Abstract
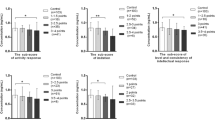
Existing data demonstrate a significant correlation between autism spectrum disorder (ASD) and the status of biologically essential and toxic trace elements. However, there is still a lack of data on the steady state of trace elements in ASD. We performed a case–control study to explore the association between the risk of ASD and 23 trace elements in plasma. The results showed that children with ASD had considerably decreased lithium (Li), manganese (Mn), selenium (Se), barium (Ba), mercury (Hg), and tin (Sn) levels when compared to their age- and sex-matched controls. Meanwhile, children with ASD had considerably increased plasma chromium (Cr) and vanadium (V) concentrations. We also divided each group into subgroups based on age and gender and created element-related networks for each subgroup. We detected significant element correlations within or between subgroups, as well as changes in correlations that included all elements examined. Finally, more element correlations were observed among males, which may open a new avenue for understanding the complicated process behind the sex ratio of children with ASD. Overall, our data revealed a novel relationship between elements and ASD, which may extend current understanding about ASD.





Similar content being viewed by others
Data Availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Shen L, Zhang K, Feng C, Chen Y, Li S, Iqbal J et al (2018) iTRAQ‐based proteomic analysis reveals protein profile in plasma from children with autism. Proteomics Clin Appl 12(3):1700085
Yao F, Zhang K, Feng C, Gao Y, Shen L, Liu X et al (2021) Protein biomarkers of autism spectrum disorder identified by computational and experimental methods. Front Psych 12:93
Maenner MJ, Shaw KA, Baio J, Washington A, Patrick M, Dirienzo M et al (2020) Prevalence of autism spectrum disorder among children aged 8 years—autism and developmental disabilities monitoring network, 11 sites, United States, 2016. MMWR Surveill Summ 69(4):1–12
Shen L, Zhao Y, Zhang H, Feng C, Gao Y, Zhao D et al (2019) Advances in biomarker studies in autism spectrum disorders. Reviews on Biomarker Studies in Psychiatric and Neurodegenerative Disorders, p. 207–233
Baxter AJ, Brugha TS, Erskine HE, Scheurer RW, Vos T, Scott JG (2015) The epidemiology and global burden of autism spectrum disorders. Psychol Med 45(3):601–613
Raina SK, Kashyap V, Bhardwaj AK, Kumar D, Chander V (2015) Prevalence of autism spectrum disorders among children (1–10 years of age)–findings of a mid-term report from Northwest India. J Postgrad Med 61(4):243–246
Zablotsky B, Black LI, Maenner MJ, Schieve LA (2014) Blumberg SJ (2015) Estimated prevalence of autism and other developmental disabilities following questionnaire changes in the. Natl Health Interview Survey 87:1–20
Wiśniowiecka-Kowalnik B, Nowakowska BA (2019) Genetics and epigenetics of autism spectrum disorder—current evidence in the field. J Appl Genet 60(1):37–47
Macedoni-Lukšič M, Gosar D, Bjørklund G, Orazem J, Kodric J, Lesnik-Musek P et al (2015) Levels of metals in the blood and specific porphyrins in the urine in children with autism spectrum disorders. Biol Trace Elem Res 163(1):2–10
Sharma SR, Gonda X, Tarazi FI (2018) Autism spectrum disorder: classification, diagnosis and therapy. Pharmacol Ther 190:91–104
Essa MM, Braidy N, Vijayan KR, Subash S, Guillemin GJ (2013) Excitotoxicity in the pathogenesis of autism. Neurotox Res 23(4):393–400
Shen L, Liu X, Zhang H, Lin J, Feng C, Iqbal J (2020) Biomarkers in autism spectrum disorders: current progress. Clin Chim Acta 502:41–54
Matelski L, Van de Water J (2016) Risk factors in autism: thinking outside the brain. J Autoimmun 67:1–7
Endreffy I, Bjørklund G, Dicső F, Urbina MA, Endreffy E (2016) Acid glycosaminoglycan (aGAG) excretion is increased in children with autism spectrum disorder, and it can be controlled by diet. Metab Brain Dis 31(2):273–278
Saad K, Abdel-Rahman AA, Elserogy YM, Al-Atram AA, Cannell JJ, Bjorklund G et al (2016) Vitamin D status in autism spectrum disorders and the efficacy of vitamin D supplementation in autistic children. Nutr Neurosci 19(8):346–351
