Abstract
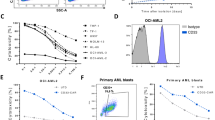
Chimeric antigen receptors (CARs) offer a promising new approach for targeting B cell malignancies through the immune system. Despite the proven effectiveness of CAR T cells targeting CD19 and CD22 in hematological malignancies, it is imperative to note that their production remains a highly complex process. Unlike T cells, NK cells eliminate targets in a non-antigen-specific manner while avoiding graft vs. host disease (GvHD). CAR-NK cells are considered safer than CAR-T cells because they have a shorter lifespan and produce less toxic cytokines. Due to their unlimited ability to proliferate in vitro, NK-92 cells can be used as a source for CAR-engineered NK cells. We found that CARs created from the m971 anti-CD22 mAb, which specifically targets a proximal CD22 epitope, were more effective at anti-leukemic activity compared to those made with other binding domains. To further enhance the anti-leukemic capacity of NK cells, we used lentiviral transduction to generate the m971-CD28-CD3ζ NK-92. CD22 is highly expressed in B cell lymphoma. To evaluate the potential of targeting CD22, Raji cells were selected as CD22-positive cells. Our study aimed to investigate CD22 as a potential target for CAR-NK-92 therapy in the treatment of B cell lymphoma. We first generated m971-CD28-CD3ζ NK-92 that expressed a CAR for binding CD22 in vitro. Flow cytometric analysis was used to evaluate the expression of CAR. The 7AAD determined the cytotoxicity of the m971-CD28-CD3ζ NK-92 towards target lymphoma cell lines by flow cytometry assay. The ELISA assay evaluated cytokine production in CAR NK-92 cells in response to target cells. The m971-CD28-CD3ζ NK-92 cells have successfully expressed the CD22-specific CAR. m971-CD28-CD3ζ NK-92 cells efficiently lysed CD22-expressing lymphoma cell lines and produced large amounts of cytokines such as IFN-γ and GM-CSF but a lower level of IL-6 after coculturing with target cells. Based on our results, it is evident that transferring m971-CD28-CD3ζ NK-92 cells could be a promising immunotherapy for B cell lymphoma.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- CARs :
-
Chimeric antigen receptors
- GvHD :
-
Graft vs. host disease
- scFv :
-
Single-chain variable fragment
- NK :
-
Natural killer
- CRS :
-
Cytokine release syndrome
- GM-CSF :
-
Granulocyte-macrophage colony-stimulating factor
- NCBI :
-
National Cell Bank of Iran
- HEK293T :
-
Human embryonic kidney cells
- IBRC :
-
Iranian Biological Resource Center
- DMEM :
-
Dulbeccoʼs modified Eagleʼs medium
- FBS :
-
Fetal bovine serum
- PCR :
-
Polymerase chain reaction
- MOI :
-
Multiplicity of infection
- ELISA :
-
Enzyme-linked immunosorbent assay
- TH-1 :
-
T helper-1
- TNFα :
-
Tumor necrosis factor α
References
Basar R, Daher M, Rezvani K (2020) Next-generation cell therapies: the emerging role of CAR-NK cells. Hematology 2020:570–578. https://doi.org/10.1182/hematology.2020002547
Bashiri Dezfouli A, Yazdi M, Pockley AG et al (2021) NK cells armed with chimeric antigen receptors (CAR): roadblocks to successful development. Cells 10:3390. https://doi.org/10.3390/cells10123390
Colamartino ABL, Lemieux W, Bifsha P et al (2019) Efficient and robust NK-cell transduction with baboon envelope pseudotyped lentivector. Front Immunol 10:1–7. https://doi.org/10.3389/fimmu.2019.02873
Daher M, Melo Garcia L, Li Y, Rezvani K (2021) CAR-NK cells: the next wave of cellular therapy for cancer. Clin Transl Immunol 10:1–16. https://doi.org/10.1002/cti2.1274
