Abstract
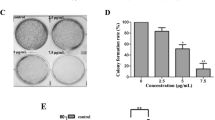
When used in combination with certain chemotherapies, curcumin has been shown to increase apoptosis in several cancer cell lines. Here, we report the combined effects of curcumin and cinobufacini on human cervical carcinoma cells. The aim of this study was to examine whether curcumin could enhance apoptosis induced by cinobufacini. 3-(4,5-Dimethylthiazol-2-y1)-2,5-diphenytetrazolium bromide (MTT) assays revealed that the growth and proliferation of HeLa cells could be inhibited by 75% after a combined treatment of 25 μg/mL cinobufacini and 8 μg/mL curcumin. The combined treatment is 3 times more effective than treatment with 25 μg/mL cinobufacini alone. Annexin V-FITC/PI staining, morphological changes and immunofluorescence verified a significant enhancement in cinobufacini-induced apoptosis when cells were also exposed to curcumin. The data showed that the proportion of early apoptotic cells significantly increased from 15.43% in cells treated only with 25 μg/mL cinobufacini to 49.2% in cells treated with 25 μg/mL cinobufacini and 8 μg/mL curcumin. Moreover, compared with treatment of only 25 μg/mL cinobufacini, ROS production increased 1.7-fold, the intracellular free Ca2+ concentration increased 1.5-fold, and the mitochondrial membrane potential decreased by 20% in the combined treatment. Simultaneously, the atomic force microscopy (AFM) results suggest that cells treated with a combination of cinobufacini and curcumin varied significantly in shape and ultrastructure. Collapsed cells with leaking cytoplasm, blebbing pores and emerging apoptotic bodies were prevalent. The nanoparticle size increased from 70 nm when the cells were treated with 25 μg/mL cinobufacini to 190 nm when the cells were treated with 25 μg/mL cinobufacini and 8 μg/mL curcumin. The size increase resulted in the cell membrane becoming considerably rough. These results can improve our understanding of combination treatments. Specifically, the combination of cinobufacini and curcumin may potentially find use as a novel cervical carcinoma treatment. Additionally, AFM is a powerful tool that can be used to explore cellular morphologies and ultrastructures.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bonomi P, Blessing J A, Stehman F B, et al. Randomized trial of three cisplatin dose schedules in squamous-cell carcinoma of the cervix: A Gynecologic Oncology Group study. J Clin Oncol, 1985, 3: 1079–1085
Omura G A. Chemotherapy for cervix cancer. Semin Oncol, 1994, 21: 54–62
McGuire W P, Blessing J A, Moore D, et al. Paclitaxel has moderate activity in squamous cervix cancer. A Gynecologic Oncology Group study. J Clin Oncol, 1996, 14: 792–795
Sutton G P, Blessing J A, McGuire W P, et al. Phase II trial of ifosfamide and mesna in patients with advanced or recurrent squamous carcinoma of the cervix who had never received chemotherapy: A Gynecologic Oncology Group study. Am J Obstet Gynecol, 1993, 168: 805–807
Muderspach L I, Blessing J A, Levenback C, et al. A phase II study of topotecan in patients with squamous cell carcinoma of the cervix: A Gynecologic Oncology Group study. Gynecol Oncol, 2001, 81: 213–215
Long III H J, Bundy B N, Grendys E C, et al. Randomized phase III trial of cisplatin with orwithout topotecan in carcinoma of the uterine cervix: A Gynecologic Oncology Group study. J Clin Oncol, 2005, 23: 4626–4633
Luk J M, Wang X, Liu P, et al. Traditional Chinese herbal medicines for treatment of liver fibrosis and cancer: From laboratory discovery to clinical evaluation. Liver Int, 2007, 27: 879–890
Zuo X D, Cui Y A, Qin S K, et al. Clinical research progress on the antitumor effects of cinobufacini (in Chinese). Chin Clin Oncol, 2003, 8: 232–235
