Abstract
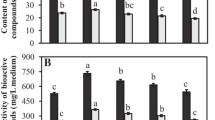
Indiscriminate collection of Arnebia benthamii roots from wild for extraction of red pigments by traders prompted to put this Himalayan plant species under critically endangered category. Concerning the development of sustainable phytoconstituents sources, the present study is aimed at understanding the factors crucial for the scalability of A. benthamii cell suspension cultures. Here, the effect of orbital shaking speed (60–80 rpm) and inoculum density (5–15%) on leaf-induced cell suspension cultures during sequential scale up in shake flask (0.25–5 L) was assessed. The suspension cultures were established on Murashige and Skoog medium supplemented with 10 μM 6-benzylaminopurine and 5 μM indole-3-butyric acid having a 20-days cultivation cycle. The data revealed a significantly (p ≤ 0.05) high biomass productivity (523.80 ± 5.95 g/L, fresh weight) in 0.25 L vessel with 10% inoculum at 70 rpm as compared to large (5.0 L) volume flask (310.80 ± 32.85 g/L). Interestingly, the optimum inoculum density was found to vary with the size of culture vessels. Furthermore, noticeable effects of sequential scaleup with shaking speed and inoculum density were also observed on hydrogen peroxide (stress indicator) production as well as enzymatic (catalase and phenylalanine ammonia-lyase activity) and non-enzymatic (phenolic compounds) antioxidant potential. A. benthamii cells cultivated with optimized conditions had a significant amount of red pigment, with 669.17 ± 26.71 mg dry weight/L yield of total shikonin derivatives. In conclusion, the results clearly demonstrated the likelihood of scalability using optimized process variables for the production of natural red pigments using A. benthamii cell suspension cultures.







Similar content being viewed by others
Data availability
The data presented in the article is available to access.
Abbreviations
- CAT:
-
Catalase
- DPPH:
-
2,2-Diphenyl-1-picrylhydrazyl
- DW:
-
Dry weight
- H2O2 :
-
Hydrogen peroxide
- MS:
-
Murashige and Skoog
- PAL:
-
Phenylalanine ammonia-lyase
- RGR:
-
Relative growth rate
- ROS:
-
Reactive oxygen species
- TPC:
-
Total polyphenol contents
- APM:
-
Arnebia pigment production medium
References
Alvarez-Yela AC, Chiquiza-Montano LN, Hoyos R, Orozco-Sanchez F (2016) Rheology and mixing analysis of plant cell cultures (Azadirachta indica, Borojoa patinoi and Thevetia peruviana) in shake flasks. Biochem Eng J 114:18–25. https://doi.org/10.1016/j.bej.2016.06.019
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Bagheria F, Tahviliana R, Karimi N, Chalabi M, Azami M (2018) Shikonin production by callus culture of Onosma bulbotrichom as active pharmaceutical ingredient. Iran J Pharm Res. 17:495–504
Baque MA, Shiragi MHK, Moh SH, Lee EJ, Paek KY (2013) Production of biomass and bioactive compounds by adventitious root suspension cultures of Morinda citrifolia (L.) in a liquidphase airlift balloon- type bioreactor. In Vitro Cell Dev Biol Plant 49:737–749. https://doi.org/10.1007/s11627-013-9555-3
Bisht SS, Mehrotra SC (1989) Iron cobalt interaction in growth and metabolism of maize (Zea mays). Indian J Agric Sci 59:650–654
Boudet AM (2007) Evolution and current status of research in phenolic compounds. Phytochemistry 68:2722–2735. https://doi.org/10.1016/j.phytochem.2007.06.012
Bradford MM (1976) A Rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Chan LK, Lim PS, Choo ML, Boey PL (2010) Establishment of Cyperusaromaticus cell suspension cultures for the production of juvenile hormone. In Vitr Cell Dev Biol 46:8–12. https://doi.org/10.1007/s11627-009-9240-8
Chaudhury A, Pal M (2010) Induction of shikonin production in hairy root cultures of Arnebia hispidissima via Agrobacterium rhizogenes-mediated genetic transformation. Crop Sci Biotech 13:99–106
Cui HY, Baque AMMd, Lee JE, Paek KY (2013) Scale-up of adventitious root cultures of Echinacea angustifolia in a pilot-scale bioreactor for the production of biomass and caffeic acid derivatives. Plant Biotechnol Rep 7:297–308. https://doi.org/10.1007/s11816-012-0263-y
Duetz WA, Witholt B (2004) Oxygen transfer by orbital shaking of square vessels and deepwell microtiter plates of various dimensions. Biochem Eng J 17:181–185. https://doi.org/10.1016/S1369-703X(03)00177-3
Folin O, Ciocalteu V (1927) On tyrosine and tryptophan determinations in proteins. J Biol Chem 73:627–650
Fremont F (2017) Cell culture: an innovative approach for production of plant actives. https://www.newfoodmagazine.com/article/33682/cell-culture-plant-actives/.
