Abstract
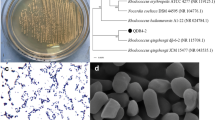
At present, the study on exopolysaccharid is mainly focused on lactic acid bacteria, and the research on exopolysaccharide produced by yeast, especially Sporidiobolus pararoseus, is relatively few. Therefore, the aim of this study was to explore the characterization and antioxidant activities of a novel neutral exopolysaccharide SPZ, which was isolated and purified from S. pararoseus PFY-Z1. The results showed that SPZ was mainly composed of mannose, followed by glucose, with a molecular weight was 24.98 kDa, had O-glycosidic bonds, no crystalline, and no triple helix structure. Based on fourier transform-infrared, high-performance liquid chromatography and nuclear magnetic resonance analyses, SPZ was identified to be a exopolysaccharide with some side chains, presence of α-, β-pyranose ring and nine sugar residues. Furthermore, the morphology features of SPZ have performed a relatively rough and uneven surface, covered with small pores and fissures. Moreover, SPZ had higher antioxidant activities and the maximum scavenging abilities of ⋅OH, NO2− and reducing power were 28.05 ± 0.73%, 92.76 ± 1.86% and 0.345 ± 0.024, respectively. Hence, SPZ could be used as a potential antioxidant application in the food and pharmaceutical industries.







Similar content being viewed by others
References
Alban S, Schauerte A, Franz G (2002) Anticoagulant sulfated polysaccharides: part I. Synthesis and structure-activity relationships of new pullulan sulfates. Carbohyd Polym 47:267–276. https://doi.org/10.1016/S0144-8617(01)00178-3
Bai YY, Luo BL, Zhang Y, Li X, Wang Z, Shan YQ, Lu M, Tian FW, Ni YQ (2021) Exopolysaccharides produced by Pediococcus acidilactici MT41-11 isolated from camel milk: structural characteristics and bioactive properties. Int J Biol Macromol 185:1036–1049. https://doi.org/10.1016/j.ijbiomac.2021.06.152
Bitter T, Muir HM (1962) A modified uronic acid carbazole reaction. Anal Biochem 4:330–334. https://doi.org/10.1016/0003-2697(62)90095-7
Bouallegue A, Casillo A, Chaari F, Gatta AL, Lanzetta R, Corsaro MM, Bachoual R, Ellouz-Chaabouni S (2020) Levan from a new isolated Bacillus subtilis AF17: purification, structural analysis and antioxidant activities. Int J Biol Macromol 144:316–324. https://doi.org/10.1016/j.ijbiomac.2019.12.108
Chen JW, Meng Q, Jiang B, Chen JJ, Zhang T, Zhou LC (2021) Structure characterization and in vitro hypoglycemic effect of partially degraded alginate. Food Chem 356:129728. https://doi.org/10.1016/j.foodchem.2021.129728
Chen ZN, Shi JL, Yang XJ, Liu Y, Nan B, Wang ZF (2016) Isolation of exopolysaccharide-producing bacteria and yeasts from Tibetan Kefir and characterisation of the exopolysaccharides. Int J Dairy Technol 69:410–417. https://doi.org/10.1111/1471-0307.12276
Chi Z, Zhao S (2003) Optimization of medium and cultivation conditions for pullulan production by a new pullulan-producing yeast strain. Enzyme Microb Tech 33:206–211. https://doi.org/10.1016/S0141-0229(03)00119-4
Choudhuri I, Khanra K, Maity P, Patra A, Maity GN, Pati BR, Nag A, Mondal S, Bhattacharyya N (2021) Structure and biological properties of exopolysaccharide isolated from Citrobacter freundii. Int J Biol Macromol 168:537–549. https://doi.org/10.1016/j.ijbiomac.2020.12.063
