Abstract
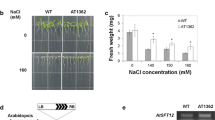
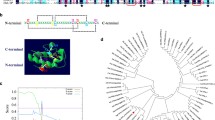
Aquaporins are implicated in a wide variety of plant physiological processes, although the mechanisms involved in their regulation are not fully understood. To gain further insight into the regulatory factors involved in this process, we used a yeast two-hybrid system to screen for potential binding partners to the Arabidopsis tonoplast intrinsic protein (TIP) AtTIP1;1. This was the first protein identified to be associated with high water permeability in vacuolar membranes from Arabidopsis thaliana. Using AtTIP1;1 as bait, a novel binding protein was identified in both yeast and plant cells. This prey protein, named AtSM34, was a 309 aa polypeptide with a predicted molecular mass of 34 kD and contained a single MYB/SANT-like domain. AtSM34 promoter:: GUS histochemical staining analysis detected AtSM34 expression in flowers, stems and leaves, particularly in the vascular tissues, in response to osmotic stress. AtSM34 expression was localized in the endoplasmic reticulum membrane, and sequence deletion analysis revealed that the N-terminal coding region (amino acids 1-83) was critical for this localization. Overexpression of AtSM34 resulted in hypersensitivity to exogenous mannitol, sorbitol and abscisic acid, and caused a significant delay in germination. AtSM34 interacted with AtTIP1;2 and AtTIP2;1, which are essential proteins for modulation of tonoplast permeability and highly expressed in germinating seedlings. These data indicate AtSM34 is a novel TIPs binding protein involved in the osmotic stress response of seedlings at an early stage of development.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alexandersson E, Fraysse L, Sjovall-Larsen S, et al. Whole gene family expression and drought stress regulation of aquaporins. Plant Mol Biol, 2005, 59: 469–484
Jang J Y, Rhee J Y, Kim D G, et al. Ectopic expression of a foreign aquaporin disrupts the natural expression patterns of endogenous aquaporin genes and alters plant responses to different stress conditions. Plant Cell Physiol, 2007, 48: 1331–1339
Gerbeau P, Amodeo G, Henzler T, et al. The water permeability of Arabidopsis plasma membrane is regulated by divalent cations and pH. Plant J, 2002, 30: 71–81
Uehlein N, Lovisolo C, Siefritz F, et al. The tobacco aquaporin NtAQP1 is a membrane CO2 pore with physiological functions. Nature, 2003, 425: 734–737
Chaumont F, Moshelion M, Daniels M J. Regulation of plant aquaporin activity. Biol Cell, 2005, 97: 749–764
Fischer M, Kaldenhoff R. On the pH regulation of plant aquaporins. J Biol Chem, 2008, 283: 33889–33892
Reichow S L, Gonen T. Noncanonical binding of calmodulin to aquaporin-0: Implications for channel regulation. Structure, 2008, 16: 1389–1398
Lu H A, Sun T X, Matsuzaki T, et al. Heat shock protein 70 interacts with aquaporin-2 and regulates its trafficking. J Biol Chem, 2007, 282: 28721–28732
Kim M J, Kim H R, Paek K H. Arabidopsis tonoplast proteins TIP1 and TIP2 interact with the cucumber mosaic virus 1a replication protein. J Gen Virol, 2006, 87: 3425–3431
Masalkar P, Wallace I S, Hwang J H, et al. Interaction of cytosolic glutamine synthetase of soybean root nodules with the C-terminal domain of the symbiosome membrane nodulin 26 aquaglyceroporin. J Biol Chem, 2010, 285: 23880–23888
Lee H K, Cho S K, Son O, et al. Drought stress-induced Rma1H1, a RING membrane-anchor E3 ubiquitin ligase homolog, regulates aquaporin levels via ubiquitination in transgenic Arabidopsis plants. Plant Cell, 2009, 21: 622–641
Wudick M M, Luu D T, Maurel C. A look inside: Localization patterns and functions of intracellular plant aquaporins. New Phytol, 2009, 184: 289–302
Kaldenhoff R, Fischer M. Aquaporins in plants. Acta Physiol (Oxf), 2006, 187: 169–176
Vera-Estrella R, Barkla B J, Bohnert H J, et al. Novel regulation of aquaporins during osmotic stress. Plant Physiol, 2004, 135: 2318–2329
Maurel C, Chrispeels M, Lurin C, et al. Function and regulation of seed aquaporins. J Exp Bot, 1997, 48: 421–430
Schüssler M D, Alexandersson E, Bienert G P, et al. The effects of the loss of TIP1;1 and TIP1;2 aquaporins in Arabidopsis thaliana. Plant J, 2008, 56: 756–767
Beebo A, Thomas D, Der C, et al. Life with and without AtTIP1;1, an Arabidopsis aquaporin preferentially localized in the apposing tonoplasts of adjacent vacuoles. Plant Mol Biol, 2009, 70: 193–209
Slabaugh E, Held M, Brandizzi F. Control of root hair development in Arabidopsis thaliana by an endoplasmic reticulum anchored member of the R2R3-MYB transcription factor family. Plant J, 2011, DOI: 10.1111/j.1365-313X.2011.04602.x
Stracke R, Werber M, Weisshaar B. The R2R3-MYB gene family in Arabidopsis thaliana. Curr Opin Plant Biol, 2001, 4: 447–456
Walter M, Chaban C, Schutze K, et al. Visualization of protein interactions in living plant cells using bimolecular fluorescence complementation. Plant J, 2004, 40: 428–438
