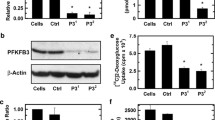
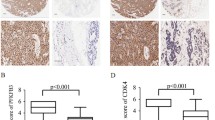
Abstract
Cyclin-dependent kinases CDK4 and CDK6 are essential for the control of the cell cycle through the G1 phase. Aberrant expression of CDK4 and CDK6 is a hallmark of cancer, which would suggest that CDK4 and CDK6 are attractive targets for cancer therapy. Herein, we report that calcein AM (the calcein acetoxymethyl-ester) is a potent specific inhibitor of CDK4 and CDK6 in HCT116 human colon adenocarcinoma cells, inhibiting retinoblastoma protein (pRb) phosphorylation and inducing cell cycle arrest in the G1 phase. The metabolic effects of calcein AM on HCT116 cells were also evaluated and the flux between the oxidative and non-oxidative branches of the pentose phosphate pathway was significantly altered. To elucidate whether these metabolic changes were due to the inhibition of CDK4 and CDK6, we also characterized the metabolic profile of a CDK4, CDK6 and CDK2 triple knockout of mouse embryonic fibroblasts. The results show that the metabolic profile associated with the depletion of CDK4, CDK6 and CDK2 coincides with the metabolic changes induced by calcein AM on HCT116 cells, thus confirming that the inhibition of CDK4 and CDK6 disrupts the balance between the oxidative and non-oxidative branches of the pentose phosphate pathway. Taken together, these results indicate that low doses of calcein can halt cell division and kill tumor cells. Thus, selective inhibition of CDK4 and CDK6 may be of greater pharmacological interest, since inhibitors of these kinases affect both cell cycle progression and the robust metabolic profile of tumors.




Similar content being viewed by others
Abbreviations
- Calcein AM:
-
Calcein acetoxymethyl-ester
- CDK:
-
Cyclin-dependent kinase
- DMEM:
-
Dulbecco’s modified eagle medium
- FCS:
-
Fetal calf serum
- Ct MEF:
-
Mouse embryonic fibroblast
- PBS:
-
Phosphate buffer saline
- PPP:
-
Pentose phosphate pathway
- pRb:
-
Retinoblastoma protein
- TKO MEF:
-
Mouse embryonic fibroblast knockout for CDK4, CDK6 and CDK2
References
Boros, L. G., Puigjaner, J., Cascante, M., Lee, W. N., Brandes, J. L., Bassilian, S., et al. (1997). Oxythiamine and dehydroepiandrosterone inhibit the nonoxidative synthesis of ribose and tumor cell proliferation. Cancer Research, 57, 4242–4248.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
Comin-Anduix, B., Boros, L. G., Marin, S., Boren, J., Callol-Massot, C., Centelles, J. J., et al. (2002). Fermented wheat germ extract inhibits glycolysis/pentose cycle enzymes and induces apoptosis through poly(ADP-ribose) polymerase activation in Jurkat T-cell leukemia tumor cells. Journal of Biological Chemistry, 277, 46408–46414.
Edwards, J. S., Ramakrishna, R., & Palsson, B. O. (2002). Characterizing the metabolic phenotype: a phenotype phase plane analysis. Biotechnology and Bioengineering, 77, 27–36.
Frangioni, J. V., & Neel, B. G. (1993). Solubilization and purification of enzymatically active glutathione S-transferase (pGEX) fusion proteins. Analytical Biochemistry, 210, 179–187.
Fry, D. W., Harvey, P. J., Keller, P. R., Elliott, W. L., Meade, M., Trachet, E., et al. (2004). Specific inhibition of cyclin-dependent kinase 4/6 by PD 0332991 and associated antitumor activity in human tumor xenografts. Molecular Cancer Therapeutics, 3, 1427–1438.
Graf, F., Koehler, L., Kniess, T., Wuest, F., Mosch, B., & Pietzsch, J. (2009). Cell cycle regulating kinase Cdk4 as a potential target for tumor cell treatment and tumor imaging. Journal of Oncology, 2009, 106378.
Hall, M., & Peters, G. (1996). Genetic alterations of cyclins, cyclin-dependent kinases, and Cdk inhibitors in human cancer. Advances in Cancer Research, 68, 67–108.
Harlow, E. & Lane, D. (Eds.). (1988). Antibodies: a laboratory manual (p. 469). New York: Cold Spring Harbor Laboratory Press.
Jonsson, B., Liminga, G., Csoka, K., Fridborg, H., Dhar, S., Nygren, P., et al. (1996). Cytotoxic activity of calcein acetoxymethyl ester (calcein/AM) on primary cultures of human haematological and solid tumours. European Journal of Cancer, 32A, 883–887.
