Abstract
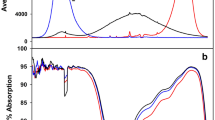
Camellia petelotii (Merr.) Sealy is a rare medicinal and edible plant in China whose wild species have a narrow distribution range and poor adaptability to strong light. To explore the light-response mechanism, the transcriptome-and photosynthesis-related physical and chemical indicators of leaves were analyzed under gradient light intensity of three processing conditions (100, 300, 700 µmol·m−2·s−1, photosynthetically active radiation, PAR) with 5 h of treatment in this study. A preliminary comparative analysis found that under strong light stress (700 PAR), the thylakoid morphology is abnormal in the leaves, the antioxidant system is damaged and the superoxide dismutase (SOD) and catalase (CAT) contents are lower than those of leaves under the 300 PAR light treatment, the synthesis of photosystem I (PSI) and photosystem II (PSII) protein members and the stability of the photosystem complex are reduced, and starch accumulation is also reduced. However, strong light stress promotes the endogenous synthesis of abscisic acid (ABA) and soluble sugar accumulation. Our research revealed that 300 PAR is a relatively appropriate growth light condition and that strong light stress (700 PAR) causes damage to the photosystem of C. petelotii. This is the first study to determine the molecular mechanism of the C. petelotii leaf responds to strong light stress at the transcription level and provides important insights for the future scientific cultivation of C. petelotii.






Similar content being viewed by others
References
Ahmed A, Nasim F, Batool K, Bibi A (2017) Microbial β-Glucosidase: sources, production and applications. J Appl Environ Microbiol 5:31–46. https://doi.org/10.12691/jaem-5-1-4
Akram NA, Iqbal M, Muhammad A, Ashraf M, Al-Qurainy F, Shafiq S (2018) Aminolaevulinic acid and nitric oxide regulate oxidative defense and secondary metabolisms in canola (Brassica napus L.) under drought stress. Protoplasma 255:163–174. https://doi.org/10.1007/s00709-017-1140-x
Andlar M, Kracher D, Rezic T, Ludwig R (2017) LPMO as a key player in the enzymatic conversion of biomass. J Biotechnol 256:S12. https://doi.org/10.1016/j.jbiotec.2017.06.043
Araújo WL, Fernie AR, Nunes-Nesi A (2011) Control of stomatal aperture: A renaissance of the old guard. Plant Signal Behav 6:1305–1311. https://doi.org/10.4161/psb.6.9.16425
Breen GAM, Jordan EM (1999) Transcriptional activation of the F1F0 ATP synthase α-subunit initiator element by USF2 is mediated by p300. Biochim Biophys Acta- Gen Subj 1428:169–176. https://doi.org/10.1016/S0304-4165(99)00061-6
Castrejón SE, Yatsimirsky AK, La S (1997) Malonaldehyde by the thiobarbituric acid method. Talanta 44:951–957. https://doi.org/10.1016/S0039-9140(96)02141-8
Chai S, Tang J, Mallik A, Shi Y, Zou R, Li J, Wei X (2018) Eco-physiological basis of shade adaptation of Camellia nitidissima, a rare and endangered forest understory plant of Southeast Asia. BMC Ecol 18:1–10. https://doi.org/10.1186/s12898-018-0159-y
Hansen J, Møller I (1975) Percolation of starch and soluble carbohydrates from plant tissue for quantitative determination with anthrone. Anal Biochem 68:87–94. https://doi.org/10.1016/0003-2697(75)90682-X
He YZ, Fan KQ, Jia CJ, Wang ZJ, Bin PW, Huang L, Yang KQ, Dong ZY (2007) Characterization of a hyperthermostable Fe-superoxide dismutase from hot spring. Appl Microbiol Biotechnol 75:367–376. https://doi.org/10.1007/s00253-006-0834-3
He X, Li H, Zhan M, Li H, Jia A, Lin S, Sun L, Du H, Yuan S, Li Y (2019) Camellia nitidissima chi extract potentiates the sensitivity of gastric cancer cells to paclitaxel via the induction of autophagy and apoptosis. Onco Targets Ther 12:10811–10825. https://doi.org/10.2147/OTT.S220453