Dong YH, Chun C, Huang Q, Fu X (2021) Study on a novel spherical polysaccharide from Fructus Mori with good antioxidant activity. Carbohyd Polym 256:117516. https://doi.org/10.1016/j.carbpol.2020.117516
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–366. https://doi.org/10.1021/ac60111a017.1
El-Dein AN, El-Deen AMN, El-Shatoury EH, Awad GA, Ibrahim MK, Awad HM, Farid MA (2021) Assessment of exopolysaccharides, bacteriocins and in vitro and in vivo hypocholesterolemic potential of some Egyptian Lactobacillus spp. Int J Biol Macromol 173:66–78. https://doi.org/10.1016/j.ijbiomac.2021.01.107
Gan LG, Jiang GY, Li XG, Zhang SH, Tian YQ, Peng BY (2021) Structural elucidation and physicochemical characteristics of a novel high-molecular-weight fructan from halotolerant Bacillus sp. SCU-E108. Food Chem 365:130496. https://doi.org/10.1016/j.foodchem.2021.130496
Ghada SI, Manal GM, Mohsen MSA, Pr EAG (2012) Production and biological evaluation of exopolysaccharide from isolated Rhodotorula glutinins. Aust J Basic Appl Sci 6:401–408
Grineva VS, Tregubova KV, Anis’kov AA, Sigida EN, Shirokov AA, Fedonenko YP, Yegorenkov IV (2020) Isolation, structure, and potential biotechnological applications of the exopolysaccharide from Paenibacillus polymyxa 92. Carbohyd Polym 232:115780. https://doi.org/10.1016/j.carbpol.2019.115780
Gu YG, Qiu Y, Wei X, Li Z, Hu ZQ, Gu YY, Zhao YZ, Wang YD, Yue TL, Yuan YH (2020) Characterization of selenium-containing polysaccharides isolated from selenium-enriched tea and its bioactivities. Food Chem 316:126371. https://doi.org/10.1016/j.foodchem.2020.126371
Hamidi M, Gholipour AR, Delattre C, Sesdighi F, Seveiri RM, Pasdara A, Kheirandish S, Pierre G, Kozani PS, Kozani PS, Karimitabar F (2020) Production, characterization and biological activities of exopolysaccharides from a new cold-adapted yeast: Rhodotorula mucilaginosa sp. GUMS16. Int J Biol Macromol 151:268–277. https://doi.org/10.1016/j.ijbiomac.2020.02.206
Han M, Xu JZ, Liu ZM, Qin H, Zhang WG (2018) Co-production of microbial oil and exopolysaccharide by the oleaginous yeast Sporidiobolus pararoseusgrown in fed-batch culture. Rsc Adv 8:3348–3356. https://doi.org/10.1039/c7ra12813d
Jeddou KB, Chaari F, Maktouf S, Nouri-Ellouz O, Helbert CB, Ghorbel RE (2016) Structural, functional, and antioxidant properties of water-soluble polysaccharides from potatoes peels. Food Chem 205:97–105. https://doi.org/10.1016/j.foodchem.2016.02.108
Jia YN, Xue ZH, Wang YJ, Lu YP, Li RL, Li NN, Wang Q, Zhang M, Chen HX (2021) Chemical structure and inhibition on α-glucosidase of polysaccharides from corn silk by fractional precipitation. Carbohyd Polym 252:117185. https://doi.org/10.1016/j.carbpol.2020.117185
Jiang J, Guo SX, ** WX, Zhao D, Ge JP (2020) Optimization production of exopolysaccharide from Leuconostoc lactis L2 and its partial characterization. Int J Biol Macromol 159:630–639. https://doi.org/10.1016/j.ijbiomac.2020.05.101
Kawai Y, Seno N, Anno K (1969) A modified method for chondrosulfatase assay. Anal Biochem 32:314–321. https://doi.org/10.1016/0003-2697(69)90091-8
Kim KN, Lee YJ, Cho DH, Ahnu KH, Hong JE, Park YI, Kim HS (2020) A novel galactoglucomannan exopolysaccharide produced by oil fermentation with Pseudozyma sp. SY16. Biotechnol Bioproc E 25:742–748. https://doi.org/10.1007/s12257-020-0066-x
