Abstract
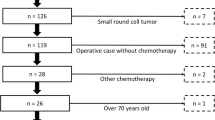
The purpose of this study was to investigate the feasibility and efficacy of pirarubicin (THP)–cisplatin (DDP) chemotherapy for refractory and recurrent high-grade osteosarcoma. Between 2008 and 2010, 23 patients with refractory and recurrent high-grade osteosarcoma were included in this analysis. THP was given at a dose of 50 mg/m2 i.v. d1 and DDP 100–120 mg/m2 i.v. d2–3 every 3 weeks. Treatment was continued until evidence of disease progression or unacceptable toxicity. Tumor response was usually evaluated every two chemotherapy cycles by CT/MRI scan. The primary end point was overall response rate, secondary endpoint including progression-free survival (PFS), overall survival (OS), disease control rate, and toxicities. A total of 68 cycles were given, median 2 per patient (range 2–7). Overall response rate was 13% and disease control rate was 34.5%, with 3 partial responses and 5 stable diseases. Median time to progression and overall survival time were 2 (95%CI 2–11) and 10 months (95%CI 6–23), respectively. Major severe toxicities were grade 3 or 4 leucopenia occurred 12 times (17.7%) in total cycles; Mild toxicities included grade 1 or 2 nausea and vomiting (80.9%), leucopenia (61.8%), fatigue (50.0%), and alopecia (79.4%). THP–DDP regimen chemotherapy represents an active and well-tolerated treatment for Chinese refractory and recurrent high-grade osteosarcoma patients. Further assessment is necessary to confirm the safety and efficacy of this treatment.


Similar content being viewed by others
References
Ta HT, Dass CR, Choong PF, Dunstan DE. Osteosarcoma treatment: state of the art. Cancer Metastasis Rev. 2009;28:247–63.
Wilkins RM, Cullen JW, Odom L, Jamroz BA, Cullen PM, et al. Superior survival in treatment of primary non-metastatic pediatric osteosarcoma of the extremity. Ann Surg Oncol. 2003;10:498–507.
Bielack SS, Kempf-Bielack B, Delling G, Exner GU, Flege S, et al. Prognostic factors in high-grade osteosarcoma of the extremities or trunk: an analysis of 1,702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J Clin Oncol. 2002;20:776–90.
Enneking WF. An abbreviated history of orthopaedic oncology in North America. Clin Orthop Relat Res. 2000;374:l15–124.
Arndt CA, Crist WM. Common musculoskeletal tumors of childhood and adolescence. N Engl J Med. 1999;341(5):342–52.
Ward WG, Mikaelian K, Dorey F, Mirra JM, Sassoon A, et al. Pulmonary metastases of stage IIB extremity osteosarcoma and subsequent pulmonary metastases. J Clin Oncol. 1994;12:1849–58.
Patel SR, Vadhan-Raj S, Papadopolous N, Plager C, Burgess MA, et al. High-dose ifosfamide in bone and soft tissue sarcomas: results of phase II and pilot studies-dose response and schedule dependence. J Clin Oncol. 1997;15:2378–84.
Kebudi R, Gorgun O, Ayan I. Oral etoposide for recurrent/progressive sarcomas of childhood. Pediatr Blood Cancer. 2004;42:320–4.
Seibel NL, Krailo M, Chen Z, Healey J, Breitfeld PP, et al. Upfront window trial of topotecan in previously untreated children and adolescents with poor prognosis metastatic osteosarcoma: children’s Cancer Group (CCG) 7943. Cancer. 2007;109:1646–53.
Crews KR, Stewart CF, Liu T, Rodriguez-Galindo C, Santana VM, et al. Effect of fractionated ifosfamide on the pharmacokinetics of irinotecan in pediatric patients with osteosarcoma. J Pediatr Hematol Oncol. 2004;26:764–7.
Bond M, Bernstein ML, Pappo A, Schultz KR, Krailo M, et al. A phase II study of imatinib mesylate in children with refractory or relapsed solid tumors. A children’s oncology group study. Pediatr Blood Cancer. 2008;50:254–8.
De Sio L, Milano GM, Castellano A, Jenkner A, Fidani P, et al. Temozolamide in resistant or relapsed pediatric solid tumors. Pediatr Blood Cancer. 2006;47:30–6.
Zou HY, Wu HL, Zhang Y, Li SF, Nie JF, et al. Studying the interaction of pirarubicin with DNA and determining pirarubicin in human urine samples: combining excitation—emission fluorescence matrices with second-order calibration methods. J Fluoresc. 2009;19:955–66.
Sridhar KS, Hussein AM, Benedetto P, Ardalan B, Savaraj N, et al. Phase II trial of 4’-0-tetrahydropyranyladriamycin (pirarubicin) in head and neck carcinoma. Cancer. 1992;70:1591–7.
Tsurumi H, Hara T, Goto N, Kanemura N, Kasahara S, et al. A phase II study of a THP-COP regimen for the treatment of elderly patients aged 70 years or older with diffuse large B-cell lymphoma. Hematol Oncol. 2007;25:107–14.
Shinozaki T, Watanabe H, Yanagawa T, Shirakura K, Takagishi K. Pirarubicin-based versus doxorubicin-based osteosarcoma chemotherapy. Ann Pharmacother. 2002;36:996–9.
Zhao H, Yao Y, Wang Z, Lin F, Sun Y, et al. Therapeutic effect of pirarubicin-based chemotherapy for osteosarcoma patients with lung metastasis. J Chemother. 2010;22:119–24.
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92:205–16.
Navid F, Willert JR, McCarville MB, Furman W, Watkins A, et al. Combination of gemcitabine and docetaxel in the treatment of children and young adults with refractory bone sarcoma. Cancer. 2008;113:419–25.
Berger M, Grignani G, Ferrari S, Biasin E, Brach del Prever A, et al. Phase 2 trial of two courses of cyclophosphamide and etoposide for relapsed high-risk osteosarcoma patients. Cancer. 2009;115:2980–7.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qi, WX., He, AN., Tang, LN. et al. Evaluation of pirarubicin–cisplatin chemotherapy in the treatment for refractory and recurrent high-grade osteosarcoma: experience of a single institute. Med Oncol 29, 2229–2233 (2012). https://doi.org/10.1007/s12032-011-0021-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-011-0021-y