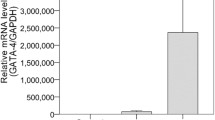
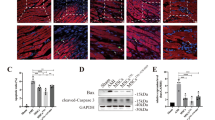
Abstract
Current myocardial infarction (MI) treatments are suboptimal, necessitating deeper pathogenesis understanding of MI. This research explored how exosomes (Exo) derived from bone marrow mesenchymal stem cells (BMSCs) contribute to MI mitigation and their therapeutic potential. Isolated BMSCs was identified by microscope, flow cytometry, alizarin red and oil red O staining. Exo were identified by TEM, NTA and western blot. HE staining, masson staining, and cardiac function parameters were used to assess the cardiac function in MI mice. TUNEL staining, western blot and qRT-PCR were used to detect apoptosis, inflammatory factors and M1/M2 markers. The NF-κB pathway activation was detected through western blot assays. Immunofluorescence, qRT-PCR, western blot, and flow cytometry were employed to evaluate macrophage polarization. MI mice showed cardiac injury, increased apoptosis and inflammation, while BMSCs-Exo treatment alleviated these effects. In MI mice, the macrophage M1 polarization was increased and the NF-κB pathway was activated, whereas BMSCs-Exo treatment reversed these changes. Furthermore, CISH expression was reduced in MI mice, but was elevated with BMSCs-Exo treatment. In vitro, LPS shifted RAW264.7 cells to M1 phenotype and activated the NF-κB pathway, yet BMSCs-Exo shifted them to M2 phenotype and inhibited the NF-κB pathway. Mechanistically, BMSCs-Exo induced macrophage M2 polarization by transmitting CISH to inhibit NF-κB activation. BMSCs-Exo mitigates MI by transmitting CISH to inhibit the NF-κB pathway, promoting macrophages to M2 type. This implies BMSCs-Exo could be a useful treatment for MI, and CISH could be a potential therapy target.






Similar content being viewed by others
Data Availability
All data generated or analysed during this study are included in this published article.
References
Thygesen, K., Alpert, J. S., Jaffe, A. S., Chaitman, B. R., Bax, J. J., Morrow, D. A., & White, H. D. (2018). Fourth Universal Definition of Myocardial Infarction. Circulation, 138, e618–e651.
Reed, G. W., & Menon, V. (2022). Reducing the incidence and mortality from myocardial infarction. Lancet Public Health, 7, e202–e203.
Pan, Q., Xu, J., Wen, C. J., **ong, Y. Y., Gong, Z. T., & Yang, Y. J. (2021). Nanoparticles: promising tools for the treatment and prevention of myocardial infarction. International Journal of Nanomedicine, 16, 6719–6747.
O’Rourke, S. A., Dunne, A., & Monaghan, M. G. (2019). The role of macrophages in the infarcted myocardium: orchestrators of ecm remodeling. Frontiers in cardiovascular medicine, 6, 101.
Jung, M., Dodsworth, M., & Thum, T. (2018). Inflammatory cells and their non-coding RNAs as targets for treating myocardial infarction. Basic Research in Cardiology, 114, 4.
Hume, R. D., & Chong, J. J. H. (2020). The cardiac injury immune response as a target for regenerative and cellular therapies. Clinical Therapeutics, 42, 1923–1943.
Heidt, T., Courties, G., Dutta, P., Sager, H. B., Sebas, M., Iwamoto, Y., Sun, Y., Da Silva, N., Panizzi, P., van der Laan, A. M., Swirski, F. K., Weissleder, R., & Nahrendorf, M. (2014). Differential contribution of monocytes to heart macrophages in steady-state and after myocardial infarction. Circulation Research, 115, 284–295.
Ma, Y., Mouton, A. J., & Lindsey, M. L. (2018). Cardiac macrophage biology in the steady-state heart, the aging heart, and following myocardial infarction. Translational Research, 191, 15–28.
Zhang, J., Huang, F., Chen, L., Li, G., Lei, W., Zhao, J., Liao, Y., Li, Y., Li, C., & Chen, M. (2021). Sodium lactate accelerates m2 macrophage polarization and improves cardiac function after myocardial infarction in mice. Cardiovascular Therapeutics, 2021, 5530541.
Pegtel, D. M., & Gould, S. J. (2019). Exosomes. Annual Review of Biochemistry, 88, 487–514.
Wu, X., Iroegbu, C. D., Peng, J., Guo, J., Yang, J., & Fan, C. (2021). Cell death and exosomes regulation after myocardial infarction and ischemia-reperfusion. Frontiers in Cell and Developmental Biology, 9, 673677.
Wu, Z., Cheng, S., Wang, S., Li, W., & Liu, J. (2021). BMSCs-derived exosomal microRNA-150-5p attenuates myocardial infarction in mice. International Immunopharmacology, 93, 107389.