Daher M, Rezvani K (2021) Outlook for new CAR-based therapies with a focus on CAR NK cells: what lies beyond CAR-engineered T cells in the race against cancer. Cancer Discov 11:45–58. https://doi.org/10.1158/2159-8290.CD-20-0556
Fujiwara K, Masutani M, Tachibana M, Okada N (2020) Impact of scFv structure in chimeric antigen receptor on receptor expression efficiency and antigen recognition properties. Biochem Biophys Res Commun 527:350–357. https://doi.org/10.1016/j.bbrc.2020.03.071
Gheidari F, Arefian E, Adegani FJ et al (2021) The miR-142 suppresses U-87 glioblastoma cell growth by targeting EGFR oncogenic signaling pathway. Iran J Pharm Res 20:202–212. https://doi.org/10.22037/ijpr.2021.115089.15193
Gheidari F, Arefian E, Saadatpour F et al (2022) The miR-429 suppresses proliferation and migration in glioblastoma cells and induces cell-cycle arrest and apoptosis via modulating several target genes of ERBB signaling pathway. Mol Biol Rep 49:11855–11866. https://doi.org/10.1007/s11033-022-07903-2
Grote S, Mittelstaet J, Baden C et al (2020) Adapter chimeric antigen receptor (AdCAR)-engineered NK-92 cells: an off-the-shelf cellular therapeutic for universal tumor targeting. Oncoimmunology 9:1–11. https://doi.org/10.1080/2162402X.2020.1825177
Guo Z, Tu S, Yu S et al (2021) Preclinical and clinical advances in dual-target chimeric antigen receptor therapy for hematological malignancies. Cancer Sci 112:1357–1368. https://doi.org/10.1111/cas.14799
Han C, Jiang Y, Wang Z, Wang H (2019) Natural killer cells involved in tumour immune escape of hepatocellular carcinomar. Int Immunopharmacol 73:10–16. https://doi.org/10.1016/j.intimp.2019.04.057
Haso W, Lee DW, Shah NN et al (2013) Anti-CD22-chimeric antigen receptors targeting B-cell precursor acute lymphoblastic leukemia. Blood 121:1165–1171. https://doi.org/10.1182/blood-2012-06-438002
Heipertz EL, Zynda ER, Stav-Noraas TE et al (2021) Current perspectives on “off-the-shelf” allogeneic NK and CAR-NK cell therapies. Front Immunol 12:. https://doi.org/10.3389/fimmu.2021.732135
Herrera L, Juan M, Eguizabal C (2020) Purification, culture, and CD19-CAR lentiviral transduction of adult and umbilical cord blood NK cells. Curr Protoc Immunol 131:2–13. https://doi.org/10.1002/cpim.108
Kalos M, Levine BL, Porter DL et al (2011) T cells with chimeric antigen receptors have potent antitumor effects and can establish memory in patients with advanced leukemia. Sci Transl Med 3:. https://doi.org/10.1126/scitranslmed.3002842
Kawalekar OU, O’Connor RS, Fraietta JA et al (2016) Distinct signaling of coreceptors regulates specific metabolism pathways and impacts memory development in CAR T cells. Immunity 44:380–390. https://doi.org/10.1016/j.immuni.2016.01.021
Khawar MB, Sun H (2021) CAR-NK cells: from natural basis to design for kill. Front Immunol 12:1–22. https://doi.org/10.3389/fimmu.2021.707542
Klingemann H (2014) Are natural killer cells superior CAR drivers? Oncoimmunology 3:37–41. https://doi.org/10.4161/onci.28147
Konjević GM, Vuletić AM, Mirjačić Martinović KM et al (2019) The role of cytokines in the regulation of NK cells in the tumor environment. Cytokine 117:30–40. https://doi.org/10.1016/j.cyto.2019.02.001
Kotzur R, Duev-Cohen A, Kol I et al (2022) NK-92 cells retain vitality and functionality when grown in standard cell culture conditions. PLoS One 17:1–13. https://doi.org/10.1371/journal.pone.0264897
