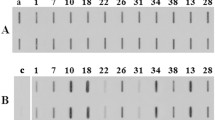
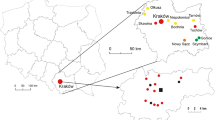
Abstract
Pollen allergies are an important and relevant question in the personal life of millions of people. They are well characterized on the genomic and proteomic level of allergens per se and on medicinal level, but the very few information exists about the natural variability and background of pollen allergen transcription patterns. Here, the variability of expression of Bet v 1, Bet v 2, Cor a 1 and Cor a 2 were performed. Both of the analysed species, Betulla verrucosa as well as Corylus avellana are clinically relevant in spring pollinosis in the area of central Europe. Bet v 1 and Bet v 2 expressions were subjected to compare in pollen for a total of twelve accessions from different areas. Cor a 1 and Cor a 2 expressions were subjected to comparison in pollen for a total of thirty-one natural pollen germplasm accessions. Expression changes of Bet v 1 were changed in three different manners – in six pollen samples was the expression higher than 1.5, three pollen samples have no relevant change and in three pollen samples was the expression lower about 0.5 times. Expression changes of Bet v 2 were as follows - five of the analysed samples were expressed more than 3 times when compared to the calibrator and in all of the other samples was the expression change in the difference that was not above 0.7 times. Expression changes of Cor a 1 were very similar among themselves and only in five samples was expression relevantly higher with at least 1.1 fold change. Expression changes of Cor a 2 were very similar among themselves and only in four samples was expression relevantly higher with at least 1.4 fold change.




Similar content being viewed by others
References
Aman S, Haq NU, Shakeel SN (2012) Identification and validation of stable internal control for heat induced gene expression of Agave Americana. Pak J Bot 44:1289–1296
Asam C, Hofer H, Wolf M, Aglas L, Wallner M (2015) Tree pollen allergens – an update from a molecular perspective. Allergy 70:1201–1211. https://doi.org/10.1111/all.12696
Behrendt H, Becker WM (2001) Localization, release and bioavailability of pollen allergens: the influence of environmental factors. Curr Opin Immunol 13:709–715. https://doi.org/10.1016/S0952-7915(01)00283-7
Bousquet PJ, Chinn S, Janson C, Kogevinas M, Burney P, Jarvis D (2007) Geographical variation in the prevalence of positive skin tests to environmental aeroallergens in the European Community respiratory health survey I. Allergy 62:301–309. https://doi.org/10.1111/j.1398-9995.2006.01293.x
Breiteneder H, Ferreira F, Hoffmann-Sommergruber K, Ebner C, Breitenbach M, Kraft D, Scheiner O (1993) Four recombinant isoforms of Cor a 1, the major allergen of hazel pollen, show different IgE-binding properties. Eur J Biochem 212:355–362. https://doi.org/10.1111/j.1432-1033.1993.tb17669.x
Bryce M, Drews O, Schenk MF, Menzel A, Estrella N, Weichenmeier I, Smulders MJM, Buters J, Ring J, Görg A, Behrendt H, Traidl-Hoffmann C (2010) Impact of urbanization on the proteome of birch pollen and its chemotactic activity on human granulocytes. Int Arch Allergy Immunol 151:46–55. https://doi.org/10.1159/000232570
Buters JTM, Kasche A, Weichenmeier I, Schober W, Klaus S, Traidl-Hoffmann C, Menzel A, Huss-Marp J, Kramer U, Behrendt H (2008) Year-to-year variation in release of bet v 1 allergen from birch pollen: evidence for geographical differences between west and South Germany. Int Arch Allergy Immunol 145:122–130. https://doi.org/10.1159/000108137
Buters JTM, Weichenmeier I, Ochs S, Pusch G, Kreyling W, Boere AJF, Schober W, Behrendt H (2010) The allergen bet v 1 in fractions of ambient air deviates from birch pollen counts. Allergy 65:850–858. https://doi.org/10.1111/j.1398-9995.2009.02286.x
Buters JTM, Thibaudon M, Smith M, Kennedy R, Rantio-Lehtimäki A, Albertini R, Reese G, Weber B, Galan C, Brandao R, Antunes CM, Jäger S, Berger U, Celenk S, Grewling L, Jackowiak B, Sauliene I, Weichenmeier I, Cecchio L (2012) Release of bet v 1 from birch pollen from 5 European countries. Results from the HIALINE study. Atmos Environ 55:496–505. https://doi.org/10.1016/j.atmosenv.2012.01.054