Feng C, Chen Y, Pan J, Yang AC, Niu L, Min J et al (2017) Redox proteomic identification of carbonylated proteins in autism plasma: insight into oxidative stress and its related biomarkers in autism. Clin Proteomics 14(1):1–8
Zhang J, Li X, Shen L, Khan NU, Zhang X, Chen L et al (2021) Trace elements in children with autism spectrum disorder: a meta-analysis based on case-control studies. J Trace Elem Med Biol 67:126782
Zhang Y (2017) Trace elements and healthcare: a bioinformatics perspective. Transl Inform Smart Healthc 1005:63–98
El-Ansary A, Bjørklund G, Tinkov AA, Skalny AV, Al Dera H (2017) Relationship between selenium, lead, and mercury in red blood cells of Saudi autistic children. Metab Brain Dis 32(4):1073–1080
Khaled EM, Meguid NA, Bjørklund G, Gouda A, Bahary MH, Hashish A et al (2016) Altered urinary porphyrins and mercury exposure as biomarkers for autism severity in Egyptian children with autism spectrum disorder. Metab Brain Dis 31(6):1419–1426
Yassa HA (2014) Autism: a form of lead and mercury toxicity. Environ Toxicol Pharmacol 38(3):1016–1024
Mostafa GA, Bjørklund G, Urbina MA, Al-Ayadhi LY (2016) The positive association between elevated blood lead levels and brain-specific autoantibodies in autistic children from low lead-polluted areas. Metab Brain Dis 31(5):1047–1054
Rahbar MH, Samms-Vaughan M, Dickerson AS, Loveland KA, Ardjomand-Hessabi M, Bressler J et al (2014) Blood manganese concentrations in Jamaican children with and without autism spectrum disorders. Environ Health 13(1):1–14
Shaw CA, Tomljenovic L (2013) Aluminum in the central nervous system (CNS): toxicity in humans and animals, vaccine adjuvants, and autoimmunity. Immunol Res 56(2):304–316
Al-Ayadhi LY (2005) Heavy metals and trace elements in hair samples of autistic children in central Saudi Arabia. Neurosci J 10(3):213–218
Blaurock-Busch E, Amin OR, Rabah T (2011) Heavy metals and trace elements in hair and urine of a sample of Arab children with autistic spectrum disorder. Maedica 6(4):247–257
Yorbik Ö, Kurt İ, Haşimi A, Ozturk O (2010) Chromium, cadmium, and lead levels in urine of children with autism and typically develo** controls. Biol Trace Elem Res 135(1):10–15
Wu L, Mao S, Lin X, Yang RW, Zhu ZW (2019) Evaluation of whole blood trace element levels in Chinese children with autism spectrum disorder. Biol Trace Elem Res 191(2):269–275
Maret W (2013) Zinc biochemistry: from a single zinc enzyme to a key element of life. Adv Nutr 4(1):82–91
Jarosz M, Olbert M, Wyszogrodzka G, Yniec KM, Librowski T (2017) Antioxidant and anti-inflammatory effects of zinc. Zinc-dependent NF-κB signaling. Inflammopharmacology 25(1):11–24
Bjørklund G (2013) The role of zinc and copper in autism spectrum disorders. Acta Neurobiol Exp (Wars) 73(2):225–236
Jory J, McGinnis WR (2008) Red-cell trace minerals in children with autism. Am J Biochem Biotechnol 4(2):101–104
Skalny AV, Skalnaya MG, Bjørklund G, Gritsenko VA, Aaseth A, Tinkov AA (2018) Selenium and autism spectrum disorder. Selenium. Springer, Cham, p. 193–210
Reynolds A, Krebs NF, Stewart PA, Austin H, Hyman SL (2012) Iron status in children with autism spectrum disorder. Pediatrics 130(Suppl 2):154–159
Nuckols CC (2013) The diagnostic and statistical manual of mental disorders, (DSM-5). American Psychiatric Association, Philadelphia
Crowell MD, Umar SB, Lacy BE, Jones MP, DiBaise JK, Talley NJ (2015) Multi-dimensional gastrointestinal symptom severity index: validation of a brief GI symptom assessment Tool. Dig Dis Sci 60(8):2270–2279
Caito S, Aschner M (2015) Neurotoxicity of metals. Handb Clin Neurol 131:169–189
Ray RS, Rana B, Swami B, Venu V, Chatterjee M (2006) Vanadium mediated apoptosis and cell cycle arrest in MCF7 cell line. Chem Biol Interact 163(3):239–247
Li H, Zhou D, Zhang Q, Feng C, Zheng W, He K et al (2013) Vanadium exposure-induced neurobehavioral alterations among Chinese workers. Neurotoxicology 36:49–54
Jiang M, Li Y, Zhang B, Zhou AF, Zheng TZ, Qian ZM et al (2016) A nested case–control study of prenatal vanadium exposure and low birthweight. Hum Reprod 31(9):2135–2141
