Abstract
Objective
To investigate the molecular mechanisms underlying the effect of baicalin on prostate cancer (PCa) progression both in vivo and in vitro.
Methods
The in situ PCa stem cells (PCSCs)-injected xenograft tumor models were established in BALB/c nude mice. Tumor volume and weight were respectively checked after baicalin (100 mg/kg) treatment. Hematoxylin-eosin (HE) staining was used to observe the growth arrest and cell necrosis. mRNA expression levels of acetaldehyde dehydrogenase 1 (ALDH1), CD44, CD133 and Notch1 were determined by reverse transcription-polymerase chain reaction. Protein expression levels of ALDH1, CD44, CD133, Notch1, nuclear factor κB (NF-κB) P65 and NF-κB p-P65 were detected by Western blot. Expression and subcellular location of ALDH1, CD44, CD133, Notch1 and NF-κB p65 were detected by immunofluorescence analysis. In vitro, cell cycle distribution and cell apoptosis of PC3 PCSCs was assessed by flow cytometry after baicalin (125 µmol/L) treatment. The migration and invasion abilities of PCSCs were assessed using Transwell assays. Transmission electron microscopy scanning was utilized to observe the structure and autophagosome formation of baicalin-treated PCSCs. In addition, PCSCs were infected with lentiviruses expressing human Notch1.
Results
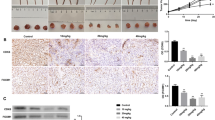
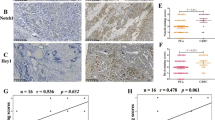
Compared with the control group, the tumor volume and weight were notably reduced in mice treated with 100 mg/kg baicalin (P<0.05 or P<0.01). Histopathological analysis showed that baicalin treatment significantly inhibited cell proliferation and promoted cell apoptosis. Furthermore, baicalin treatment reduced mRNA and protein expression levels of CD44, CD133, ALDH1, and Notch1 as well as the protein expression of NF-κB p-P65 in the xenograft tumor (P<0.01). In vitro, the cell proliferation of PCSCs was significantly attenuated after treatment with 125 µmol/L baicalin for 72 h (P<0.01). The cell migration and invasion rates were decreased following treatment with baicalin for 48 and 72 h (P<0.01). Baicalin notably induced cell apoptosis and seriously damaged the structure of PCSCs. The mRNA and protein expressions of CD133, CD44, ALDH1 and Notch1 in PCSCs were significantly downregulated following baicalin treatment (P<0.01). Importantly, the inhibitory effects of baicalin on PCa progression and stemness were reversed by Notch1 overexpression (P<0.05 or P<0.01).
Conclusion
Mechanistically, baicalin exhibited a potential therapeutic effect on PCa via inhibiting the Notch1/NF-κB signaling pathway and its mediated cancer stemness.
Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin 2019;69:7–34.
Gupta S, Pungsrinont T, Zenata O, Neubert L, Vrzal R, Baniahmad A. Interleukin-23 represses the level of cell senescence induced by the androgen receptor antagonists enzalutamide and darolutamide in castration-resistant prostate cancer cells. Horm Cancer 2020;11:182–190.
Escudero-Lourdes C, Alvarado-Morales I, Tokar EJ. Stem cells as target for prostate cancer therapy: opportunities and challenges. Stem Cell Rev Rep 2022;18:2833–2851.
Leao R, Domingos C, Figueiredo A, Hamilton R, Tabori U, Castelo-Branco P. Cancer stem cells in prostate cancer: implications for targeted therapy. Urol Int 2017;99:125–136.
Bahmad HF, Cheaito K, Chalhoub RM, Hadadeh O, Monzer A, Ballout F, et al. Sphere-formation assay: three-dimensional in vitro culturing of prostate cancer stem/progenitor sphere-forming cells. Front Oncol 2018;8:347.
Di Stefano C, Grazioli P, Fontanella RA, De Cesaris P, D’Amore A, Regno M, et al. Stem-like and highly invasive prostate cancer cells expressing CD44v8-10 marker originate from CD44-negative cells. Oncotarget 2018;9:30905–30918.