Kuo, W., Lin, J., & Tang, T. K. (2000). Human glucose-6-phosphate dehydrogenase (G6PD) gene transforms NIH 3T3 cells and induces tumors in nude mice. International Journal of Cancer, 85, 857–864.
Laemmli, U. K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227, 680–685.
Landis, M. W., Pawlyk, B. S., Li, T., Sicinski, P., & Hinds, P. W. (2006). Cyclin D1-dependent kinase activity in murine development and mammary tumorigenesis. Cancer Cell, 9, 13–22.
Lee, W. N. P. (2006). Characterizing phenotype with tracer based metabolomics. Metabolomics, 2, 31–39.
Lee, W. N., Boros, L. G., Puigjaner, J., Bassilian, S., Lim, S., & Cascante, M. (1998). Mass isotopomer study of the nonoxidative pathways of the pentose cycle with [1,2–13C2]glucose. American Journal of Physiology, 274, E843–E851.
Lee, W. N., Byerley, L. O., Bergner, E. A., & Edmond, J. (1991). Mass isotopomer analysis: theoretical and practical considerations. Biological Mass Spectrometry, 20, 451–458.
Liminga, G., Jonsson, B., Nygren, P., & Larsson, R. (1999). On the mechanism underlying calcein-induced cytotoxicity. European Journal of Pharmacology, 383, 321–329.
Liminga, G., Martinsson, P., Jonsson, B., Nygren, P., & Larsson, R. (2000). Apoptosis induced by calcein acetoxymethyl ester in the human histiocytic lymphoma cell line U-937 GTB. Biochemical Pharmacology, 60, 1751–1759.
Liminga, G., Nygren, P., Dhar, S., Nilsson, K., & Larsson, R. (1995). Cytotoxic effect of calcein acetoxymethyl ester on human tumor cell lines: drug delivery by intracellular trap**. Anticancer Drugs, 6, 578–585.
Lundberg, A. S., & Weinberg, R. A. (1998). Functional inactivation of the retinoblastoma protein requires sequential modification by at least two distinct cyclin-cdk complexes. Molecular and Cellular Biology, 18, 753–761.
Mahale, S., Aubry, C., Jenkins, P. R., Marechal, J. D., Sutcliffe, M. J., & Chaudhuri, B. (2006). Inhibition of cancer cell growth by cyclin dependent kinase 4 inhibitors synthesized based on the structure of fascaplysin. Bioorganic Chemistry, 34(5), 287–297.
Malumbres, M., & Barbacid, M. (2001). To cycle or not to cycle: a critical decision in cancer. Nature Reviews Cancer, 1, 222–231.
Malumbres, M., & Barbacid, M. (2006). Is cyclin D1-CDK4 kinase a bona fide cancer target? Cancer Cell, 9, 2–4.
Malumbres, M., Sotillo, R., Santamaria, D., Galan, J., Cerezo, A., Ortega, S., et al. (2004). Mammalian cells cycle without the D-type cyclin-dependent kinases Cdk4 and Cdk6. Cell, 118, 493–504.
Marzec, M., Kasprzycka, M., Lai, R., Gladden, A. B., Wlodarski, P., Tomczak, E., et al. (2006). Mantle cell lymphoma cells express predominantly cyclin D1a isoform and are highly sensitive to selective inhibition of CDK4 kinase activity. Blood, 108, 1744–1750.
Matito, C., Mastorakou, F., Centelles, J. J., Torres, J. L., & Cascante, M. (2003). Antiproliferative effect of antioxidant polyphenols from grape in murine Hepa-1c1c7. European Journal of Nutrition, 42, 43–49.
McInnes, C. (2008). Progress in the evaluation of CDK inhibitors as anti-tumor agents. Drug Discovery Today, 13, 875–881.
Menu, E., Garcia, J., Huang, X., Di Liberto, M., Toogood, P. L., Chen, I., et al. (2008). A novel therapeutic combination using PD 0332991 and bortezomib: study in the 5T33MM myeloma model. Cancer Research, 68, 5519–5523.
Mosmann, T. (1983). Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. Journal of Immunological Methods, 65, 55–63.
Myohanen, S. K., Baylin, S. B., & Herman, J. G. (1998). Hypermethylation can selectively silence individual p16ink4A alleles in neoplasia. Cancer Research, 58, 591–593.
Poulsen, H. S., & Frederiksen, P. (1981). Glucose-6-phosphate dehydrogenase activity in human breast cancer. Lack of association with oestrogen receptor content. Acta Pathol Microbiol Scand [A], 89, 263–270.
