Abstract
Introduction
Plant cell walls play an important role in providing physical strength and defence against abiotic stress. Rice brittle culm (bc) mutants are a strength-decreased mutant because of abnormal cell walls, and it has been reported that the causative genes of bc mutants affect cell wall composition. However, the metabolic alterations in each organ of bc mutants have remained unknown.
Objectives
To evaluate the metabolic changes in rice bc mutants, comparative analysis of the primary metabolites was conducted.
Methods
The primary metabolites in leaves, internodes, and nodes of rice bc mutants and wild-type control were measured using CE- and LC-MS/MS. Multivariate analyses using metabolomic data was performed.
Results
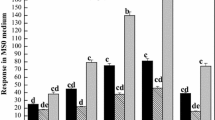
We found that mutations in each bc mutant had different effects on metabolism. For example, higher oxalate content was observed in bc3 and bc1 bc3 mutants, suggesting that surplus carbon that was not used for cell wall components might be used for oxalate synthesis. In addition, common metabolic alterations such as a decrease of sugar nucleotides in nodes were found in bc1 and Bc6, in which the causative genes are involved in cellulose accumulation.
Conclusion
These results suggest that metabolic analysis of the bc mutants could elucidate the functions of causative gene and improve the cell wall components for livestock feed or bioethanol production.






Similar content being viewed by others
Abbreviations
- DHAP:
-
Dihydroxyacetone phosphate
- G1P:
-
Glucose-1-phosphate
- G-3-P:
-
Glyceraldehyde-3-phosphate
- G6P:
-
Glucose-6-phosphate
- GABA:
-
Gamma-aminobutyrate
- gEC:
-
Gamma-glutamylcysteine
- GSH:
-
Glutathione (reduced)
- GSSG:
-
Glutathione disulphide
- F6P:
-
Fructose-6-phosphate
- FBP:
-
Fructose-1,6-bisphosphate
- 2OG:
-
2-Oxoglutarate
- PC:
-
Principal component
- PEP:
-
Phosphoenolpyruvate
- 6PG:
-
6-Phosphogluconate
- 3PGA:
-
3-Phosphoglycerate
- RuBP:
-
Ribulose-1,5-bisphosphate
- R5P:
-
Ribose-5-phosphate
- Ru5P:
-
Ribulose-5-phosphate
- S7P:
-
Sedoheptulose-7-phosphate
- T65:
-
Taichung 65
- TCA:
-
Tricarboxylic acid
- UDP-Ara:
-
UDP-arabinose
- UDP-Gal:
-
UDP-galactose
- UDP-GalA:
-
UDP-galacturonate
- UDP-Glc:
-
UDP-glucose
- UDP-GlcA:
-
UDP-glucuronate
- UDP-GlcNAc:
-
UDP-N-acetylglucosamine
- UDP-Xyl:
-
UDP-xylose
References
Adachi, S., Tanaka, Y., Miyagi, A., Kashima, M., Tezuka, A., Toya, Y., Kobayashi, S., Ohkubo, S., Shimizu, H., Kawai-Yamada, M., Sage, R. F., Nagano, A. J., & Yamori, W. (2019). High-yielding rice Takanari has superior photosynthetic response to a commercial rice Koshihikari under fluctuating light. Journal of Experimental Botany, 70, 5287–5297.
Aohara, T., Kotake, T., Kaneko, Y., Takatsuji, H., Tsumuraya, Y., & Kawasaki, S. (2009). Rice BRITTLE CULM5 (BRITTLE NODE) is involved in secondary cell wall formation in the sclerenchyma tissue of nodes. Plant, Cell and Physiology, 50, 1886–1897.
Barros, J., Escamilla-Trevino, L., Song, L., Rao, X., Serrani-Yarce, J., Palacios, M., Engle, N., Choudhury, F., Tschaplinski, T., Venables, B., Mittler, R., & Dixon, R. (2019). 4-Coumarate 3-hydroxylase in the lignin biosynthesis pathway is a cytosolic ascorbate peroxidase. Nature Communications, 10, 1994.
Brown, D. M., Zeef, L. A. H., Ellis, J., Goodacre, R., & Turner, S. R. (2005). Identification of novel genes in Arabidopsis involved in secondary cell wall formation using expression profiling and reverse genetics. The Plant Cell, 17, 2281–2295.
Carpita, N., & McCann, M. (2015). Characterizing visible and invisible cell wall mutant phenotypes. Journal of Experimental Botany, 66, 4145–4163.
Franceschi, V., & Nakata, P. (2005). Calcium oxalate in plants: Formation and function. Annual Review of Plant Biology, 56, 41–71.
Halliwell, B. (1978). Lignin synthesis: The generation of hydrogen peroxide and superoxide by horseradish peroxidase and its stimulation by manganese (II) and phenols. Planta, 140, 81–88.
Hirano, K., Kotake, T., Kamihara, K., Tsuna, K., Aohara, T., Kaneko, Y., Takatsuji, H., Tsumuraya, Y., & Kawasaki, S. (2010). Rice BRITTLE CULM3 (BC3) encodes a classical dynamin OsDRP2B essential for proper secondary cell wall synthesis. Planta, 232, 95–108.