Lee DW, Gardner R, Porter DL et al (2014) Current concepts in the diagnosis and management of cytokine release syndrome. Blood 124:188–195. https://doi.org/10.1182/blood-2014-05-552729
Liu S, Deng B, Yin Z et al (2021) Combination of CD19 and CD22 CAR-T cell therapy in relapsed B-cell acute lymphoblastic leukemia after allogeneic transplantation. Am J Hematol 96:671–679. https://doi.org/10.1002/ajh.26160
Long AH, Haso WM, Shern JF et al (2015) 4–1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat Med 21:581–590. https://doi.org/10.1038/nm.3838
Milone MC, Fish JD, Carpenito C et al (2009) Chimeric receptors containing CD137 signal transduction domains mediate enhanced survival of T cells and increased antileukemic efficacy in vivo. Mol Ther 17:1453–1464. https://doi.org/10.1038/mt.2009.83
Müller S, Bexte T, Gebel V et al (2020) High cytotoxic efficiency of lentivirally and alpharetrovirally engineered CD19-specific chimeric antigen receptor natural killer cells against acute lymphoblastic leukemia. Front Immunol 10:1–16. https://doi.org/10.3389/fimmu.2019.03123
Nitschke L (2009) CD22 and Siglec-G: B-cell inhibitory receptors with distinct functions. Immunol Rev 230:128–143. https://doi.org/10.1111/j.1600-065X.2009.00801.x
Overview E (2019) Natural killer cells for cancer immunotherapy: a new CAR is catching up. EBioMedicine 39:1–2. https://doi.org/10.1016/j.ebiom.2019.01.018
Quintarelli C, Sivori S, Caruso S et al (2018) CD19 redirected CAR NK cells are equally effective but less toxic than CAR T cells. Blood 132:3491–3491. https://doi.org/10.1182/blood-2018-99-118005
Rafiq S, Hackett CS, Brentjens RJ (2020) Engineering strategies to overcome the current roadblocks in CAR T cell therapy. Nat Rev Clin Oncol 17:147–167. https://doi.org/10.1038/s41571-019-0297-y
Rassaei N, Dibavar MA, Soleimani M et al (2023) The effect of microvesicles derived from K562 cells on proliferation and apoptosis of human bone marrow mesenchymal stem cells. Iran J Basic Med Sci 26:295–300. https://doi.org/10.22038/IJBMS.2023.66903.14675
Roex G, Campillo-Davo D, Flumens D et al (2022) Two for one: targeting BCMA and CD19 in B-cell malignancies with off-the-shelf dual-CAR NK-92 cells. J Transl Med 20:1–13. https://doi.org/10.1186/s12967-022-03326-6
Romanski A, Uherek C, Bug G et al (2016) CD19-CAR engineered NK-92 cells are sufficient to overcome NK cell resistance in B-cell malignancies. J Cell Mol Med 20:1287–1294. https://doi.org/10.1111/jcmm.12810
Savani M, Oluwole O, Dholaria B (2021) New targets for CAR T therapy in hematologic malignancies. Best Pract Res Clin Haematol 34:. https://doi.org/10.1016/j.beha.2021.101277
Schönfeld K, Sahm C, Zhang C et al (2015) Selective inhibition of tumor growth by clonal NK cells expressing an ErbB2/HER2-specific chimeric antigen receptor. Mol Ther 23:330–338. https://doi.org/10.1038/mt.2014.219
Shah NN, Stevenson MS, Yuan CM et al (2015) Characterization of CD22 expression in acute lymphoblastic leukemia. Pediatr Blood Cancer 62:964–969. https://doi.org/10.1002/pbc.25410
Spiegel JY, Patel S, Muffly L et al (2021) CAR T cells with dual targeting of CD19 and CD22 in adult patients with recurrent or refractory B cell malignancies: a phase 1 trial. Nat Med 27:1419–1431. https://doi.org/10.1038/s41591-021-01436-0
Suck G, Linn YC, Tonn T (2016) Natural killer cells for therapy of leukemia. Transfus Med Hemotherapy 43:89–95. https://doi.org/10.1159/000445325