Clark MJ, Prentice JR, Hoggard N, Paley MN, Hadjivassiliou M, Wilkinson JM (2014) Brain structure and function in patients after metal-on-metal hip resurfacing. Am J Neuroradiol 35(9):1753–1758
Carson BL, Ellis HV, McCann JL (1986) Toxicology and biological monitoring of metals in humans: including feasibility and need, 1st edn. CRC Press. https://doi.org/10.1201/9781351077385
Costa M, Klein CB (2006) Toxicity and carcinogenicity of chromium compounds in humans. Crit Rev Toxicol 36(2):155–163
Chehbani F, Gallello G, Brahim T, Ouanes S, Douki W, Gaddour N et al (2020) The status of chemical elements in the blood plasma of children with autism spectrum disorder in Tunisia: a case-control study. Environ Sci Pollut Res 27(28):35738–35749
Al-Farsi YM, Waly MI, Al-Sharbati MM, Al-Shafaee MA, Al-Farsi OA, Al-Khaduri MM et al (2013) Levels of heavy metals and essential minerals in hair samples of children with autism in Oman: a case–control study. Biol Trace Elem Res 151(2):181–186
Yorbik O, Sayal A, Akay C, Akbiyik DI, Sohmen T (2002) Investigation of antioxidant enzymes in children with autistic disorder. Prostaglandins Leukot Essent Fatty Acids 67(5):341–343
Sezgin C, Kaya S, Keskin S (2010) Comparison of blood toxic and plasma essential elements of the autistic Turkish infants. FEBS J 277:88
Audyha T, McGinnis WR (2004) Nutrient, toxin and enzyme profile of autistic children. International Meeting for Autism Research, Sacramento, California, May 7–8, p 74
Arteel GE, Sies H (2001) The biochemistry of selenium and the glutathione system. Environ Toxicol Pharmacol 10(4):153–158
Schrauzer GN (1987) Effects of selenium antagonists on cancer susceptibility: new aspects of chronic heavy metal toxicity. J UOEH 9:208–215
Ming X, Johnson WG, Stenroos ES, Mars A, Lambert GH, Buyske S (2010) Genetic variant of glutathione peroxidase 1 in autism[J]. Brain Develop 32(2):105–109
Chapman L, Chan HM (2000) The influence of nutrition on methyl mercury intoxication. Environ Health Perspect 108(suppl 1):29–56
Bjørklund G, Mutter J, Aaseth J (2017) Metal chelators and neurotoxicity: lead, mercury, and arsenic. Arch Toxicol 91(12):3787–3797
Rahman MM, Hossain KFB, Banik S, Sikder MT, Akter M, Bondad SEC et al (2019) Selenium and zinc protections against metal-(loids)-induced toxicity and disease manifestations: a review. Ecotoxicol Environ Saf 168:146–163
Rahbar MH, Samms-Vaughan M, Ma J, Bressler J, Dickerson AS, Hessabi M et al (2015) Synergic effect of GSTP1 and blood manganese concentrations in autism spectrum disorder. Res Autism Spectr Disord 18:73–82
Lindenau J, Noack H, Possel H, Asayama K, Wolf G (2000) Cellular distribution of superoxide dismutases in the rat CNS. Glia 29(1):25–34
Kaur K, Chauhan V, Gu F, Chauhan A (2014) Bisphenol A induces oxidative stress and mitochondrial dysfunction in lymphoblasts from children with autism and unaffected siblings. Free Radic Biol Med 76:25–33
Gaetke LM, Chow CK (2003) Copper toxicity, oxidative stress, and antioxidant nutrients. Toxicology 189(1–2):147–163
Ercal N, Gurer-Orhan H, Aykin-Burns N (2001) Toxic metals and oxidative stress part I: mechanisms involved in metal-induced oxidative damage. Curr Top Med Chem 1(6):529–539
Adams JB, Holloway CE, George F, Quig D (2006) Analyses of toxic metals and essential minerals in the hair of Arizona children with autism and associated conditions, and their mothers. Biol Trace Elem Res 110(3):193–209
Sherman WR, Leavitt AL, Honchar MP, Hallcher LM, Phillips BE (1981) Evidence that lithium alters phosphoinositide metabolism: chronic administration elevates primarily D-myo-inositol-1-phosphate in cerebral cortex of the rat. J Neurochem 36(6):1947–1951
Berridge MJ, Downes CP, Hanley MR (1982) Lithium amplifies agonist-dependent phosphatidylinositol responses in brain and salivary glands. Biochem J 206(3):587–595
Pesavento JA, Kolli V, Garcia-Delgar B, Coffey BJ (2016) Autoimmune thyroiditis in an adolescent girl on lithium. J Child Adolesc Psychopharmacol 26(8):756–759
Ryu HU, Chung JY, Shin BS, Kang HG (2020) Lithium induced reversible splenial lesion in neuroleptic malignant syndrome like symptoms: two case reports. BMC Neurol 20(1):1–5
