson, T. F., Paredes, R., Sweeney, D. A., Short, W. R., … Lane, H. C. (2020). Remdesivir for the treatment of Covid-19—preliminary report. New England Journal of Medicine, 1, 1–12. https://doi.org/10.1056/nejmoa2007764
de Ruyck, J., Brysbaert, G., Blossey, R., & Lensink, M. F. (2016). Molecular docking as a popular tool in drug design, an in silico travel. Advances and Applications in Bioinformatics and Chemistry, 9, 1–11. https://doi.org/10.2147/AABC.S105289
Ruba, S., Arooj, M., & Naz, D. G. (2014). In silico molecular docking studies and design of dengue virus inhibitors. Journal Of Pharmacy and Biologycal Science, 9, 15–23. https://doi.org/10.9790/3008-09211523
Huang, H. J., Yu, H. W., Chen, C. Y., Hsu, C. H., Chen, H. Y., Lee, K. J., Tsai, F. J., & Chen, C. Y. C. (2010). Current developments of computer-aided drug design. Journal of the Taiwan Institute of Chemical Engineers, 41, 623–635. https://doi.org/10.1016/j.jtice.2010.03.017
Morris, G. M., Huey, R., Lindstrom, W., Sanner, M. F., Belew, R. K., Goodsell, D. S., & Olson, A. J. (2009). AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. Journal of Computational Chemistry, 30, 2785–2791. https://doi.org/10.1002/jcc.21256
Yan, J., Zhang, G., Pan, J., & Wang, Y. (2014). α-Glucosidase inhibition by luteolin: Kinetics, interaction and molecular docking. International Journal of Biological Macromolecules, 64, 213–223. https://doi.org/10.1016/j.ijbiomac.2013.12.007
Kumar, Y., Singhc, H., & Patel, C. N. (2020). In silico prediction of potential inhibitors for the main protease of SARS-CoV-2 using molecular. Journal of Infection and Public Health, 13, 1–14. https://doi.org/10.1016/j.jiph.2020.06.016
Biovia. (2015). Dassault systemes BIOVIA, discovery studio modelling environment, release 4.5. Accelrys Softw. Inc.
Marinho, E. M., de Andrade, Batista, Neto, J., Silva, J., Rocha da Silva, C., Cavalcanti, B. C., Marinho, E. S., & Nobre Júnior, H. V. (2020). Virtual screening based on molecular docking of possible inhibitors of Covid-19 main protease. Microbial Pathogenesis, 148, 104365. https://doi.org/10.1016/j.micpath.2020.104365
Shityakov, S., & Förster, C. (2014). In silico predictive model to determine vector-mediated transport properties for the blood-brain barrier choline transporter. Advances and Applications in Bioinformatics and Chemistry. https://doi.org/10.2147/AABC.S63749
Imberty, A., Hardman, K. D., Carver, J. P., & Perez, S. (1991). Molecular modelling of protein-carbohydrate interactions. Docking of monosaccharides in the binding site of concanavalin A. Glycobiology, 1, 631–642. https://doi.org/10.1093/glycob/1.6.631
Schrödinger, L. (2010). Maestro, version 9.1. New York, NY
Jorgensen, W. L., Chandrasekhar, J., Madura, J. D., Impey, R. W., & Klein, M. L. (1983). Comparison of simple potential functions for simulating liquid water. The Journal of Chemical Physics, 79, 926–935. https://doi.org/10.1063/1.445869
Ryckaert, J. P., Ciccotti, G., & Berendsen, H. J. C. (1977). Numerical integration of the cartesian equations of motion of a system with constraints: Molecular dynamics of n-alkanes. Journal of Computational Physics, 23, 327–341. https://doi.org/10.1016/0021-9991(77)90098-5
Cheatham, T. E., Miller, J. L., Fox, T., Darden, T. A., & Kollman, P. A. (1995). Molecular dynamics simulations on solvated biomolecular systems: The particle mesh Ewald method leads to stable trajectories of DNA, RNA, and proteins. Journal of the American Chemical Society, 117, 4193–4194.