Kot AM, Kieliszek M, Piwowarek K, Błażejak S, Mussagya CU (2021) Sporobolomyces and Sporidioboluse non-conventional yeasts for use in industries. Fungal Biol Rev 37:41–58. https://doi.org/10.1016/j.fbr.2021.06.001
Li SS, **a HQ, **e AQ, Wang ZY, Ling KJ, Zhang QX, Zou X (2020a) Structure of a fucose-rich polysaccharide derived from EPS produced by Kosakonia sp. CCTCC M2018092 and its application in antibacterialfilm. Int J Biol Macromol 159:295–303. https://doi.org/10.1016/j.ijbiomac.2020.05.029
Li XW, Lv S, Shi TT, Liu K, Li QM, Pan LH, Zha XQ, Luo JP (2020b) Exopolysaccharides from yoghurt fermented by Lactobacillus paracasei: production, purification and its binding to sodium caseinate. Food Hydrocolloid 102:105635. https://doi.org/10.1016/j.foodhyd.2019.105635
Li X, Wei ZL, Wang XY, Duan FX, **ong LD, Li JW, Tian J, Jia LR, Gao H (2021) Premna microphylla Turcz leaf pectin exhibited antioxidant and anti-inflammatory activities in LPS-stimulated RAW 264.7 macrophages. Food Chem 349:129164. https://doi.org/10.1016/j.foodchem.2021.129164
Liu LN, Xu JJ, Du RP, ** WX, Ge JP, Zhao D (2021) The response surface optimization of exopolysaccharide produced by Saccharomyces cerevisiae Y3 and its partial characterization. Prep Biochem Biotech 52:566–577. https://doi.org/10.1080/10826068.2021.1972428
Liu LN, Xu JJ, Na RY, Du RP, ** WX, Ge JP (2022) Purification, characterization and partial biological activities of exopolysaccharide produced by Saccharomyces cerevisiae Y3. Int J Biol Macromol 206:777–787. https://doi.org/10.1016/j.ijbiomac.2022.03.083
Liu T, Zhou K, Yin S, Liu SL, Zhu YT, Yang Y, Wang CT (2019) Purification and characterization of an exopolysaccharide produced by Lactobacillus plantarum HY isolated from home-made Sichuan pickle. Int J Biol Macromol 134:516–526. https://doi.org/10.1016/j.ijbiomac.2019.05.010
Ma WJ, Chen XF, Wang B, Lou WJ, Chen X, Hua JL, Sun YJ, Zhao Y, Peng T (2018) Characterization, antioxidativity, and anti-carcinoma activity of exopolysaccharide extract from Rhodotorula mucilaginosa CICC 33013. Carbohyd Polym 181:768–777. https://doi.org/10.1016/j.carbpol.2017.11.080
Mao YH, Song AX, Li LQ, Yang Y, Yao ZP, Wu JY (2020) A high-molecular weight exopolysaccharide from the Cs-HK1 fungus: ultrasonic degradation, characterization and in vitro fecal fermentation. Carbohyd Polym 246:116636. https://doi.org/10.1016/j.carbpol.2020.116636
Mathivanan K, Chandirika JU, Mathimani T, Vinothkanna A, Rajaram R, Annadurai G (2020) Optimization, compositional analysis, and characterization of exopolysaccharides produced by multi-metal resistant Bacillus cereus KMS3-1. Carbohyd Polym 227:115369. https://doi.org/10.1016/j.carbpol.2019.115369
Mehwish HM, Liu G, Riaz Rajoka MS, Cai HM, Zhong JF, Song X, **a LX, Wang MZ, Aadil RM, Raheem MIU, **ong YG, Wu HQ, Amirzada MI, Zhu QC, He ZD (2021) Therapeutic potential of Moringa oleifera seed polysaccharide embedded silver nanoparticles in wound healing. Int J Biol Macromol 184:144–158. https://doi.org/10.1016/j.ijbiomac.2021.05.202