Fu JY, Zhao H, Bao JX, Wen ZL, Fang RJ, Fazal A, Yang MK, Liu B, Yin TM, Pang YJ, Lu GH, Qi JL, Yang YH (2020) Establishment of the hairy root culture of Echium plantagineum L. and its shikonin production. 3 Biotech. 10:429.
Gechev TS, Breusegem FV, Stone JM, Denev I, Laloi C (2006) Reactive oxygen species as signals that modulate plant stress responses and programmed cell death. Bioessay 28:1091–1101. https://doi.org/10.1002/bies.20493
Georgiev V, Slavov A, Vasileva I, Pavlov A (2018) Plant cell culture as emerging technology for production of active cosmetic ingredients. Eng Life Sci 18:779–798. https://doi.org/10.1002/elsc.201800066
Haida Z, Syahida A, Ariff SM, Mahmood M, Mansor H (2019) Factors affecting cell biomass and flavonoid production of Ficus deltoidea var. kunstleri in cell suspension culture system. Sci Rep 9:1–8. https://doi.org/10.1038/s41598-019-46042-w
Hatano T, Edamatsu R, Hiramatsu M, Mori A, Fujita Y, Yasuhara T, Yoshida T, Okuda T (1989) Effects of the interaction of tannins with co-existing substances. VI. Effects of tannins and related polyphenols on superoxide anion radical, and on 1,1-diphenyl-pierylhydrazyl radical. Chem Pharm 37:2016–2021. https://doi.org/10.1248/cpb.37.2016
Heide L, Nishioka N, Fukui H, Tabata M (1989) Enzymatic regulation of shikonin biosynthesis in Lithospermum erythrorhizon cell cultures. Phytochemistry 28:1873–1877. https://doi.org/10.1016/S0031-9422(00)97877-4
Ho TT, Lee KJ, Lee JD, Bhushan S, Paek KY, Park SY (2017) Adventitious root culture of Polygonum multiflorum for phenolic compounds and its pilot-scale production in 500 L-tank. Plant Cell Tiss Organ Cult 130:167–181. https://doi.org/10.1007/s11240-017-1212-9
Huang ML-H, Chiang S, Kalinowski DS, Bae D-H, Sahni S, Richardson DR (2019) The Role of the antioxidant response in mitochondrial dysfunction in degenerative diseases: cross-talk between antioxidant defense, autophagy, and apoptosis. Hindawi Oxid Med Cell Longev. https://doi.org/10.1155/2019/6392763
Jiménez JA, Alonso-Blázquez N, López-Vela D, Celestino C, Toribio M, Alegre J (2011) Influence of culture vessel characteristics and agitation rate on gaseous exchange, hydrodynamic stress, and growth of embryogenic cork oak (Quercus suber L.) cultures. In Vitro Cell Dev Biol Plant 47:578–588. https://doi.org/10.1007/s11627-011-9399-7
Karuppanapandian T, Moon JC, Kim C, Manoharan K, Kim W (2011) Reactive oxygen species in plants: their generation, signal transduction, and scavenging mechanisms. Aust J Crop Sci 5:709–725
Katoch P, Rana S, Kumar D, Kumar S, Bhushan S (2016) Concurrent HPTLC determination of shikonin and β, β-dimethylacryl shikonin in Arnebia benthamii. J Chromatogr Sci 54:1421–1427. https://doi.org/10.1093/chromsci/bmw096
Kumar R, Sharma N, Malik S, Bhushan S, Sharma UK, Kumari D, Sinha AK, Sharma M, Ahuja PS (2011) Cell suspension culture of Arnebia euchroma (Royle) Johnston – a potential source of naphthoquinone pigments. J Med Plant Res 5:6048–6054
Le KC, Jeong CS, Lee H, Paek KY, Park SY (2019) Ginsenoside accumulation profiles in long- and short-term cell suspension and adventitious root cultures in Panax ginseng. Hortic Environ Biotechnol 60:125–134