Hess, B., Bekker, H., Berendsen, H. J. C., & Fraaije, J. G. E. M. (1997). LINCS: A linear constraint solver for molecular simulations. Journal of Computational Chemistry, 18, 1463–1472. https://doi.org/10.1002/(SICI)1096-987X(199709)18:12%3c1463::AID-JCC4%3e3.0.CO;2-H
Phillips, J. C., Braun, R., Wang, W., Gumbart, J., Tajkhorshid, E., Villa, E., Chipot, C., Skeel, R. D., Kalé, L., & Schulten, K. (2005). Scalable molecular dynamics with NAMD. Journal of Computational Chemistry, 26, 1781–1802. https://doi.org/10.1002/jcc.20289
Turner, P. (2005). XMGRACE, Version 5.1. 19. Center for Coastal and Land-Margin Research, Oregon Graduate Institute of Science and Technology, Beaverton
Humphrey, W., Dalke, A., & Schulten, K. (1996). VMD: Visual molecular dynamics. Journal of Molecular Graphics, 14, 33–38. https://doi.org/10.1016/0263-7855(96)00018-5
Diez, M., Petuya, V., Martínez-Cruz, L. A., & Hernández, A. (2014). Insights into mechanism kinematics for protein motion simulation. BMC Bioinformatics. https://doi.org/10.1186/1471-2105-15-184
Arshia, A. H., Shadravan, S., Solhjoo, A., Sakhteman, A., & Sami, A. (2021). De novo design of novel protease inhibitor candidates in the treatment of SARS-CoV-2 using deep learning, docking, and molecular dynamic simulations. Computers in Biology and Medicine. https://doi.org/10.1016/j.compbiomed.2021.104967
Gohlke, H., Kiel, C., & Case, D. A. (2003). Insights into protein-protein binding by binding free energy calculation and free energy decomposition for the Ras-Raf and Ras-RalGDS complexes. Journal of Molecular Biology, 330, 891–913. https://doi.org/10.1016/S0022-2836(03)00610-7
DasGupta, D., Mandalaparthy, V., & Jayaram, B. (2017). A component analysis of the free energies of folding of 35 proteins: A consensus view on the thermodynamics of folding at the molecular level. Journal of Computational Chemistry, 38, 2791–2801. https://doi.org/10.1002/jcc.25072
Genheden, S., & Ryde, U. L. F. (2010). How to obtain statistically converged MM/GBSA results. Journal of Computational Chemistry, 31, 837–846. https://doi.org/10.1002/jcc.21366
Fan, J., Fu, A., & Zhang, L. (2019). Progress in molecular docking. Quantitative Biology, 7, 83–89. https://doi.org/10.1007/s40484-019-0172-y
Lee, J., Lee, Y., Jung, Y. M., Park, J. H., Yoo, H. S., & Park, J. (2022). Discovery of E3 ligase ligands for target protein degradation. Molecules, 27, 1–23. https://doi.org/10.3390/molecules27196515
Pettersen, E. F., Goddard, T. D., Huang, C. C., Meng, E. C., Couch, G. S., Croll, T. I., Morris, J. H., & Ferrin, T. E. (2021). UCSF ChimeraX: Structure visualization for researchers, educators, and developers. Protein Science, 30, 70–82. https://doi.org/10.1002/pro.3943
**, Z., Du, X., Xu, Y., Deng, Y., Liu, M., Zhao, Y., Zhang, B., Li, X., Zhang, L., Peng, C., Duan, Y., Yu, J., Wang, L., Yang, K., Liu, F., Jiang, R., Yang, X., You, T., Liu, X., … Yang, H. (2020). Structure of Mpro from COVID-19 virus and discovery of its inhibitors. bioRxiv, 582, 289–293.
Coutsias, E. A., & Wester, M. J. (2019). RMSD and symmetry. Journal of Computational Chemistry, 40, 1496–1508. https://doi.org/10.1002/jcc.25802
Hevener, K. E., Zhao, W., Ball, D. M., Babaoglu, K., Qi, J., White, S. W., & Lee, R. E. (2009). Validation of molecular docking programs for virtual screening against dihydropteroate synthase. Journal of Chemical Information and Modeling, 49, 444–460. https://doi.org/10.1021/ci800293n
Aniszewski, T. (2015). Alkaloid chemistry. Alkaloids (pp. 99–193). Elsevier.