Honda, S., Ohkubo, S., San, N. S., Nakkasame, A., Tomisawa, K., Katsura, K., Ookawa, T., Nagano, A. J., & Adachi, S. (2021). Maintaining higher leaf photosynthesis after heading stage could promote biomass accumulation in rice. Scientific Reports, 11, 7579.
Ito, J., Herter, T., Baidoo, E., Lao, J., Vega-Sanchez, M., Smith-Moritz, M., Adams, P., Keasling, J., Usabel, B., Petzold, C., & Heazlewood, J. (2014). Analysis of plant nucleotide sugars by hydrophilic interaction liquid chromatography and tandem mass spectrometry. Analytical Biochemistry, 448, 14–22.
Kokubo, A., Kuraishi, S., & Sakurai, N. (1989). Culm strength of barley. Plant Physiology, 91, 876–882.
Kokubo, A., Sakurai, N., Kuraishi, S., & Takeda, K. (1991). Culm brittleness of barley (Hordeum vulgare L.) mutants is caused by smaller number of cellulose molecules in cell wall. Plant Physiology, 97, 509–514.
Kotake, T., Aohara, T., Hirano, K., Sato, A., Kaneko, Y., Tsumuraya, Y., Takatsuji, H., & Kawasaki, S. (2011). Rice Brittle culm 6 encodes a dominant-negative form of CesA protein that perturbs cellulose synthesis in secondary cell walls. Journal of Experimental Botany, 62, 2053–2062.
Li, Y., Qian, Q., Zhou, Y., Yan, M., Sun, L., Zhang, M., Fu, Z., Wang, Y., Han, B., Pan, X., Chen, M., & Li, J. (2003). Brittle culm 1, which encodes a COBRA-like protein, affects the mechanical properties of rice plants. The Plant Cell, 15, 2020–2031.
Miyagi, A., Kawai-Yamada, M., Uchimiya, M., Ojima, N., Suzuki, K., & Uchimiya, H. (2013a). Metabolome analysis of food-chain between plants and insects. Metabolomics, 9, 1254–1261.
Miyagi, A., Noguchi, K., Tokida, T., Usui, Y., Nakamura, H., Sakai, H., Hasegawa, T., & Kawai-Yamada, M. (2019). Oxalate contents in leaves of two rice cultivars grown at a free-air CO2 enrichment (FACE) site. Plant Production Science, 22, 407–411.
Miyagi, A., Takahashi, H., Takahara, K., Hirabayashi, T., Nishimura, Y., Tezuka, T., Kawai-Yamada, M., & Uchimiya, H. (2010). Principal component and hierarchical clustering analysis of metabolites in destructive weeds; polygonaceous plants. Metabolomics, 6, 146–155.
Miyagi, A., Uchimiya, M., Kawai-Yamada, M., & Uchimiya, H. (2013b). An antagonist treatment in combination with tracer experiments revealed isocitrate pathway dominant to oxalate biosynthesis in Rumex obtusifolius L. Metabolomics, 9, 590–598.
Noguchi, K., Tsunoda, T., Miyagi, A., Kawai-Yamada, M., Sugiura, D., Miyazawa, S.-I., Tokida, T., Usui, Y., Nakamura, H., Sakai, H., & Hasegawa, T. (2018). Effects of elevated atmospheric CO2 on respiratory rates in mature leaves of two rice cultivars grown at a free-air CO2 enrichment site and analyses of the underlying mechanisms. Plant and Cell Physiology, 59, 637–649.
Ohkubo, S., Tanaka, Y., Yamori, W., & Adachi, S. (2020). Rice cultivar Takanari has higher photosynthetic performance under fluctuating light than Koshihikari, especially under limited nitrogen supply and elevated CO2. Frontiers in Plant Science, 11, 1308.
Ookawa, T., Aoba, R., Yamamoto, T., Ueda, T., Takai, T., Fukuoka, S., Ando, T., Adachi, S., Matsuoka, M., Ebitani, T., Kato, Y., Mulsanti, I., Kishii, M., Reynolds, M., Piñera, F., Kotake, T., Kawasaki, S., Motobayashi, T., & Hirasawa, T. (2016). Precise estimation of genomic regions controlling lodging resistance using a set of reciprocal chromosome segment substitution lines in rice. Scientific Reports, 6, 30572.
Sanchez, A., & Khush, G. (1994). Chromosomal location of some marker genes in rice using the primary trisomics. The Journal of Heredity, 85, 297–300.
Sato-Izawa, K., Nakamura, S., & Matsumoto, T. (2020). Mutation of rice bc1 gene affects internode elongation and induces delayed cell wall deposition in develo** internodes. Plant Signaling & Behavior, 15, e1749786.
Sharma, U., Brillouet, J., Scalbert, A., & Monties, B. (1986). Studies on a brittle stem mutant of rice, Oryza sativa L.; characterization of lignin fractions, associated phenolic acids and polysaccharides from rice stem. Agronomie, 6, 265–271.