Deng, S., Zhou, X., Ge, Z., Song, Y., Wang, H., Liu, X., & Zhang, D. (2019). Exosomes from adipose-derived mesenchymal stem cells ameliorate cardiac damage after myocardial infarction by activating S1P/SK1/S1PR1 signaling and promoting macrophage M2 polarization. International Journal of Biochemistry & Cell Biology, 114, 105564.
Naser, W., Maymand, S., Rivera, L. R., Connor, T., Liongue, C., Smith, C. M., Aston-Mourney, K., McCulloch, D. R., McGee, S. L., & Ward, A. C. (2022). Cytokine-inducible SH2 domain containing protein contributes to regulation of adiposity, food intake, and glucose metabolism. The FASEB Journal, 36, e22320.
Mujalli, A., Banaganapalli, B., Alrayes, N. M., Shaik, N. A., Elango, R., & Al-Aama, J. Y. (2020). Myocardial infarction biomarker discovery with integrated gene expression, pathways and biological networks analysis. Genomics, 112, 5072–5085.
Lachtermacher, S., Esporcatte, B. L., Montalvao, F., Costa, P. C., Rodrigues, D. C., Belem, L., Rabischoffisky, A., Faria Neto, H. C., Vasconcellos, R., Iacobas, S., Iacobas, D. A., Dohmann, H. F., Spray, D. C., Goldenberg, R. C., & Campos-de-Carvalho, A. C. (2010). Cardiac gene expression and systemic cytokine profile are complementary in a murine model of post-ischemic heart failure. Brazilian Journal of Medical and Biological Research, 43, 377–389.
Zhang, S., Rautela, J., Bediaga, N. G., Kolesnik, T. B., You, Y., Nie, J., Dagley, L. F., Bedo, J., Wang, H., Sun, L., Sutherland, R., Surgenor, E., Iannarella, N., Allan, R., Souza-Fonseca-Guimaraes, F., **e, Y., Wang, Q., Zhang, Y., Xu, Y., … Zhan, Y. (2023). CIS controls the functional polarization of GM-CSF-derived macrophages. Cellular & Molecular Immunology, 20, 65–79.
Hu, G., Zhou, R., Liu, J., Gong, A. Y., Eischeid, A. N., Dittman, J. W., & Chen, X. M. (2009). MicroRNA-98 and let-7 confer cholangiocyte expression of cytokine-inducible Src homology 2-containing protein in response to microbial challenge. The Journal of Immunology, 183, 1617–1624.
Yin, C., Ye, Z., Wu, J., Huang, C., Pan, L., Ding, H., Zhong, L., Guo, L., Zou, Y., Wang, X., Wang, Y., Gao, P., **, X., Yan, X., Zou, Y., Huang, R., & Gong, H. (2021). Elevated Wnt2 and Wnt4 activate NF-kappaB signaling to promote cardiac fibrosis by cooperation of Fzd4/2 and LRP6 following myocardial infarction. eBioMedicine, 74, 103745.
Dan, H., Liu, S., Liu, J., Liu, D., Yin, F., Wei, Z., Wang, J., Zhou, Y., Jiang, L., Ji, N., Zeng, X., Li, J., & Chen, Q. (2020). RACK1 promotes cancer progression by increasing the M2/M1 macrophage ratio via the NF-kappaB pathway in oral squamous cell carcinoma. Molecular Oncology, 14, 795–807.
Ni, L., Lin, Z., Hu, S., Shi, Y., Jiang, Z., Zhao, J., Zhou, Y., Wu, Y., Tian, N., Sun, L., Wu, A., Pan, Z., Zhang, X., & Wang, X. (2022). Itaconate attenuates osteoarthritis by inhibiting STING/NF-kappaB axis in chondrocytes and promoting M2 polarization in macrophages. Biochemical Pharmacology, 198, 114935.
Zhang, L., & Chan, C. (2010). Isolation and enrichment of rat mesenchymal stem cells (MSCs) and separation of single-colony derived MSCs. Journal of Visualized Experiments, 37, e1852.
Zhu, Q., Tang, S., Zhu, Y., Chen, D., Huang, J., & Lin, J. (2022). Exosomes derived from CTF1-modified bone marrow stem cells promote endometrial regeneration and restore fertility. Frontiers in Bioengineering and Biotechnology, 10, 868734.
Chen, B., Ni, Y., Liu, J., Zhang, Y., & Yan, F. (2018). Bone marrow-derived mesenchymal stem cells exert diverse effects on different macrophage subsets. Stem Cells International, 2018, 8348121.
Li, X., Bi, T., & Yang, S. (2022). Exosomal microRNA-150-5p from bone marrow mesenchymal stromal cells mitigates cerebral ischemia/reperfusion injury via targeting toll-like receptor 5. Bioengineered, 13, 3030–3043.