Schrauzer GN, Shrestha KP (1990) Lithium in drinking water and the incidences of crimes, suicides, and arrests related to drug addictions. Biol Trace Elem Res 25(2):105–113
Adams JB, Audhya T, McDonough-Means S, Rubin RA, Quig D, Geis E et al (2011) Nutritional and metabolic status of children with autism vs. neurotypical children, and the association with autism severity. Nutr Metab 8(1):1–32
Deixonne B, Baumel H, Mauras Y, Allain P, Robert C, Raffanel C (1983) A case of barium-peritoneum with neurological involvement. Importance of barium determination in biological fluids. J Chir 120(11):611–613
Skalny AV, Simashkova NV, Klyushnik TP, Grabeklis AR, Bjorklund G, Skalnaya MG et al (2017) Hair toxic and essential trace elements in children with autism spectrum disorder. Metab Brain Dis 32(1):195–202
Gerren RA, Groswald DE, Luttges MW (1976) Triethyltin toxicity as a model for degenerative disorders. Pharmacol Biochem Behav 5(3):299–307
Marshall LF, Bruce DA, Graham DI, Langfitt TW (1976) Alterations in behavior, brain electrical activity, cerebral blood flow, and intracranial pressure produced by triethyl tin sulfate induced cerebral edema. Stroke 7(1):21–25
Holmes AS, Blaxill MF, Haley BE (2003) Reduced levels of mercury in first baby haircuts of autistic children. Int J Toxicol 22(4):277–285
Kern JK, Grannemann BD, Trivedi MH, Adams JB (2007) Sulfhydryl-reactive metals in autism. J Toxicol Environ Health A 70(8):715–721
Kern JK, Jones AM (2006) Evidence of toxicity, oxidative stress, and neuronal insult in autism. J Toxicol Environ Health B 9(6):485–499
Qin YY, Jian B, Wu C, Jiang CZ, Kang Y, Zhou JX et al (2018) A comparison of blood metal levels in autism spectrum disorder and unaffected children in Shenzhen of China and factors involved in bioaccumulation of metals. Environ Sci Pollut Res 25(18):17950–17956
Majewska MD, Urbanowicz E, Rok-Bujko P, Namyslowska I, Mierzejewski P (2010) Age-dependent lower or higher levels of hair mercury in autistic children than in healthy controls. Acta Neurobiol Exp 70(2):196–208
Adams JB, Romdalvik J, Levine KE, Hu LW (2008) Mercury in first-cut baby hair of children with autism versus typically-develo** children. Toxicol Environ Chem 90(4):739–753
James SJ, Cutler P, Melnyk S, Jernigan S, Janak L, Gaylor DW et al (2004) Metabolic biomarkers of increased oxidative stress and impaired methylation capacity in children with autism. Am J Clin Nutr 80(6):1611–1617
Zhang Y, Xu Y, Zheng L (2020) Disease ionomics: understanding the role of ions in complex disease. Int J Mol Sci 21(22):8646
Skalny AV, Simashkova NV, Skalnaya AA, Klyushnik TP, Bjørklund G, Skalnaya MG et al (2017) Assessment of gender and age effects on serum and hair trace element levels in children with autism spectrum disorder. Metab Brain Dis 32(5):1675–1684
Funding
This study was supported by the National Natural Science Foundation of China (Grant No. 31870825), the Shenzhen Bureau of Science, Technology, and Information (No. JCYJ20170412110026229), and the Shenzhen-Hong Kong Institute of Brain Science-Shenzhen Fundamental Research Institutions (No. 2021SHIBS0003).
Author information
Authors and Affiliations
Contributions
LMS and JZ conceived and designed the experiments and drafted the manuscript. JZ, JL, XYZ, FY, ZJH, and XSC performed the experiments and analyzed the data. CYF and YG recruited patients and collected the blood samples. NUK, MC, and PL helped to revise the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
The Maternal and Child Health Hospital of Baoan and the Shenzhen University approved study procedures. Written informed consent was obtained from the parents and/or from legal guardians of each child prior to enrollment and all analyses in this study.
Consent to Participate and for Publication
All authors agree to publish.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, J., Lin, J., Zhao, X. et al. Trace Element Changes in the Plasma of Autism Spectrum Disorder Children and the Positive Correlation Between Chromium and Vanadium. Biol Trace Elem Res 200, 4924–4935 (2022). https://doi.org/10.1007/s12011-021-03082-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-03082-6