Zuo X D, Cui Y A, Qin S K, et al. Effect of cinobufacini on tumor cell cycle and expression of Bcl-2 protein (in Chinese). Mod J Integr Chin Tradit West Med, 2003, 12: 567–568
Deshpande S S, Maru G B. Effects of curcumin on the formation of benzo [a] pyrene derived DNA adducts in vitro. Cancer Lett, 1995, 96: 71–80
Sharma R A, Gescher A J, Steward W P. Curcumin: The story so far. Eur J Cancer, 2005, 41: 1955–1968
Maheshwari R K, Singh A K, Gaddipati J, et al. Multiple biological activities of curcumin: A short review. Life Sci, 2006, 78: 2081–2087
Ono K, Hasegawa K, Naike H, et al. Curcumin has potent anti-amyloidogenic effects for Alzheimer’s beta-amyloidfibrils in vitro. Neurosci Res, 2004, 75: 742–750
Aggarwal B B, Kumar A, Bharti A C. Anticancer potential of curcu min: Preclinical and clinical studies. Anticancer Res, 2003, 23: 363–398
Syng-ai C, Kumari A L, Khar A. Effect of curcumin on normal and tumor cells: Role of glutathione and Bcl-2. Mol Cancer Ther, 2004, 3: 1101–1108
Kunwar A, Barik A, Mishra B, et al. Quantitative cellular uptake, localization and cytotoxicitof curcumin in normal and tumor cells. Biochim Biophys Acta-Gen Subj, 2008, 1780: 673–679
Scarlett J L, Sheard P W, Hughes G, et al. Changes in mitochondrial membrane potential during staurosporine-induced apoptosis in Jurkat cells. FEBS Lett, 2000, 475: 267–272
Puech P H, Poole K, Knebel D, et al. A new technical approach to quantify cell-cell adhesion forces by AFM. Ultramicroscopy, 2006, 106: 637–644
Alarmo E, Pärssinen J, Ketolainen J M, et al. BMP7 influences proliferation, migration, and invasion of breast cancer cells. Cancer Lett, 2009, 275: 35–43
Heidemann S R, Wirtz D. Towards a regional approach to cellmechanics. Trends Cell Biol, 2004, 14: 160–166
Suresh S. Biomechanics and biophysics of cancer cells. Acta Mater, 2007, 55: 3989–4014
Shimizu S, Eguchi Y, Kamiike W. Induction of apoptosis as well as necrosis by hypoxia and predominant prevention of apoptosis by Bcl-2 and Bcl-XL. Cancer Res, 1996, 56: 2161–2166
Grzanka A, Grzanka D, Orlikowska M. Cytoskeletal reorganization during process of apoptosis induced by cytostatic drugs in K-562 and HL-60 leukemia cell lines. Biochem Pharmacol, 2003, 66: 1611–1617
Melanie H, Amanda M G V, Pieter S, et al. Rho-kinase-dependent F-actin rearrangement in involved in the inhibition of PI3-kinase/Akt during ischemia-reperfusion-induced endothelial cell apoptosis. Apoptosis, 2008, 13: 404–412
Olivia N T, Jocelyne H, Jacqueline B. Cytoskeleton and apoptosis. Biochem Pharmacol, 2008, 76: 11–18
Escobar M L, Echeverría R O, Vázquez-Nin G H. Combined apoptosis and autophagy, the process that eliminates the oocytes of atretic follicles in immature rats. Apoptosis, 2008, 13: 1253–1266
Lekka M, Laidler P. Applicability of AFM in cancer detection. Nat Nanotechnol, 2009, 4: 72–73
** H, Huang X, Chen Y, et al. Photoinactivation effects of hematoporphyrin monomethyl ether on Gram-positive and -negative bacteria detected by atomic force microscopy. Appl Microbiol Biotechnol, 2010, 88: 761–770
Christian L G, Lesniewska E, Giocondi M, et al. Imaging of the surface of living cells by low-force contact-mode atomic force microscopy. Biophys J, 1998, 75: 695–703
Wang M, Ruan Y X, Chen Q, et al. Curcumin induced HepG2 cell apoptosis-associated mitochondrial membrane potential and intracellular free Ca2+ concentration. Eur J Pharmacol, 2011, 650: 41–47
Denning T L, Takaishi H, Crowe S E, et al. Oxidative stress induces the expression of Fas and Fas ligand and apoptosis in murine intestinal epithelial cell. Free Radic Biol Med, 2002, 33: 1641–1650
Lin Y, Choksi S, Shen H M, et al. Tumor necrosis factor-induced nonapoptotic cell death requires receptor-interacting protein-mediated cellular reactive oxygen species accumulation. J Biol Chem, 2004, 279: 10822–10828