Peng, Y., Chen, B., Zhao, J., Peng, Z., Xu, W., & Yu, G. (2019). Effect of intravenous transplantation of hUCB-MSCs on M1/M2 subtype conversion in monocyte/macrophages of AMI mice. Biomedicine & Pharmacotherapy, 111, 624–630.
Wang, X., Guo, Z., Ding, Z., & Mehta, J. L. (2018). Inflammation, autophagy, and apoptosis after myocardial infarction. Journal of the American Heart Association, 7, e008024.
Meeran, M. F. N., Azimullah, S., Adeghate, E., & Ojha, S. (2021). Nootkatone attenuates myocardial oxidative damage, inflammation, and apoptosis in isoproterenol-induced myocardial infarction in rats. Phytomedicine, 84, 153405.
Viola, A., Munari, F., Sanchez-Rodriguez, R., Scolaro, T., & Castegna, A. (2019). The metabolic signature of macrophage responses. Frontiers in Immunology, 10, 1462.
Cheng, Y., & Rong, J. (2018). Macrophage polarization as a therapeutic target in myocardial infarction. Current Drug Targets, 19, 651–662.
Zhao, J., Li, X., Hu, J., Chen, F., Qiao, S., Sun, X., Gao, L., **e, J., & Xu, B. (2019). Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovascular Research, 115, 1205–1216.
Liu, L., Guo, H., Song, A., Huang, J., Zhang, Y., **, S., Li, S., Zhang, L., Yang, C., & Yang, P. (2020). Progranulin inhibits LPS-induced macrophage M1 polarization via NF-small ka. CyrillicB and MAPK pathways. BMC Immunol, 21, 32.
Kawano, S., Kubota, T., Monden, Y., Tsutsumi, T., Inoue, T., Kawamura, N., Tsutsui, H., & Sunagawa, K. (2006). Blockade of NF-kappaB improves cardiac function and survival after myocardial infarction. American Journal of Physiology. Heart and Circulatory Physiology, 291, H1337-1344.
Wen, Z., Zheng, S., Zhou, C., Wang, J., & Wang, T. (2011). Repair mechanisms of bone marrow mesenchymal stem cells in myocardial infarction. Journal of Cellular and Molecular Medicine, 15, 1032–1043.
Barron, C. C., Lalu, M. M., Stewart, D. J., Fergusson, D., Yang, H., Moher, D., Liu, P., Mazer, D., Devereaux, P. J., McIntyre, L., Trials, C. P. A. C., & G. (2017). Assessment of safety and efficacy of mesenchymal stromal cell therapy in preclinical models of acute myocardial infarction: A systematic review protocol. Systematic Reviews, 6, 226.
Gnecchi, M., Danieli, P., & Cervio, E. (2012). Mesenchymal stem cell therapy for heart disease. Vascular Pharmacology, 57, 48–55.
Motavaf, M., Pakravan, K., Babashah, S., Malekvandfard, F., Masoumi, M., & Sadeghizadeh, M. (2016). Therapeutic application of mesenchymal stem cell-derived exosomes: A promising cell-free therapeutic strategy in regenerative medicine. Cellular and Molecular Biology, 62, 74–79.
Hu, M., Guo, G., Huang, Q., Cheng, C., Xu, R., Li, A., Liu, N., & Liu, S. (2018). The harsh microenvironment in infarcted heart accelerates transplanted bone marrow mesenchymal stem cells injury: The role of injured cardiomyocytes-derived exosomes. Cell Death & Disease, 9, 357.
Ju, C., Shen, Y., Ma, G., Liu, Y., Cai, J., Kim, I. M., Weintraub, N. L., Liu, N., & Tang, Y. (2018). Transplantation of Cardiac Mesenchymal Stem Cell-Derived Exosomes Promotes Repair in Ischemic Myocardium. Journal of Cardiovascular Translational Research, 11, 420–428.
Ni, J., Sun, Y., & Liu, Z. (2019). The potential of stem cells and stem cell-derived exosomes in treating cardiovascular diseases. Journal of Cardiovascular Translational Research, 12, 51–61.
Zheng, J., Zhang, X., Cai, W., Yang, Y., Guo, T., Li, J., & Dai, H. (2022). Bone marrow mesenchymal stem cell-derived exosomal microRNA-29b-3p promotes angiogenesis and ventricular remodeling in rats with myocardial infarction by targeting ADAMTS16. Cardiovascular Toxicology, 22, 689–700.
Li, Y., Yang, R., Guo, B., Zhang, H., Zhang, H., Liu, S., & Li, Y. (2019). Exosomal miR-301 derived from mesenchymal stem cells protects myocardial infarction by inhibiting myocardial autophagy. Biochemical and Biophysical Research Communications, 514, 323–328.