Ciprandi G, Comite P, Mussap M, De Amici M, Quaglini S, Barocci F, Marseglia GL, Scala E (2016) Profiles of Birch Sensitization (Bet v 1, Bet v 2, and Bet v 4) and Oral Allergy Syndrome Across Italy. J Investig Allergol Clin Immunol 26:244–248. https://doi.org/10.18176/jiaci.0041
D’Amato G, Liccardi G, D’Amato M, Holgatew S (2005) Environmental risk factors and allergic bronchial asthma. Clin Expl Allergy 35:1113–1124. https://doi.org/10.1111/j.1365-2222.2005.02328.x
D’Amato G, Cecchi L, Bonini S, Nunes C, Annesi-Maesano I, Behrendt H, Liccardi G, Popov T, van Cauwenberge P (2007) Allergenic pollen and pollen allergy in Europe. Allergy 62:976–990. https://doi.org/10.1111/j.1398-9995.2007.01393.x
D’Amato G, Cecchi L, D’Amato M, Liccardi G (2010) Urban air pollution and climate change as environmental risk factors of respiratory allergy: an update. J Investig Allergol Clin Immunol 20:95–102
D'Amato G, Vitale C, De Martino A, Viegi G, Lanza M, Molino A, Sanduzzi A, Vatrella A, Annesi-Maesano I, D'Amato M (2015) Effects on asthma and respiratory allergy of climate change and air pollution. Multidiscip Respir Med 10:39. https://doi.org/10.1186/s40248-015-0036-x
Eder W, Ege MJ, von Mutius E (2006) The asthma epidemic. N Engl J Med 355:2226–2235. https://doi.org/10.1056/NEJMra054308
Erler A, Hawranek T, Krückemeier L, Asam C, Egger M, Ferreira F, Briza P (2011) Proteomic profiling of birch (Betula verrucosa) pollen extracts from different origins. Proteomics 11:1486–1489. https://doi.org/10.1002/pmic.201000624
Gilmour MI, Jaakkola MS, London SJ, Nel AE, Rogers CA (2006) How exposure to environmental tobacco smoke, outdoor air pollutants, and increased pollen burdens influences the incidence of asthma. Environ Health Perspect 114:627–633. https://doi.org/10.1289/ehp.8380
Grote M, Valenta R, Reichelt R (2003) Abortive pollen germination: a mechanism of allergen release in birch, alder and hazel revealed by immunogold electron microscopy. J Allergy Clin Immunol 111:1017–1023. https://doi.org/10.1067/mai.2003.1452
Ianovici N (2007) The principal airborne and allergenic pollen species in Timisoara. Ann West Univ Timiş, Ser Biol 10:11–26
Kazemi-Shirazi L, Pauli G, Purohit A, Spitzauer S, Froschl R, Hoffmann-Sommergruber K, Breiteneder H, Scheiner O, Kraft D, Valenta R (2000) Quantitative IgE-inhibition experiments with purified recombinant allergens as the sensitizing agents responsible for many forms of food allergy. J Allergy Clin Immunol 105:116–125. https://doi.org/10.1016/S0091-6749(00)90186-6
Koloušková P, Stone JD, Štorchová H (2017) Evaluation of reference genes for reverse transcription quantitative real-time PCR (RT-qPCR) studies in Silene vulgaris considering the method of cDNA preparation. PLoS One. https://doi.org/10.1371/journal.pone.0183470
Kozera B, Rapacz M (2013) Reference genes in real-time PCR. J Appl Genet 54:391–406. https://doi.org/10.1007/s13353-013-0173-x
Laurent GS, Shtokalo D, Tackett MR, Yang Z, Vyatkin Y, Milos PM, Seilheimer B, McCaffrey TA, Kapranov P (2013) On the importance of small changes in RNA expression. Methods. https://doi.org/10.1016/j.ymeth.2013.03.027
Leister D, Wang L, Kleine T (2017) Organellar gene expression and acclimation of plants to environmental stress. Front Plant Sci 8:387. https://doi.org/10.3389/fpls.2017.00387
Liang C, Hao J, Meng Y, Luo L, Li L (2018) Identifying optimal reference genes for the normalization of microRNA expression in cucumber under viral stress. PLoS One. https://doi.org/10.1371/journal.pone.0194436
Liscombe DK, MacLeod BP, Loukanina N (2005) Evidence for the monophyletic evolution of benzylisoquinoline alkaloid biosynthesis in angiosperms. Phytochem 66:2501–2520
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(Delta Delta C(T)) method. Methods 25:402–408
Longhi S, Cristofori A, Gatto P, Cristofolini F, Grando MS, Gottardini E (2009) Biomolecular identification of allergenic pollen: a new perspective for aerobiological monitoring? Ann Allergy Asthma Immunol 103:508–514. https://doi.org/10.1016/S1081-1206(10)60268-2