Ramos-Montoya, A., Lee, W.-N. P., Bassilian, S., Lim, S., Trebukhina, R. V., Kazhyna, M. V., et al. (2006). Pentose phosphate cycle oxidative and non-oxidative balance: a new vulnerable target for overcoming drug resistance in cancer. International Journal of Cancer, 119, 2733–2741.
Rubio-Martinez, J., Pinto, M., Tomas M.S., Perez, J. J. (2005). Dock_Dyn: a program for fast molecular docking using molecular dynamics information. University of Barcelona and Technical University of Catalonia.
Santamaria, D., & Ortega, S. (2006). Cyclins and CDKS in development and cancer: lessons from genetically modified mice. Frontiers in Bioscience, 11, 1164–1188.
Shapiro, G. I. (2006). Cyclin-dependent kinase pathways as targets for cancer treatment. Journal of Clinical Oncology, 24, 1770–1783.
Sherr, C. J. (1996). Cancer cell cycles. Science, 274, 1672–1677.
Sherr, C. J., & Roberts, J. M. (2004). Living with or without cyclins and cyclin-dependent kinases. Genes and Development, 18, 2699–2711.
Smith, D. B., & Johnson, K. S. (1988). Single-step purification of polypeptides expressed in Escherichia coli as fusions with glutathione S-transferase. Gene, 67, 31–40.
Villacanas, O., Perez, J. J., & Rubio-Martinez, J. (2002). Structural analysis of the inhibition of Cdk4 and Cdk6 by p16(INK4a) through molecular dynamics simulations. Journal of Biomolecular Structure and Dynamics, 20, 347–358.
Villacanas, O., & Rubio-Martinez, J. (2006). Reducing CDK4/6–p16(INK4a) interface. Computational alanine scanning of a peptide bound to CDK6 protein. Proteins, 63, 797–810.
Vizan, P., Alcarraz-Vizán, G., Diaz-Moralli, S., Rodriguez-Prados, J. C., Zanuy, M., Centelles, J. J., et al. (2007). Quantification of intracellular phosphorylated carbohydrates in HT29 human colon adenocarcinoma cell line using liquid chromatography-electrospray ionization tandem mass spectrometry. Analytical Chemistry, 79(13), 5000–5005.
Vizan, P., Alcarraz-Vizan, G., Diaz-Moralli, S., Solovjeva, O. N., Frederiks, W. M., & Cascante, M. (2009). Modulation of pentose phosphate pathway during cell cycle progression in human colon adenocarcinoma cell line HT29. International Journal of Cancer, 124, 2789–2796.
Vizán, P., Mazurek, S., & Cascante, M. (2008). Robust metabolic adaptation underlying tumor progression. Metabolomics, 4, 1–12.
Warburg, O. (1956). Origin of cancer cells. Oncologia, 9, 75–83.
Yu, Q., Sicinska, E., Geng, Y., Ahnstrom, M., Zagozdzon, A., Kong, Y., et al. (2006). Requirement for CDK4 kinase function in breast cancer. Cancer Cell, 9, 23–32.
Acknowledgments
The authors thank Mrs Ursula Valls for her technical support in the experiments and Dr David Santamaria for his help in MEF procedures. MEF cells were a generous gift from Dr Mariano Barbacid, CNIO-Madrid (Spain). This study was supported by the projects SAF2008-00164 (to MC) and SAF2007-60491 (to NA) and by RD06/0020/0046 (to MC), RD06/0020/0010 (to OB) from Red Temática de Investigación Cooperativa en Cáncer (RTICC), Instituto de Salud Carlos III, all of them funded by the Ministerio de Ciencia e Innovación-Spanish government and European Regional Development Funds (ERDF) “Una manera de hacer Europa”. It has also received financial support from the European Union-funded project ETHERPATHS (FP7-KBBE-222639) (http://www.etherpaths.org/) and from the Agència de Gestió d’Ajuts Universitaris i de Recerca (AGAUR)-Generalitat de Catalunya (2009SGR01308 and predoctoral fellowship of M.Z.). Mass spectrometry facility was supported by NIH grants to WNP Lee from UCLA Center of Excellence (PO1 AT003960-01) and from Harbor-UCLA GCRC (MO1 RR00425-33). MC acknowledges the support received through the prize “ICREA Academia” for excellence in research, funded by ICREA foundation-Generalitat de Catalunya.
Author information
Authors and Affiliations
Corresponding author
Additional information
Miriam Zanuy and Antonio Ramos-Montoya contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zanuy, M., Ramos-Montoya, A., Villacañas, O. et al. Cyclin-dependent kinases 4 and 6 control tumor progression and direct glucose oxidation in the pentose cycle. Metabolomics 8, 454–464 (2012). https://doi.org/10.1007/s11306-011-0328-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11306-011-0328-x