Rule, A. M. (2004). American society of health-system pharmacists’ pain management network. Journal of Pain & Palliative Care Pharmacotherapy. https://doi.org/10.1300/J354v18n03_06
Majumdar, A., Kitson, M. T., & Roberts, S. K. (2016). Systematic review: Current concepts and challenges for the direct-acting antiviral era in hepatitis C cirrhosis. Alimentary Pharmacology & Therapeutics, 43, 1276–1292. https://doi.org/10.1111/apt.13633
Giannini, A. J., & Slaby, A. E. (1989). Drugs of abuse. Medical Economics Books.
Ernst, E. (2010). Index Nominum 2000. International drug director. Focus on Alternative and Complementary Therapies, 5, 233–233. https://doi.org/10.1111/j.2042-7166.2000.tb02559.x
Si, B., & Song, E. (2018). Recent advances in the detection of neurotransmitters. Chemosensors, 6, 1–24. https://doi.org/10.3390/chemosensors6010001
Diener, H. C., Jansen, J. P., Reches, A., Pascual, J., Pitei, D., & Steiner, T. J. (2002). Efficacy, tolerability and safety of oral eletriptan and ergotamine plus caffeine (Cafergot®) in the acute treatment of migraine: A multicentre, randomised, double-blind, placebo-controlled comparison. European Neurology, 47, 99–107. https://doi.org/10.1159/000047960
Sayfan, J. (2002). Ergotamine-induced anorectal strictures: Report of five cases. Diseases of the Colon and Rectum, 45, 271–272. https://doi.org/10.1007/s10350-004-6160-x
Kanfer, I., & Shargel, L. (2020). Approved drug products with therapeutic equivalence evaluations. Generic drug product development (pp. 36–51). The Orange Book.
Schiff, P. L. (2006). Ergot and its alkaloids. American Journal of Pharmaceutical Education, 70, 1–10. https://doi.org/10.5688/aj700598
Mevada, V., Dudhagara, P., Gandhi, H., Vaghamshi, Nilam Beladiya, & U, Patel R,. (2020). Drug repurposing of approved drugs Elbasvir, Ledipasvir, Paritaprevir, Velpatasvir, Antrafenine and Ergotamine for combating COVID19. ChemRxiv. https://doi.org/10.26434/chemrxiv.12115251.v2
Arcon, J. P., Defelipe, L. A., Modenutti, C. P., López, E. D., Alvarez-Garcia, D., Barril, X., Turjanski, A. G., & Martí, M. A. (2017). Molecular dynamics in mixed solvents reveals protein-ligand interactions, improves docking, and allows accurate binding free energy predictions. Journal of Chemical Information and Modeling, 57, 846–863. https://doi.org/10.1021/acs.jcim.6b00678
Struchtrup, H. (2020). Entropy and the second law of thermodynamics-The nonequilibrium perspective. Entropy, 22, 1–61. https://doi.org/10.3390/e22070793
Beretta, G. P. (2020). The fourth law of thermodynamics: Steepest entropy ascent. Philosophical Transactions of the Royal Society A: Mathematical Physical and Engineering Sciences, 378, 1–17. https://doi.org/10.1098/rsta.2019.0168
Chinaka, T. W. (2021). Introducing the second law of thermodynamics using Legitimation Code Theory among first year chemistry students. Cypriot Journal of Educational Sciences, 16, 981–994.