Lüttkopf D, Müller U, Skov PS, Ballmer-Weber BK, Wüthrich B, Skamstrup Hansen K, Poulsen LK, Kästner M, Haustein D, Vieths S (2002) Comparison of four variants of a major allergen in hazelnut (Corylus avellana) Cor a 1.04 with the major hazel pollen allergen Cor a 1.01. Mol Immunol 38:515–525
Mari A, Wallner M, Ferreira F (2003) Fagales pollen sensitization in a birch-free area: a respiratory cohort survey using Fagales pollen extracts and birch recombinant allergens. Clin Exp Allergy 33:1419–1428. https://doi.org/10.1046/j.1365-2222.2003.01773.x
Markovic-Housley Z, Degano M, Lamba D (2003) Crystal structure of a hypoallergenic isoform of the major birch pollen allergen bet v 1 and its likely biological function as a plant steroid carrier. J Mol Biol 325:123–133. https://doi.org/10.1016/S0022-2836(02)01197-X
Mogensen JE, Ferreras M, Wimmer R, Petersen SV, Enghild JJ, Otzen DE (2007) The major allergen from birch tree pollen, bet v 1, binds and permeabilizes membranes. Biochem 46:3356–3365. https://doi.org/10.1021/bi062058h
Moverare R, Westritschnig K, Svensson M, Hayek B, Bende M, Pauli G, Sorva R, Haahtela T, Valenta R, Elfman L (2002) Different IgE reactivity profiles in birch pollen-sensitive patients from six European populations revealed by recombinant allergens: an Imprit of local sensitization. Int Arch Allergy Immunol 128:325–335. https://doi.org/10.1159/000063855
Ortolani C, Ballmer-Weber B, Hansen K, Ispano M, Wuthrich B, Bindslev-Jensen C, Ansaloni R, Vannucci L, Pravettoni V, Scibilia J, Poulsen LK, Pastorello EA (2000) Hazelnut allergy: a double-blind placebo-controlled food challenge multicenter study. J Allergy Clin Immunol 105:577–581. https://doi.org/10.1067/mai.2000.103052
Pablos I, Wildner S, Asam C, Wallner M, Gadermaier G (2016) Pollen Allergens for Molecular Diagnosis. Curr Allergy Asthma Rep 16:31. https://doi.org/10.1007/s11882-016-0603-z
Pesti R, Kontra L, Paul K, Vass I, Csorba T, Havelda Z, Várallyay E (2019) Differential gene expression and physiological changes during acute or persistent plant virus interactions may contribute to viral symptom differences. PLoS One 14:e0216618. https://doi.org/10.1371/journal.pone.0216618
Prado N, De Linares C, Sanz ML, Gamboa P, Villalba M, Rodríguez R, Batanero E (2015) Pollensomes as natural vehicles for pollen allergens. J Immunol 195:445–449. https://doi.org/10.4049/jimmunol.1500452
Puc M (2003) Characterization of pollen allergens. An Agric Environ Med 10:143–149
Ražná K, Bežo M, Nikolaieva N, Garkava K, Brindza J, Žiarovská J (2014) Variability of Corylus avellana, L. CorA and profilin pollen allergens expression. J Environ Sci Health B 49:639–645. https://doi.org/10.1080/03601234.2014.922403
Rossi RE, Monasterolo G, Monasterolo S (2003) Detection of specific IgE antibodies in the sera of patients allergic to birch pollen using recombinant allergens bet v 1, bet v 2, bet v 4: evaluation of different IgE reactivity profiles. Allergy 55:929–932. https://doi.org/10.1034/j.1398-9995.2003.00245.x
Samanani N, Liscombe DK, Facchini PJ (2004) Molecular cloning and characterization of norcoclaurine synthase, an enzyme catalyzing the first committed step in benzylisoquinoline alkaloid biosynthesis. Plant J 40:302–313. https://doi.org/10.1111/j.1365-313X.2004.02210.x
Ščevková J, Dušička J, Hrubiško M, Mičieta K (2015) Influence of airborne pollen counts and length of pollen season of selected allergenic plants on the concentration of sIgE antibodies on the population of Bratislava, Slovakia. Ann Agric Environ Med 22:451–455. https://doi.org/10.5604/12321966.1167712
Sekerková A, Polačková M (2011) Detection of bet v 1, bet v 2 and bet v 4 specific IgE antibodies in the sera of children and adult patients allergic to birch pollen: evaluation of different IgE reactivity profiles depending on age and local sensitization. Int Arch Allergy Immunol 154:278–285. https://doi.org/10.1159/000321819