Sindhu, A., Langewisch, T., Olek, A., Multani, D., McCann, M., Vermerris, W., Cartita, N., & Johal, G. (2007). Maize brittle stalk2 encodes a COBRA-like protein expressed in early organ development but required for tissue flexibility at maturity. Plant Physiology, 145, 1444–1459.
Song, X., Liu, L., Jiang, Y., Zhang, B., Gao, Y., Liu, X., Lin, Q., Ling, H., & Zhou, Y. (2013). Disruption of secondary wall cellulose biosynthesis alters cadmium translocation and tolerance in rice plants. Molecular Plant, 6, 768–780.
Takahashi, M., Kinoshita, T., & Takeda, K. (1968). Character expressions and causal genes of some mutants in rice plant: (Genetical studies on rice plant, XXXIII). Journal of the Faculty of Agriculture, Hokkaido University, 55, 496–512.
Takenaka, Y., Watanabe, Y., Schuetz, M., Unda, F., Hill, J., Phookaew, P., Yoneda, A., Mansfield, S., Samuels, L., Ohtani, M., & Demura, T. (2018). Patterned deposition of xylan and lignin is independent from that of the secondary wall cellulose of arabidopsis xylem vessels. The Plant Cell, 30, 2663–2676.
Tanaka, K., Murata, K., Yamazaki, M., Onosato, K., Miyao, A., & Hirochika, H. (2003). Three distinct rice cellulose synthase catalytic subunit genes required for cellulose synthesis in the secondary wall. Plant Physiology, 133, 73–83.
Vaughan, P., & Butt, V. (1970). The action of o-dihydric phenols in the hydroxylation of p-coumaric acid by a phenolase from leaves of spinach beet (Beta vulgaris L.). Biochemical Journal, 119, 89–94.
**ong, G., Li, R., Qian, Q., Song, X., Liu, X., Yu, Y., Zeng, D., Wan, J., Li, J., & Zhou, Y. (2010). The rice dynamin-related protein DRP2B mediates membrane trafficking, and thereby plays a critical role in secondary cell wall cellulose biosynthesis. The Plant Journal, 64, 56–70.
Yan, C., Yan, S., Zeng, X., Zhang, Z., & Gu, M. (2007). Fine map** and isolation of Bc7(t), allelic to OsCesA4. Journal of Genetics and Genomics, 34, 1019–1027.
Zhang, B., Deng, L., Qian, Q., **ong, G., Zeng, D., Li, R., Guo, L., Li, J., & Zhou, Y. (2009). A missense mutation in the transmembrane domain of CESA4 affects protein abundance in the plasma membrane and results in abnormal cell wall biosynthesis in rice. Plant Molecular Biology, 71, 509–524.
Zhang, M., Zhang, B., Qian, Q., Yu, Y., Li, R., Zhang, J., Liu, X., Zeng, D., Li, J., & Zhou, Y. (2010). Brittle Culm 12, a dual-targeting kinesin-4 protein, controls cell-cycle progression and wall properties in rice. The Plant Journal, 63, 312–328.
Zhong, R., Burk, D., Morrison, W., & Ye, Z. (2002). A Kinesin-like protein is essential for oriented deposition of cellulose microfibrils and cell wall strength. The Plant Cell, 14, 3101–3117.
Zhong, R., Burk, D., Morrison, W., & Ye, Z. (2004). Fragile fiber 3, an Arabidopsis gene encoding a type II inositol polyphosphate 5-phosphatase, is required for secondary wall synthesis and actin organization in fiber cells. The Plant Cell, 16, 3242–3259.
Zhong, R., Pena, M., Zhou, G., Nairn, C., Wood-Jones, A., Richardson, E., Morrison, W., Darvill, A., York, W., & Ye, Z. (2005). Arabidopsis fragile fiber 8, which encodes a putative glucuronyltransferase, is essential for normal secondary wall synthesis. The Plant Cell, 17, 3390–3408.
Acknowledgements
We are grateful to Dr. Shinji Kawasaki (NIAS) for providing the rice bc mutant seeds. We thank Ms. Tomoe Nishimoto (Saitama University) for technical assistance. The present study was supported by MEXT Kakenhi Grant Number 18K14386 and 22K05435, Japan.
Author information
Authors and Affiliations
Contributions
A.M. and K.M. wrote the manuscript. A.M., K.M., S.O., S.A., T.O., T.K. and M.K.-Y. designed research. A.M., K.M., S.O., S.A., T.I. and T.O. performed experiments. A.M., K.M., T.I., M.Y., T.K. and M.K.-Y. analysed data. All authors contributed to approve the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors of this manuscript have no competing interests; they do not have any other interests that influence the results and discussion of this paper.
Research involving human and/or animal participants
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Miyagi, A., Mori, K., Ishikawa, T. et al. Metabolomic analysis of rice brittle culm mutants reveals each mutant- specific metabolic pattern in each organ. Metabolomics 18, 95 (2022). https://doi.org/10.1007/s11306-022-01958-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-022-01958-9