Hou X, Du H, Yang R, Qi J, Huang Y, Feng S, Wu Y, Lin S, Liu Z, Jia AQ, Yuan S, Sun L (2018) The antitumor activity screening of chemical constituents from Camellia nitidissima Chi. Int J Mol Med 41:2793–2801. https://doi.org/10.3892/ijmm.2018.3502
Ido K, Kakiuchi S, Uno C, Nishimura T, Fukao Y, Noguchi T, Sato F, Ifuku K (2012) The conserved His-144 in the PsbP protein is important for the interaction between the PsbP N-terminus and the Cyt b559 subunit of photosystem II. J Biol Chem 287:26377–26387. https://doi.org/10.1074/jbc.M112.385286
Ihnatowicz A, Pesaresi P, Varotto C, Richly E, Schneider A, Jahns P, Salamini F, Leister D (2004) Mutants for photosystem I subunit D of Arabidopsis thaliana: Effects on photosynthesis, photosystem I stability and expression of nuclear genes for chloroplast functions. Plant J 37:839–852. https://doi.org/10.1111/j.1365-313X.2004.02011.x
Iuchi S, Kobayashi M, Taji T, Naramoto M, Seki M, Kato T, Tabata S, Kakubari Y, Yamaguchi-Shinozaki K, Shinozaki K (2001) Regulation of drought tolerance by gene manipulation of 9-cis-epoxycarotenoid dioxygenase, a key enzyme in abscisic acid biosynthesis in Arabidopsis. Plant J 27:325–333. https://doi.org/10.1046/j.1365-313X.2001.01096.x
Jang JC, León P, Zhou L, Sheen J (1997) Hexokinase as a sugar sensor in higher plants. Plant Cell 9:5–19. https://doi.org/10.1105/tpc.9.1.5
Johansson LH, Håkan Borg LA (1988) A spectrophotometric method for determination of catalase activity in small tissue samples. Anal Biochem 174:331–336. https://doi.org/10.1016/0003-2697(88)90554-4
Johnson MP, Vasilev C, Olsen JD, Hunter CN (2014) Nanodomains of cytochrome b6f and photosystem II complexes in spinach grana thylakoid membranes. Plant Cell 26:3051–3061. https://doi.org/10.1105/tpc.114.127233
Kaplan F, Guy CL (2005) RNA interference of Arabidopsis beta-amylase 8 preventsmaltose accumulation upon cold shock and increases sensitivity of PSII photochemical efficiency to freezing stress. Plant J 44:730–743. https://doi.org/10.1111/j.1365-313X.2005.02565.x
Li P, Shi C, Zheng F, Jian J, Huang B, Shan D, Shi M, Fang C, Yue Y, Li F, Li D, Wei S, Han B, Jiang C, Yin Y, **a T, Zhang Z, Bennetzen JL, Zhao S, Wan X (2018) Draft genome sequence of Camellia sinensis var. sinensis provides insights into the evolution of the tea genome and tea quality. Proc Natl Acad Sci U S A 115:E4151–E4158. https://doi.org/10.1073/pnas.1719622115
Lü J, Chen R, Zhang M, da Silva JAT, Ma G (2013) Plant regeneration via somatic embryogenesis and shoot organogenesis from immature cotyledons of Camellia nitidissima Chi. J Plant Physiol 170:1202–1211. https://doi.org/10.1016/j.jplph.2013.03.019
Ma H, Liu C, Li Z, Ran Q, **e G, Wang B, Fang S, Chu J, Zhang J (2018) ZmbZIP4 contributes to stress resistance in maize by regulating ABA synthesis and root development1. Plant Physiol 178:753–770. https://doi.org/10.1104/PP.18.00436
Mittelheuser CJ, van Steveninck RFM (1971) Rapid action of abscisic acid on photosynthesis and stomatal resistance. Planta 97:83–86. https://doi.org/10.1007/BF00388408
Mulo P, Sakurai I, Aro EM (2012) Strategies for psbA gene expression in cyanobacteria, green algae and higher plants: From transcription to PSII repair. Biochim Biophys Acta - Bioenerg 1817:247–257. https://doi.org/10.1016/j.bbabio.2011.04.011
Naz R, Bano A, Nosheen A, Yasmin H, Keyani R, Shah STA, Anwar Z, Roberts TH (2021) Induction of defense-related enzymes and enhanced disease resistance in maize against Fusarium verticillioides by seed treatment with Jacaranda mimosifolia formulations. Sci Rep 11:1–15. https://doi.org/10.1038/s41598-020-79306-x