Ortiz-Sánchez E, Daniels T R, Helguera G, et al. Enhanced cytotoxicity of an anti-transferrin receptor IgG3-avidin fusion protein in combination with gambogic acid against human malignant hematopoietic cells: Functional relevance of iron, the receptor, and reactive oxygen species. Leukemia, 2009, 23: 59–70
Trachootham D, Zhou Y, Zhang H, et al. Selective killing of oncogenically transformed cells through a ROS-mediated mechanism by β-phenylethyl isothiocyanate. Cancer Cell, 2006, 10: 241–252
Lu M, Bi C S, Gong X G, et al. Anti-proliferative effects of recombinant iron superoxide dismutase on HepG2 cells via a redox-dependent PI3k/Akt pathway. Appl Microbiol Biotechnol, 2007, 76: 193–201
Raj L, Ide T, Gurkar A U, et al. Selective killing of cancer cells by a small molecule targeting the stress response to ROS. Nature, 2011, 475: 231–234
Orrenius S, Zhivotovsky B, Nicotera P. Regulation of cell death: The calcium-apoptosis link. Nat Rev Mol Cell Biol, 2003, 4: 552–565
Jared L S, Philip W S, Gillian H, et al. Changes in mitochondrial membrane potential during staurosporine-induced apoptosis in Jurkat cells. FEBS Lett, 2000, 475: 267–272
Douglas R G, Guido K. The pathophysiology of mitochondrial cell death. Science, 2004, 305: 626–629
Waterhouse N J, Goldstein J C, Ahsen O V, et al. Cytochrome c maintains mitochondrial transmem-brane potential and ATP generation after outer mitochondrial membrane perme-abilization during the apoptotic process. J Cell Biol, 2001, 153: 319–328
Hu W, Kavanagh J J. Anticancer therapy targeting the apoptotic pathway. Lancet Oncol, 2003, 4: 721–729
Li Y H, Li X L, Wong Y S, et al. The reversal of cisplatin-induced nephrotoxicity by selenium nanoparticles functionalized with 11-mercapto-1-undecanol by inhibition of ROS-mediated apoptosis. Biomaterials, 2011, 32: 9068–9076
Huang C H, ** H, Song B, et al. The cytotoxicity and anticancer mechanisms of alterporriol L, a marine bianthraquinone, against MCF-7 human breast cancer cells. Appl Microbiol Biotechnol, 2012, 93: 777–785
Wang X. The expanding role of mitochondria in apoptosis. Genes Dev, 2001, 15: 2922–2933
Salganik R I. The benefits and hazards of antioxidants: Controlling apoptosis and other protective mechanisms in cancer patients and the human population. J Am Coll Nutr, 2001, 20: 464S–472S
Shackelford R E, Kaufmann W K, Paules R S. Oxidative stress and cell cycle checkpoint function. Free Radic Biol Med, 2000, 28: 1387–1404
Ma L N, Song B, ** H, et al. Cinobufacini induced MDA-MB-231 cell apoptosis-associated cell cycle arrest and cytoskeleton function. Bioorg Med Chem Lett, 2012, 22: 1459–1463
Paul S B, Yisang Y, James L R, et al. Calcium, ATP, and ROS: A mitochondrial love-hate triangle. Am J Physiol Cell Physiol, 2004, 287: 817–833
Shih C M, Ko W C, Wu J S, et al. Mediating of caspase-independent apoptosis by cadmium through the mitochondria-ROS pathway in MRC-5 fibroblasts. J Cell Biochem, 2004, 91: 384–397
Nicotera P, Bellomo G, Orrenius S. Calcium-mediated mechanisms in chemically induced cell death. Annu Rev Phar-Macol Toxicol, 1992, 32: 449–470
Melanie H, Amanda M G V, Pieter S, et al. Rho-kinase-dependent F-actin rearrangement in involved in the inhibition of PI3-kinase/Akt during ischemia-reperfusion-induced endothelial cell apoptosis. Apoptosis, 2008, 13: 404–412
Olivia N T, Jocelyne H, Jacqueline B. Cytoskeleton and apoptosis. Biochem Pharmacol, 2008, 76: 11–18
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Liu, L., **, H., Ou, J. et al. Cinobufacini-induced HeLa cell apoptosis enhanced by curcumin. Chin. Sci. Bull. 58, 2584–2593 (2013). https://doi.org/10.1007/s11434-013-5739-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-013-5739-9