Lee EJ, Moh SH, Paek KY (2011) Influence of inoculum density and aeration volume on biomass and bioactive compound production in bulb-type bubble bioreactor cultures of Eleutherococcus koreanum Nakai. Bioresour Technol 102:7165–7170
Lin L, Wu J (2002) Enhancement of shikonin production in single- and two-phase suspension cultures of Lithospermum erythrorhizon cells using low-energy ultrasound. Biotechnol Bioeng 78:81–88
Malik S, Bhushan S, Verma SC, Sharma N, Sinha AK, Sharma M, Ahuja PS (2008) Production of naphthoquinone pigments in cell suspension cultures of Arnebia euchroma (Royle) Johnston: influence of pH on growth kinetics and acetylshikonin. Med Aromat Plant Sci Biotechnol 2:43–49
Malik S, Bhushan S, Sharma M, Ahuja PS (2011) Physico-chemical factors influencing the shikonin derivatives production in cell suspension cultures of Arnebia euchroma (Royle) Johnston, a medicinally important plant species. Cell Biol Int 35:153–158. https://doi.org/10.1042/CBI20090459
Manjkhola S, Dhar U (2002) Conservation and utilization of Arnebia benthamii (Wall, ex G. Don) Johnston—A high value Himalayan medicinal plant. Curr Sci 83:484–488
McElroy C, Jennewein S (2018) Taxol biosynthesis and production: from forests to fermenters. Biotechnol Nat Prod 7:145–185. https://doi.org/10.1007/978-3-319-67903-7_7
Meijer JJ, Hoopen HJGT, Luyben KCAM, Libbengat KR (1993) Effects of hydrodynamic stress on cultured plant cells: a literature survey. Enzyme Microb Technol 15:234–238. https://doi.org/10.1016/0141-0229(93)90143-P
Murthy HN, Georgiev MI, Kim YS, Jeong CS, Kim SJ, Park SY, Paek KY (2014) Ginsenosides: prospective for sustainable biotechnological production. Appl Microbiol Biotechnol 98:6243–6254
Murthy HN, Dandin VS, Park SY, Paek KY (2018) Quality, safety and efficacy profiling of ginseng adventitious roots produced in vitro. Appl Microbiol Biotechnol 102:7309–7317
Patade VY, Bhargava S, Suprasanna P (2012) Effects of NaCl and iso-osmotic PEG stress on growth, osmolytes accumulation and antioxidant defense in cultured sugarcane cells. Plant Cell Tiss Organ Cult 108:279–286. https://doi.org/10.1007/s11240-011-0041-5
Sahraroo A, Mirjalili MH, Corchete P, Babalar M, Moghadam MRF (2016) Establishment and characterization of a Satureja khuzistanica Jamzad (Lamiaceae) cell suspension culture: a new in vitro source of rosmarinic acid. Cytotechnology 68:1415–1424. https://doi.org/10.1007/s10616-015-9901-x
Sajc L, Grubisic D, Vunjak-Novakovic G (2000) Bioreactors for plant engineering: an outlook for further research. Biochem Eng J 4:89–99. https://doi.org/10.1016/S1369-703X(99)00035-2
Sergiev I, Alexieva V, Karanov E (1997) Effect of spermine, atrazine and combination between them on some endogenous protective systems and stress markers in plants. Compt Rend Acad Bulg Sci 51:1–4
Shimomura K, Sudo H, Saga H, Kamada H (1991) Shikonin production and secretion by hairy root cultures of Lithospermum erythrorhizon. Plant Cell Rep 10:282–285