Mohammed YYM, Saad MMG, Abdelgaleil SAM (2021) Production, characterization and bio-emulsifying application of exopolysaccharides from Rhodotorula mucilaginosa YMM19. 3 Biotech 11:349. https://doi.org/10.1007/s13205-021-02898-2
Osińska-Jaroszuk M, Jarosz-Wilkołazka A, Jaroszuk-Ściseł J, Szałapata K, Nowak A, Jaszek M, Ozimek E, Majewska M (2015) Extracellular polysaccharides from Ascomycota and Basidiomycota: production conditions, biochemical characteristics, and biological properties. World J Microbiol Biotechnol 31:1823–1844. https://doi.org/10.1007/s11274-015-1937-8
Pei FY, Ma YS, Chen X, Liu H (2020) Purification and structural characterization and antioxidant activity of levan from Bacillus megaterium PFY-147. Int J Biol Macromol 161:1181–1188. https://doi.org/10.1016/j.ijbiomac.2020.06.140
Pei FY, Cao XB, Wang XM, Ren YX, Ge JP (2022a) Structural characteristics and bioactivities of polysaccharides from blue honeysuckle after probiotic fermentation. LWT-Food Sci Technol 165:113764. https://doi.org/10.1016/j.lwt.2022.113764
Pei FY, Lv YZ, Cao XB, Wang XM, Ren YX, Ge JP (2022b) Structural characteristics and the antioxidant and hypoglycemic activities of a polysaccharide from Lonicera caerulea L. Pomace Ferment 8:422. https://doi.org/10.3390/fermentation8090422
Poli A, Anzelmo G, Tommonaro G, Pavlova K, Pavlova A, Nicolaus B (2010) Production and chemical characterization of an exopolysaccharide synthesized by psychrophilic yeast strain Sporobolomyces salmonicolor AL1 isolated from Livingston Island Antarctica. Folia Microbiol 55:576–581. https://doi.org/10.1007/s12223-010-0092-8
Riaz Rajoka MS, Wu YG, Mehwish HM, Bansal M, Zhao LQ (2020) Lactobacillus exopolysaccharides: new perspectives on engineering strategies, physiochemical functions, and immunomodulatory effects on host health. Trends Food Sci Tech 103:36–48. https://doi.org/10.1016/j.tifs.2020.06.003
Riaz Rajoka MS, Thirumdas R, Mehwish HM, Umair M, Khurshid M, Hayat HF, PhimolsiripolXPallarés YN, Martí-Quijal FJ, Barba FJ (2021) Role of food antioxidants in modulating gut microbial communities: novel understandings in intestinal oxidative stress damage and their impact on host health. Antioxidants 10:1563. https://doi.org/10.3390/antiox10101563
Riaz Rajoka MS, Mehwish HM, Kitazawa H, Barba FJ, Berthelot L, Umair M, Zhu QC, He ZD, Zhao LQ (2022) Techno-functional properties and immunomodulatory potential of exopolysaccharide from Lactiplantibacillus plantarum MM89 isolated from human breast milk. Food Chem 377:131954. https://doi.org/10.1016/j.foodchem.2021.131954
Rusinova-Videva S, Pavlova K, Panchev I, Georgieva K, Kuncheva M (2010) Effect of different factors on biosynthesis of exopolysaccharide from antarctic yeast. Biotechnol Biotechnol Equip 24:507–511. https://doi.org/10.1080/13102818.2010.10817891
Saadat YR, Khosroushahi AY, Movassaghpour AA, Talebi M, Gargari BP (2020) Modulatory role of exopolysaccharides of Kluyveromyces marxianus and Pichia kudriavzevii as probiotic yeasts from dairy products in human colon cancer cells. J Funct Foods 64:103675. https://doi.org/10.1016/j.jff.2019.103675
Saadat YR, Khosroushahi AY, Gargari BP (2021) Yeast exopolysaccharides and their physiological functions. Folia Microbiol 66:171–182. https://doi.org/10.1007/s12223-021-00856-2