Du, X., Li, Y., **a, Y. L., Ai, S. M., Liang, J., Sang, P., Ji, X. L., & Liu, S. Q. (2016). Insights into protein–ligand interactions: Mechanisms, models, and methods. International Journal of Molecular Sciences, 17, 1–34. https://doi.org/10.3390/ijms17020144
Byléhn, F., Menéndez, C. A., Perez-Lemus, G. R., Alvarado, W., & De Pablo, J. J. (2021). Modeling the binding mechanism of Remdesivir, Favilavir, and Ribavirin to SARS-CoV-2 RNA-dependent RNA polymerase. ACS Central Science, 7, 164–174. https://doi.org/10.1021/acscentsci.0c01242
Miyamoto, Y., Motohashi, H., Suyama, T., & Yokoyama, J. (2014). Langevin description of gauged scalar fields in a thermal bath. Physical Review D. https://doi.org/10.1103/PhysRevD.89.085037
Qin, X., Zhong, J., & Wang, Y. (2021). A mutant T1 lipase homology modeling, and its molecular docking and molecular dynamics simulation with fatty acids. Journal of Biotechnology, 337, 24–34. https://doi.org/10.1016/j.jbiotec.2021.06.024
Roe, D. R., & Brooks, B. R. (2020). A protocol for preparing explicitly solvated systems for stable molecular dynamics simulations. The Journal of Chemical Physics, 153, 1–9. https://doi.org/10.1063/5.0013849
Mascoli, V., Liguori, N., Cupellini, L., Elias, E., Mennucci, B., & Croce, R. (2021). Uncovering the interactions driving carotenoid binding in light-harvesting complexes. Chemical Science, 12, 5113–5122. https://doi.org/10.1039/d1sc00071c
Ragunathan, A., Malathi, K., & Anbarasu, A. (2018). MurB as a target in an alternative approach to tackle the Vibrio cholerae resistance using molecular docking and simulation study. Journal of Cellular Biochemistry, 119, 1726–1732. https://doi.org/10.1002/jcb.26333
Mazola, Y., Guirola, O., Palomares, S., Chinea, G., Menéndez, C., Hernández, L., & Musacchio, A. (2015). A comparative molecular dynamics study of thermophilic and mesophilic β-fructosidase enzymes. Journal of Molecular Modeling, 21, 1–11. https://doi.org/10.1007/s00894-015-2772-4
Genheden, S., & Ryde, U. (2015). The MM/PBSA and MM/GBSA methods to estimate ligand-binding affinities. Expert Opinion on Drug Discovery, 10, 449–461.
Chen, F., Sun, H., Wang, J., Zhu, F., Liu, H., Wang, Z., Lei, T., Li, Y., & Hou, T. (2018). Assessing the performance of MM/PBSA and MM/GBSA methods. 8. Predicting binding free energies and poses of protein-RNA complexes. RNA, 24, 1183–1194. https://doi.org/10.1261/rna.065896.118
Wang, C., Greene, D., **ao, L., Qi, R., & Luo, R. (2018). Recent developments and applications of the MMPBSA method. Frontiers in Molecular Biosciences, 4, 1–18.
Wright, D. W., Hall, B. A., Kenway, O. A., Jha, S., & Coveney, P. V. (2014). Computing clinically relevant binding free energies of HIV-1 protease inhibitors. Journal of Chemical Theory and Computation, 10, 1228–1241. https://doi.org/10.1021/ct4007037
Ben-Tal, N., Honig, B., Bagdassarian, C. K., & Ben-Shaul, A. (2000). Association entropy in adsorption processes. Biophysical Journal, 79, 1180–1187. https://doi.org/10.1016/S0006-3495(00)76372-7
Šponer, J., Hobza, P., & Leszczynski, J. (1999). Chapter 3 Computational approaches to the studies of the interactions of nucleic acid bases. Theoretical and computational chemistry (pp. 85–117). Elsevier.
Emirik, M. (2022). Potential therapeutic effect of turmeric contents against SARS-CoV-2 compared with experimental COVID-19 therapies: In silico study. Journal of Biomolecular Structure & Dynamics, 40, 2024–2037. https://doi.org/10.1080/07391102.2020.1835719
Funding
The authors would like to thank the Brazilian Agencies: Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico (FUNCAP). Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Ensino Superior (CAPES) for fellowships and financial supports. The authors would like to thank project Inova Fiocruz FUNCAP (Grant#:06481104-2020), CNPq (Grant 306008/2022-0).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical Approval
Not applicable.
Informed Consent
Not applicable.
Consent to Participate
Not applicable.
Consent for publication
The authors affirm that provided informed consent for publication.
Institutional Review Board
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
da Fonseca, A.M., Caluaco, B.J., Madureira, J.M.C. et al. Screening of Potential Inhibitors Targeting the Main Protease Structure of SARS-CoV-2 via Molecular Docking, and Approach with Molecular Dynamics, RMSD, RMSF, H-Bond, SASA and MMGBSA. Mol Biotechnol (2023). https://doi.org/10.1007/s12033-023-00831-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12033-023-00831-x