Bahmad HF, Jalloul M, Azar J, Moubarak MM, Samad TA, Mukherji D, et al. Tumor microenvironment in prostate cancer: toward identification of novel molecular biomarkers for diagnosis, prognosis, and therapy development. Front Genet 2021;12:652747.
Harris KS, Kerr BA. Prostate cancer stem cell markers drive progression, therapeutic resistance, and bone metastasis. Stem Cells Int 2017;2017:8629234.
Gong WY, Zhao ZX, Liu BJ, Lu LW, Dong JC. Exploring the chemopreventive properties and perspectives of baicalin and its aglycone baicalein in solid tumors. Eur J Med Chem 2017;126:844–852.
Singh S, Meena A, Luqman S. Baicalin mediated regulation of key signaling pathways in cancer. Pharmacol Res 2021;164:105387.
Yu ZQ, Zhan CS, Du HX, Zhang LG, Liang CZ, Zhang L. Baicalin suppresses the cell cycle progression and proliferation of prostate cancer cells through the CDK6/FOXM1 axis. Mol Cell Biochem 2020;469:169–178.
Hu XJ, Cheng YL, Kang HN, Li SX, Wang YW, Liu JZ, et al. Electroacupuncture attenuates chronic sal**itis transforming growth factor-beta1/p38 mitogen-activated protein kinase signaling pathway. J Tradit Chin Med 2022;42:781–787.
Thitiset T, Damrongsakkul S, Yodmuang S, Leeanansaksiri W, Apinun J, Honsawek S. A novel gelatin/chitooligosaccharide/demineralized bone matrix composite scaffold and periosteum-derived mesenchymal stem cells for bone tissue engineering. Biomater Res 2021;25:19.
Sui X, Han X, Chen P, Wu Q, Feng J, Duan T, et al. Baicalin induces apoptosis and suppresses the cell cycle progression of lung cancer cells through downregulating Akt/mTOR signaling pathway. Front Mol Biosci 2020;7:602282.
Kong N, Chen X, Feng J, Duan T, Liu S, Sun X, et al. Baicalin induces ferroptosis in bladder cancer cells by downregulating FTH1. Acta Pharm Sin B 2021;11:4045–4054.
Li Y, Wang D, Liu J, Chen D, Zhou L, Lang T, et al. Baicalin attenuates yap activity to suppress ovarian cancer stemness. Onco Targets Ther 2020;13:7151–7163.
**n L. Cells of origin for prostate cancer. Adv Exp Med Biol 2019;1210:67–86.
Cheaito KA, Bahmad HF, Hadadeh O, Saleh E, Dagher C, Hammoud MS, et al. EMT markers in locally-advanced prostate cancer: predicting recurrence? Front Oncol 2019;9:131.
Tsao T, Beretov J, Ni J, Bai X, Bucci J, Graham P, et al. Cancer stem cells in prostate cancer radioresistance. Cancer Lett 2019;465:94–104.
Mawaribuchi S, Onuma Y, Aiki Y, Kuriyama Y, Mutoh M, Fujii G, et al. The rBC2LCN-positive subpopulation of PC-3 cells exhibits cancer stemlike properties. Biochem Biophys Res Commun 2019;515:176–182.
Hu J, Sun F, Chen W, Zhang J, Zhang T, Qi M, et al. BTF3 sustains cancer stem-like phenotype of prostate cancer via stabilization of BMI1. J Exp Clin Cancer Res 2019;38:227.
Simeckova S, Kahounova Z, Fedr R, Remsik J, Slabakova E, Suchankova T, et al. High Skp2 expression is associated with a mesenchymal phenotype and increased tumorigenic potential of prostate cancer cells. Sci Rep 2019;9:5695.
Yasumizu Y, Rajabi H, ** C, Hata T, Pitroda S, Long MD, et al. MUC1-C regulates lineage plasticity driving progression to neuroendocrine prostate cancer. Nat Commun 2020;11:338.