Sykłowska-Baranek K, Pilarek M, Cichosz M, Pietrosiuk A (2014) Liquid perfluorodecalin application for in situ extraction and enhanced naphthoquinones production in arnebia euchroma cell suspension cultures. Appl Biochem Biotechnol 172:2618–2627. https://doi.org/10.1007/s12010-013-0701-5
Thanh NT, Murthy HN, Paek KY (2014) Optimization of ginseng cell culture in airlift bioreactors and develo** the large-scale production system. Ind Crops Prod 60:343–348. https://doi.org/10.1016/j.indcrop.2014.06.036
Trejo-Tapia G, Sepulveda-Jimenez G, Trejo-Espino J, Cerda-Garcıa-Rojas CM, Torre M, Rodrıguez-Monroy M, Ramos-Valdivia AC (2007) Hydrodynamic stress induces monoterpenoid oxindole alkaloid accumulation by Uncaria tomentosa (Wild) D. C. cell suspension cultures via oxidative burst. Biotechnol Bioeng 98:230–238. https://doi.org/10.1002/bit.21384
Vasilev N, Schmtz C, Grom** U, Fischer R, Schillberg S (2014) Assessment of cultivation factors that affect biomass and geraniol production in transgenic tobacco cell suspension cultures. PLoS ONE 9:e104620. https://doi.org/10.1371/journal.pone.0104620
Villegas-Velásquez S, Martínez-Mira AD, Hoyos R, Rojano B, Orozco-Sánchez F (2017) Hydrodynamic stress and limonoid production in Azadirachta indica cell culture. Biochem Eng J 122:75–84. https://doi.org/10.1016/j.bej.2017.03.004
Zare K, Nazemiyeh H, Movafeghi A, Khosrowshahli M, Motallebi-Azar A, Dadpour M, Omidi Y (2010) Bioprocess engineering of Echium italicum L.: induction of shikonin and alkannin derivatives by two-liquid-phase suspension cultures. Plant Cell Tiss Organ Cult 100:157–164
Zhang CH, Wu JY, He GY (2002) Effects of inoculum size and age on biomass growth and paclitaxel production of elicitor-treated taxus yunnanensis cell cultures. Appl Microbiol Biotechnol 60:396–402. https://doi.org/10.1007/s00253-002-1130-5
Zhao J, Fujita K, Sakai K (2005) Oxidative stress in plant cell culture: a role in production of β-Thujaplicin by Cupresssus lusitanica suspension culture. Biotechnol Bioeng 90:621–631. https://doi.org/10.1002/bit.20465
Acknowledgements
Authors are greatly acknowledging the guidance and support received from Director, CSIR-IHBT. RK and JD are also thankful to CSIR for Ph.D. fellowship.
Funding
The study was financially supported by Council of Scientific and Industrial Research (CSIR), Government of India vide project No. MLP-0144 and MLP 201.
Author information
Authors and Affiliations
Contributions
RK executed the experiments, collected data and written the manuscript. JD participated in data interpretation and revising of the manuscript. DK has performed the UPLC analysis. SB made substantial contributions from conception and design of this study to data interpretation and reviewing the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, R., Devi, J., Kumar, D. et al. Arnebia benthamii cell suspension cultures as a source of natural red pigments: optimization of shaking speed and inoculum density to maximize process productivity during sequential scaleup. Plant Biotechnol Rep 17, 353–367 (2023). https://doi.org/10.1007/s11816-023-00835-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-023-00835-8