Spelleberg IF, Eriksson NE, Crump VSA (2006) Silver birch (Betula pendula) pollen and human health: problems for an exotic tree in New Zealand. AUF 32(4):133–137
Swoboda I, Jilek A, Ferreira F, Engel E, Hoffmann-Sommergruber K, Scheiner O, Kraft D, Breiteneder H, Pittenauer E, Schmid E, Vicente O, Heberle-Bors E, Ahorn H, Breitenbach M (1995) Isoforms of bet v 1, the major birch pollen allergen, analyzed by liquid chromatography, mass spectrometry, and cDNA cloning. J Biol Chem 10:2607–2613. https://doi.org/10.1074/jbc.270.6.2607
Taylor PE, Jonsson H (2004) Thunderstorm asthma. Curr Allergy Asthma Rep 4:409–413
Tian C, Jiang Q, Wang F, Wang GL, Xu ZS, **ong AS (2015) Selection of suitable reference genes for qPCR normalization under abiotic stresses and hormone stimuli in carrot leaves. PLoS One 10. https://doi.org/10.1371/journal.pone.0117569
Valenta R, Duchene M, Petterburger K, Sillaber C, Valent P, Bettelheim P, Breitenbach M, Rumpold H, Kraft D, Scheiner O (1991) Identification of profilin as a novel pollen allergen: IgE autoreactivity in sensitized individuals. Science 253:557–560. https://doi.org/10.1126/science.1857985
Van Rhijn BD, van Ree R, Versteeg SA, Vlieg-Boestra BJ, Sprikkelman AB, Tereehorst I, Smout AJPM, Bredenoord AJ (2013) Birch pollen sensitization with cross-reactivity to food allergens predominates in adults with eaosinophilic esophagitis. Allergy 68:1475–1481. https://doi.org/10.1111/all.12257
Vaverková MD, Adamcová D, Winkler J, Koda E, Červenková J, Podlasek A (2019) Influence of a municipal solid waste landfill on the surrounding environment: landfill vegetation as a potential risk of allergenc pollen. Int J Environ Res Public Health 16:5064. https://doi.org/10.3390/ijerph16245064
Vieths S, Scheurer S, Ballmer-Weber B (2002) Current understanding of cross-reactivity of food allergens and pollen. Ann N Y Acad Sci 964:47–68. https://doi.org/10.1111/j.1749-6632.2002.tb04132.x
Weerd NA, de Bhalla PL, Singh MB (2002) Aeroallergens and pollinosis: molecular and immunological characteristics of cloned pollen allergens. Aerobiologia 18:87–106
Weryszko-Chmielewska E, Puc M, Rapiejko P (2001) Comparative analysis of pollen counts of Corylus, Alnus and Betula in Szczecin, Warsaw and Lublin (2000-2001). Ann Agric Environ Med 8:235–240
Wimmer M, Alessandrini F, Gilles S, Frank U, Oeder S, Hauser M, Ring J, Ferreira F, Ernst D, Winkler JB, Schmitt-Kopplin P, Ohnmacht C, Behrendt H, Schmidt-Weber C, Traidl-Hoffmann C, Gutermuth J (2015) Pollen-derived adenosine is a necessary cofactor for ragweed allergy. Allergy 70:944–954. https://doi.org/10.1111/all.12642
Ye J, Coulouris G, Zaretskaya I, Cutcutache I, Rozen S, Maddenet TL (2012) Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics 8:134. https://doi.org/10.1186/1471-2105-13-134
Žiarovská J, Labajová M, Ražná K, Bežo M, Štefúnová V, Shevtsova T, Garkava K, Brindza J (2013) Changes in expression of BetV1 allergen of silver birch pollen in urbanized area of Ukraine. J Env Sci Health 48:1479–1484. https://doi.org/10.1080/10934529.2013.796788
Acknowledgments
This publication was supported by the Operational program Integrated Infrastructure within the project: Demand-driven research for the sustainable and inovative food, Drive4SIFood 313011 V336, cofinanced by the European Regional Development Fund. The authors would like to thanks to associated professor Ján Brindza for the organizing of the pollen samples collections and providing them.
Author information
Authors and Affiliations
Contributions
JŽ carried out all the data analysis and wrote this manuscript; NN, JŽ, EF and ML collected the samples of pollen; JŽ, EF and KR conceived the project and drafted the manuscript; JŽ and LU carried out the experiment of qRT-PCR; JŽ supervised the analysis and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Žiarovská, J., Urbanová, L., Fernández-Cusimamani, E. et al. Variability in expression profiles of Betulaceae spring pollen allergens in Central Europe region. Biologia 76, 2349–2358 (2021). https://doi.org/10.1007/s11756-021-00744-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-021-00744-7