Tanaka J (2021) Recent advances in cellular therapy for malignant lymphoma. Cytotherapy 23:662–671. https://doi.org/10.1016/j.jcyt.2020.12.007
Wang L, Qin W, Huo YJ et al (2020) Advances in targeted therapy for malignant lymphoma. Signal Transduct Target Ther 5:. https://doi.org/10.1038/s41392-020-0113-2
Wang WN, Zhou GY, Zhang WL (2017) NK-92 cell, another ideal carrier for chimeric antigen receptor. Immunotherapy 9:753–765. https://doi.org/10.2217/imt-2017-0022
**a J, Minamino S, Kuwabara K (2020) CAR-expressing NK cells for cancer therapy: a new hope. Biosci Trends 14:354–359. https://doi.org/10.5582/bst.2020.03308
**ao X, Ho M, Zhu Z et al (2009) Identification and characterization of fully human anti-CD22 monoclonal antibodies. Mabs 1:297–303. https://doi.org/10.4161/mabs.1.3.8113
Xu D, ** G, Chai D et al (2018) The development of CAR design for tumor CAR-T cell therapy. Oncotarget 9:13991–14004. https://doi.org/10.18632/oncotarget.24179
Acknowledgements
We would like to acknowledge our colleagues for their helpful assistance in this study.
Author information
Authors and Affiliations
Contributions
MS and MHM designed and contributed to the concept of the experiments; MAD performed experiments and collected data; MSZ discussed the results and strategy and interpretation of data; MS and MHM supervised, directed, and managed the study; All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This project was approved by Ethical Committee of Shahid Beheshti University of Medical Sciences (IR.SBMU.RETECH.REC.1402.158) (Approval date: 11/06/2023).
Consent for publication
Not applicable.
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this article.
Supplementary Information
Below is the link to the electronic supplementary material.
11626_2024_895_MOESM2_ESM.tif
Supplementary file2 Fig. S2: the effect of CD22-CAR NK-92 cells on the CFSE-labeled target cells by a fluorescence microscope. A: the left one showed CFSE-labeled Raji cells alone. The right one showed CFSE-labeled Raji cells were co-incubated with CD22-CAR NK-92 cells for 24 h at E: T ratios of 10:1. B: the left one showed CFSE-labeled K562 cells alone. The right one showed CFSE-labeled K562 cells were co-incubated with CD22-CAR NK-92 cells for 24 h at E: T ratios of 10:1. (TIF 8566 KB)
11626_2024_895_MOESM3_ESM.tif
Supplementary file3 Fig. S3: Flow cytometry plots showing the gating strategy used in cytotoxicity experiments. A: the cytotoxic activity of untransduced NK-92 cells against Raji. Each row showed different E: T ratios of 1:2, 1:1, 2:1, 5:1, and 10:1 respectively. B: CD22-CAR NK-92 -transduced cells against Raji. Each row showed different E: T ratios of 1:2, 1:1, 2:1, 5:1, and 10:1 respectively. (TIF 4010 KB)
11626_2024_895_MOESM4_ESM.tif
Supplementary file4 Fig. S4: Flow cytometry plot showing the gating strategy used in cytotoxicity experiments. A: the cytotoxic activity of untransduced NK-92 cells against K562. Each row showed different E: T ratios of 1:2, 1:1, 2:1, 5:1, and 10:1 respectively. B: CD22-CAR NK-92 -transduced cells against K562. Each row showed different E: T ratios of 1:2, 1:1, 2:1, 5:1, and 10:1 respectively. (TIF 3859 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abbaszade Dibavar, M., Soleimani, M., Mohammadi, M.H. et al. High yield killing of lymphoma cells by anti-CD22 CAR-NK cell therapy. In Vitro Cell.Dev.Biol.-Animal 60, 321–332 (2024). https://doi.org/10.1007/s11626-024-00895-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-024-00895-2