Palma F, Birren BW, Nusbaum C, Lindblad-Toh K, Friedman N, Regev A (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652. https://doi.org/10.1038/nbt.1883
Pantin F, Monnet F, Jannaud D, Costa JM, Renaud J, Muller B, Simonneau T, Genty B (2013) The dual effect of abscisic acid on stomata. New Phytol 197:65–72. https://doi.org/10.1111/nph.12013
Park SY, Fung P, Nishimura N, Jensen DR, Fujii H, Zhao Y, Lumba S, Santiago J, Rodrigues A, Chow TFF, Alfred SE, Bonetta D, Finkelstein R, Provart NJ, Desveaux D, Rodriguez PL, McCourt P, Zhu JK, Schroeder JI, Volkman BF, Cutler SR (2009) Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 324(80):1068–1071. https://doi.org/10.1126/science.1173041
Pfister B, Zeeman SC (2016) Formation of starch in plant cells. Cell Mol Life Sci 73:2781–2807. https://doi.org/10.1007/s00018-016-2250-x
Rahire M, Laroche F, Cerutti L, Rochaix JD (2012) Identification of an OPR protein involved in the translation initiation of the PsaB subunit of photosystem i. Plant J 72:652–661. https://doi.org/10.1111/j.1365-313X.2012.05111.x
Raszewski G, Renger T (2008) Light harvesting in photosystem II core complexes is limited by the transfer to the trap: Can the core complex turn into a photoprotective mode? J Am Chem Soc 130:4431–4446. https://doi.org/10.1021/ja7099826
Rock CD, Sun X (2005) Crosstalk between ABA and auxin signaling pathways in roots of Arabidopsis thaliana (L.) Heynh. Planta 222:98–106. https://doi.org/10.1007/s00425-005-1521-9
Sharma P, Chatterjee M, Burman N, Khurana JP (2014) Cryptochrome 1 regulates growth and development in Brassica through alteration in the expression of genes involved in light, phytohormone and stress signalling. Plant Cell Environ 37:961–77. https://doi.org/10.1111/pce.12212
Sullivan MB, Lindell D, Lee JA, Thompson LR, Bielawski JP, Chisholm SW (2006) Prevalence and evolution of core photosystem II genes in marine cyanobacterial viruses and their hosts. PLoS Biol 4:1344–1357. https://doi.org/10.1371/journal.pbio.0040234
Tang S, Bin X, Wang L, Zhong Y (2006) Genetic diversity and population structure of yellow camellia (Camellia nitidissima) in China as revealed by RAPD and AFLP markers. Biochem Genet 44:449–461. https://doi.org/10.1007/s10528-006-9053-y
Taylor JS, Blake TJ, Pharis RP (1982) The role of plant hormones and carbohydrates in the growth and survival of coppiced Eucalyptus seedlings. Physiol Plant 55:421–430. https://doi.org/10.1111/j.1399-3054.1982.tb04522.x
Vaistij FE, Goldschmidt-Clermont M, Wostrikoff K, Rochaix JD (2000) Stability determinants in the chloroplast psbB/T/H mRNAs of Chlamydomonas reinhardtii. Plant J 21:469–482. https://doi.org/10.1046/j.1365-313X.2000.00700.x
Wang K, Wei X, Liu K, Li K, Ma J (2019) Response of photosynthesis characteristics and leaf anatomical structure of Vietnam Camellia hirsuta under different light environment. For Research 32:105–113. https://doi.org/10.13275/j.cnki.lykxyj.2019.04.014
Yang R, Guan Y, Zhou J, Sun B, Wang Z, Chen H, He Z, Jia A (2017) Phytochemicals from Camellia nitidissima Chi flowers reduce the Pyocyanin production and motility of Pseudomonas aeruginosa PAO1. Front Microbiol 8:1–13. https://doi.org/10.3389/fmicb.2017.02640
Yang R, Guan Y, Wang W, Chen H, He Z, Jia AQ (2018) Antioxidant capacity of phenolics in Camellia nitidissima Chi flowers and their identification by HPLC Triple TOF MS/MS. PLoS ONE 13:1–21. https://doi.org/10.1371/journal.pone.0195508
Zhang J, Li Y, Liu Y, Di B, Sun J (2004) Effects of high temperature and strong light on oxidative stress of apple fruit peel on different exposures of tree crown. Sci Agric Sin 37:1976–1980. https://doi.org/10.3321/j.issn:0578-1752.2004.12.032