Li JJ, Shen MM. Prostate stem cells and cancer stem cells. Cold Spring Harb Perspect Med 2019;9:a030395.
Hofer P, Zochmeister C, Behm C, Brezina S, Baierl A, Doriguzzi A, et al. MNS16A tandem repeat minisatellite of human telomerase gene: functional studies in colorectal, lung and prostate cancer. Oncotarget 2017;8:28021–28027.
Tong D. Unravelling the molecular mechanisms of prostate cancer evolution from genotype to phenotype. Crit Rev Oncol Hematol 2021;163:103370.
Wu D, Ke Y, **ao R, Liu J, Li Q, Wang Y. Long non-coding RNA GClnc1 knockdown suppresses progression of epithelial ovarian cancer by recruiting FOXC2 to disrupt the NOTCH1/NF-kappaB/Snail pathway. Exp Cell Res 2021;399:112422.
Wu W, Nie L, Zhang L, Li Y. The notch pathway promotes NF-kappaB activation through Asb2 in T cell acute lymphoblastic leukemia cells. Cell Mol Biol Lett 2018;23:37.
Okasha SM, Itoh M, Tohda S. Sirtuin 1 activation suppresses the growth of T-lymphoblastic leukemia cells by inhibiting NOTCH and NF-kappaB pathways. Anticancer Res 2020;40:3155–3161.
Quan XX, Hawk NV, Chen W, Coupar J, Lee SK, Petersen DW, et al. Targeting Notch1 and IKKalpha enhanced NF-kappaB activation in CD133+ skin cancer stem cells. Mol Cancer Ther 2018;17:2034–2048.
Yang L, Dong Y, Li Y, Wang D, Liu S, Gao Q, et al. IL-10 derived from M2 macrophage promotes cancer stemness via JAK1/STAT1/NF-kappaB/Notch1 pathway in non-small cell lung cancer. Int J Cancer 2019;145:1099–1110.
Cai C, Wu Q, Hong H, He L, Liu Z, Gu Y, et al. In silico identification of natural products from traditional Chinese medicine for cancer immunotherapy. Sci Rep 2021;11:3332.
Hu Q, Zhang W, Wu Z, Tian X, **ang J, Li L, et al. Baicalin and the liver-gut system: pharmacological bases explaining its therapeutic effects. Pharmacol Res 2021;165:105444.
Avila-Carrasco L, Majano P, Sanchez-Tomero JA, Selgas R, Lopez-Cabrera M, Aguilera A, et al. Natural plants compounds as modulators of epithelial-to-mesenchymal transition. Front Pharmacol 2019;10:715.
Mirzaei S, Zarrabi A, Asnaf SE, Hashemi F, Zabolian A, Hushmandi K, et al. The role of microRNA-338-3p in cancer: growth, invasion, chemoresistance, and mediators. Life Sci 2021;268:119005.
Yang B, Bai H, Sa Y, Zhu P, Liu P. Inhibiting EMT, stemness and cell cycle involved in baicalin-induced growth inhibition and apoptosis in colorectal cancer cells. J Cancer 2020;11:2303–2317.
Author information
Authors and Affiliations
Contributions
Wu MH performed the experiments and wrote the paper. Wu K performed the experiments and analyzed the data. Zhu YB, Li DC and Yang H performed the experiments. Zeng H designed the study, reviewed the paper and provided the funding. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
The authors declare that they have no competing interests.
Additional information
Supported by the Science and Health Joint Chinese Medical Research Project of Chongqing Municipal Health Committee (No. 2019ZY023447)
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wu, Mh., Wu, K., Zhu, Yb. et al. Baicalin Antagonizes Prostate Cancer Stemness via Inhibiting Notch1/NF-κB Signaling Pathway. Chin. J. Integr. Med. 29, 914–923 (2023). https://doi.org/10.1007/s11655-023-3595-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-023-3595-2