Zhu, W., Sun, L., Zhao, P., Liu, Y., Zhang, J., Zhang, Y., Hong, Y., Zhu, Y., Lu, Y., Zhao, W., Chen, X., & Zhang, F. (2021). Macrophage migration inhibitory factor facilitates the therapeutic efficacy of mesenchymal stem cells derived exosomes in acute myocardial infarction through upregulating miR-133a-3p. Journal of Nanobiotechnology, 19, 61.
Zhu, W., Wang, Q., Zhang, J., Sun, L., Hong, X., Du, W., Duan, R., Jiang, J., Ji, Y., Wang, H., & Han, B. (2023). Exosomes derived from mir-214-3p overexpressing mesenchymal stem cells promote myocardial repair. Biomaterials Research, 27, 77.
Song, Y., Zhang, C., Zhang, J., Jiao, Z., Dong, N., Wang, G., Wang, Z., & Wang, L. (2019). Localized injection of miRNA-21-enriched extracellular vesicles effectively restores cardiac function after myocardial infarction. Theranostics, 9, 2346–2360.
**ao, F., Deng, J., Jiao, F., Hu, X., Jiang, H., Yuan, F., Chen, S., Niu, Y., Jiang, X., & Guo, F. (2022). Hepatic cytokine-inducible SH2-containing protein (CISH) regulates gluconeogenesis via cAMP-responsive element binding protein (CREB). The FASEB Journal, 36, e22541.
Zhu, H., Blum, R. H., Bernareggi, D., Ask, E. H., Wu, Z., Hoel, H. J., Meng, Z., Wu, C., Guan, K. L., Malmberg, K. J., & Kaufman, D. S. (2020). Metabolic Reprograming via Deletion of CISH in Human iPSC-Derived NK Cells Promotes In Vivo Persistence and Enhances Anti-tumor Activity. Cell Stem Cell, 27(224–237), e226.
Sager, H. B., Hulsmans, M., Lavine, K. J., Moreira, M. B., Heidt, T., Courties, G., Sun, Y., Iwamoto, Y., Tricot, B., Khan, O. F., Dahlman, J. E., Borodovsky, A., Fitzgerald, K., Anderson, D. G., Weissleder, R., Libby, P., Swirski, F. K., & Nahrendorf, M. (2016). Proliferation and Recruitment Contribute to myocardial macrophage expansion in chronic heart failure. Circulation Research, 119, 853–864.
Peng, Y., Pan, W., Ou, Y., Xu, W., Kaelber, S., Borlongan, C. V., Sun, M., & Yu, G. (2016). Extracardiac-lodged mesenchymal stromal cells propel an inflammatory response against myocardial infarction via paracrine effects. Cell Transplantation, 25, 929–935.
Weirather, J., Hofmann, U. D., Beyersdorf, N., Ramos, G. C., Vogel, B., Frey, A., Ertl, G., Kerkau, T., & Frantz, S. (2014). Foxp3+ CD4+ T cells improve healing after myocardial infarction by modulating monocyte/macrophage differentiation. Circulation Research, 115, 55–67.
Lawrence, T. (2009). The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harbor Perspectives in Biology, 1, a001651.
Viatour, P., Merville, M. P., Bours, V., & Chariot, A. (2005). Phosphorylation of NF-kappaB and IkappaB proteins: Implications in cancer and inflammation. Trends in Biochemical Sciences, 30, 43–52.
Zhang, Q., Wang, L., Wang, S., Cheng, H., Xu, L., Pei, G., Wang, Y., Fu, C., Jiang, Y., He, C., & Wei, Q. (2022). Signaling pathways and targeted therapy for myocardial infarction. Signal Transduction and Targeted Therapy, 7, 78.
Acknowledgements
Not applicable.
Funding
This work was supported by the Natural Science Foundation of Hunan Province (2023JJ40847) and The Voyage Project of The Second **angya Hospital of Central South University.
Author information
Authors and Affiliations
Contributions
Yi Peng is the guarantor of integrity of the entire study; Guolong Yu contributed to the study concepts; Yi Peng contributed to the study design and manuscript editing; Yi Peng and Guolong Yu contributed to the definition of intellectual content and the manuscript review; Minzhi Ouyang and Jiling Zhao contributed to the literature research and experimental studies; Yang Yang contributed to the data acquisition and statistical analysis; Minzhi Ouyang contributed to the data analysis and manuscript preparation.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: GEN SUZUKI.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ouyang, M., Yang, Y., Yu, G. et al. BMSCs-derived Exosome CISH Alleviates Myocardial Infarction by Inactivating the NF-κB Pathway to Stimulate Macrophage M2 Polarization. Cardiovasc Toxicol 24, 422–434 (2024). https://doi.org/10.1007/s12012-024-09847-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-024-09847-4