Zhang W, Fan D, **e Z, Jiang X (2005) The seasonal photosynthetic responses of seedlings of the endangered plant Cathaya argyrophylla to different growth light environments. Biodivers Sci 13:387–397. https://doi.org/10.1360/biodiv.0500142
Zhang J, Chen S, Li Y, Di B, Zhang J, Liu Y (2008) Effect of high temperature and excessive light on glutathione content in apple peel. Front Agric China 2:97–102. https://doi.org/10.1007/s11703-008-0002-x
Zhang X, Feng J, Su S, Huang L (2020a) Hepatoprotective effects of Camellia nitidissima aqueous ethanol extract against CCl4-induced acute liver injury in SD rats related to Nrf2 and NF-κB signalling. Pharm Biol 58:239–246. https://doi.org/10.1080/13880209.2020.1739719
Zhang H-L, Wu Q-X, Qin X-M (2020b) Camellia nitidissima Chi flower extract alleviates obesity and related complications and modulates gut microbiota composition in rats with high-fat-diet-induced obesity. J Sci Food Agric 100:4378–4389. https://doi.org/10.1002/jsfa.10471
Zhou XW, Fan ZQ, Chen Y, Zhu YL, Li JY, Yin HF (2013) Functional analyses of a flavonol synthase-like gene from Camellia nitidissima reveal its roles in flavonoid metabolism during floral pigmentation. J Biosci 38:593–604. https://doi.org/10.1007/s12038-013-9339-2
Acknowledgements
This study was supported by the Fundamental Research Funds for Guangxi Forestry Research Institute (201827).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
All authors declare no conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11756_2022_1172_MOESM1_ESM.jpg
Supplementary file1 (JPG 283 KB) Supplement Fig. S1 Contents of carotenoids, chlorophyll a, and chlorophyll b in the leaves of Camellia petelotii (Merr.) Sealy under the treatment of E (700 PAR), M (300 PAR), and L (100 PAR) for 0, 5, and 10 hours. All bars represent the means ± s.d., and those labeled with a letter are significantly different at P < 0.05 by Duncan’s test. Three biological replicates in the experiment were performed. For each biological replication, five leaves of similar height were selected from the same Camellia petelotii (Merr.) Sealy plant
11756_2022_1172_MOESM2_ESM.jpg
Supplementary file2 (JPG 327 KB) Supplement Fig. S2 Contents of IAA, MeJA, Zeatin, MeSA, and GA3 phytohormones in the leaves of Camellia petelotii (Merr.) Sealy under the treatment of E (700 PAR), M (300 PAR), and L (100 PAR) for 0, 5, and 10 hours. All bars represent the means ± s.d., and those labeled with a letter are significantly different at P < 0.05 by Duncan’s test. Three biological replicates in the experiment were performed. For each biological replication, five leaves of similar height were selected from the same Camellia petelotii (Merr.) Sealy plant
11756_2022_1172_MOESM3_ESM.jpg
Supplementary file3 (JPG 87 KB) Supplement Fig. S3 Contents of MDA (The malondialdehyde) in the leaves of Camellia petelotii (Merr.) Sealy under the treatment of E (700 PAR), M (300 PAR), and L (100 PAR) for 0, 5, and 10 hours. All bars represent the means ± s.d., and those labeled with a letter are significantly different at P < 0.05 by Duncan’s test. Three biological replicates in the experiment were performed. For each biological replication, five leaves of similar height were selected from the same Camellia petelotii (Merr.) Sealy plant
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, X., Qin, B., **a, S. et al. A comparative study on the effects of strong light stress on the photosynthetic characteristics of the shade plant Camellia petelotii (Merr.) Sealy. Biologia 77, 3455–3468 (2022). https://doi.org/10.1007/s11756-022-01172-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01172-x