Sheen J. A transient expression assay using Arabidopsis mesophyll protoplasts. 2002, http://genetics.mgh.harvard.edu/sheenweb/
An R, Chen Q J, Chai M F, et al. AtNHX8, a member of the monovalent cation: Proton antiporter-1 family in Arabidopsis thaliana, encodes a putative Li/H antiporter. Plant J, 2007, 49: 718–728
Xu J, Li H D, Chen L Q, et al. A protein kinase, interacting with two calcineurin B-like proteins, regulates K+ transporter AKT1 in Arabidopsis. Cell, 2006, 125: 1347–1360
Cao X, Yang K Z, **a C, et al. Characterization of DUF724 gene family in Arabidopsis thaliana. Plant Mol Biol, 2010, 72: 61–73
Wang M, Xu Q, Yu J, et al. The putative Arabidopsis zinc transporter ZTP29 is involved in the response to salt stress. Plant Mol Biol, 2010, 73: 467–479
Chen N Z, Zhang X Q, Wei P C, et al. AtHAP3b plays a crucial role in the regulation of flowering time in Arabidopsis during osmotic stress. J Biochem Mol Biol, 2007, 40: 1083–1089
Charrier B, Champion A, Henry Y, et al. Expression profiling of the whole Arabidopsis shaggy-like kinase multigene family by real-time reverse transcriptase-polymerase chain reaction. Plant Physiol, 2002, 130: 577–590
Livak K J, Schmittgen T D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods, 2001, 25: 402–408
Chen Y F, Li L Q, Xu Q, et al. The WRKY6 transcription factor modulates PHOSPHATE1 expression in response to low Pi stress in Arabidopsis. Plant Cell, 2009, 21: 3554–3566
Clough S J, Bent A F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J, 1998, 16: 735–743
Hunter P R, Craddock C P, Di Benedetto S, et al. Fluorescent reporter proteins for the tonoplast and the vacuolar lumen identify a single vacuolar compartment in Arabidopsis cells. Plant Physiol, 2007, 145: 1371–1382
Vitale A, Hinz G. Sorting of proteins to storage vacuoles: How many mechanisms? Trends Plant Sci, 2005, 10: 316–323
Jelinkova A, Malinska K, Simon S, et al. Probing plant membranes with FM dyes: Tracking, dragging or blocking? Plant J, 2010, 61: 883–892
Kim D H, Eu Y J, Yoo C M, et al. Trafficking of phosphatidylinositol 3-phosphate from the trans-Golgi network to the lumen of the central vacuole in plant cells. Plant Cell, 2001, 13: 287–301
Maurel C, Verdoucq L, Luu D T, et al. Plant aquaporins: Membrane channels with multiple integrated functions. Annu Rev Plant Biol, 2008, 59: 595–624
Johnson K D, Herman E M, Chrispeels M J. An abundant, highly conserved tonoplast protein in seeds. Plant Physiol, 1989, 91: 1006–1013
Höfte H, Hubbard L, Reizer J, et al. Vegetative and seed-specific forms of tonoplast intrinsic protein in the vacuolar membrane of Arabidopsis thaliana. Plant Physiol, 1992, 99: 561–570
Vander W C, Postaire O, Tournaire-Roux C, et al. Expression and inhibition of aquaporins in germinating Arabidopsis seeds. Plant Cell Physiol, 2006, 47: 1241–1250
Peng Y, Lin W, Cai W, et al. Overexpression of a Panax ginseng tonoplast aquaporin alters salt tolerance, drought tolerance and cold acclimation ability in transgenic Arabidopsis plants. Planta, 2007, 226: 729–740
Yamaguchi-Shinozaki K, Shinozaki K. Characterization of the expression of a desiccation-responsive rd29 gene of Arabidopsis thaliana and analysis of its promoter in transgenic plants. Mol Gen Genet, 1993, 236: 331–340
Lee M M, Schiefelbein J. Developmentally distinct MYB genes encode functionally equivalent proteins in Arabidopsis. Development, 2001, 128: 1539–1546
Abe H, Urao T, Ito T, et al. Arabidopsis AtMYC2 (bHLH) and AtMYB2 (MYB) function as transcriptional activators in abscisic acid signaling. Plant Cell, 2003, 15: 63–78
Denekamp M, Smeekens S C. Integration of wounding and osmotic stress signals determines the expression of the AtMYB102 transcription factor gene. Plant Physiol, 2003, 132: 1415–1423
Araki S, Ito M, Soyano T, et al. Mitotic cyclins stimulate the activity of c-Myb-like factors for transactivation of G2/M phase-specific genes in tobacco. J Biol Chem, 2004, 279: 32979–32988
Barg R, Sobolev I, Eilon T, et al. The tomato early fruit specific gene Lefsm1 defines a novel class of plant-specific SANT/MYB domain proteins. Planta, 2005, 221: 197–211
Hamaguchi A, Yamashino T, Koizumi N, et al. A small subfamily of Arabidopsis RADIALIS-LIKE SANT/MYB genes: A link to HOOKLESS1-mediated signal transduction during early morphogenesis. Biosci Biotechnol Biochem, 2008, 72: 2687–2696
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
This article is published with open access at Springerlink.com
Electronic supplementary material
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Li, L., Ren, F., Wei, P. et al. Identification of AtSM34, a novel tonoplast intrinsic protein-interacting polypeptide expressed in response to osmotic stress in germinating seedlings. Chin. Sci. Bull. 56, 3518–3530 (2011). https://doi.org/10.1007/s11434-011-4793-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4793-4