Saleem M, Malik S, Mehwish HM, Ali ME, Hussain N, Khurshid M, Riaz Rajoka MS, Chen YG (2021) Isolation and functional characterization of exopolysaccharide produced by Lactobacillus plantarum S123 isolated from traditional Chinese cheese. Arch Microbiol 203:3061–3070. https://doi.org/10.1007/s00203-021-02291-w
Seyfi R, Kasaai MR, Chaichi MJ (2019) Isolation and structural characterization of a polysaccharide derived from a local gum: Zedo (Amygdalus scoparia Spach). Food Hydrocolloid 87:915–924. https://doi.org/10.1016/j.foodhyd.2018.09.017
Silambarasan S, Logeswari P, Cornejo P, Kannan VR (2019) Evaluation of the production of exopolysaccharide by plant growth promoting yeast Rhodotorula sp. strain CAH2 under abiotic stress conditions. Int J Biol Macromol 121:55–62. https://doi.org/10.1016/j.ijbiomac.2018.10.016
Sun XQ, Wang L, Fu RJ, Yang YX, Cheng R, Li J, Wang SM, Zhang JF (2020) The chemical properties and hygroscopic activity of the exopolysaccharide lubcan from Paenibacillus sp. ZX1905. Int J Biol Macromol 164:2641–2650. https://doi.org/10.1016/j.ijbiomac.2020.08.129
Tan C, Wang J, Sun BG (2021) Polysaccharide dual coating of yeast capsules for stabilization of anthocyanins. Food Chem 357:129652. https://doi.org/10.1016/j.foodchem.2021.129652
Wang Y, Du RP, Qiao XX, Zhao B, Zhou ZJ, Han Y (2020) Optimization and characterization of exopolysaccharides with a highly branched structure extracted from Leuconostoc citreum B-2. Int J Biol Macromol 142:73–84. https://doi.org/10.1016/j.ijbiomac.2019.09.071
Wu DT, He Y, Fu MX, Gan RY, Hu YC, Peng LX, Zhao G, Zou L (2022) Structural characteristics and biological activities of a pectic-polysaccharide from okra affected by ultrasound assisted metal-free Fenton reaction. Food Hydrocolloid 122:107085. https://doi.org/10.1016/j.foodhyd.2021.107085
Wu MQ, Li W, Zhang YL, Shi L, Xu ZZ, **a W, Zhang WQ (2021) Structure characteristics, hypoglycemic and immunomodulatory activities of pectic polysaccharides from Rosa setate x Rosa rugosa waste. Carbohydr Polym 253:117190. https://doi.org/10.1016/j.carbpol.2020.117190
Zhang H, Zou P, Zhao HT, Qiu JQ, Regenstein JM, Yang X (2021) Isolation, purification, structure and antioxidant activity of polysaccharide from pinecones of Pinus koraiensis. Carbohyd Polym 251:117078. https://doi.org/10.1016/j.carbpol.2020.117078
Zhang M, Zeng SY, Hao LY, Yao SJ, Wang DK, Yang H, Wu CD (2022) Structural characterization and bioactivity of novel exopolysaccharides produced by Tetragenococcus Halophilus. Food Res Int 155:111083. https://doi.org/10.1016/j.foodres.2022.111083
Zhu YT, Wang CT, Jia SS, Wang BY, Zhou K, Chen SJ, Yang Y, Liu SL (2018) Purification, characterization and antioxidant activity of the exopolysaccharide from Weissella cibaria SJ14 isolated from Sichuan paocai. Int J Biol Macromol 115:820–828. https://doi.org/10.1016/j.ijbiomac.2018.04.067
Acknowledgements
All appreciation to Qiqihar Medical University, Education Department of Heilongjiang Province, and Qiqihar Science and Technology Bureau. This work was supported by the Fund project of Qiqihar Academy of Medical Sciences (Grant no. [QMSI 2020M-07]), Foundation of Heilongjiang Educational Committee (Grant no. [2021-KYYWF-0381]), and Qiqihar Science and Technology Plan Joint Guidance Project (Grant no. [LSFGG-2022037]).
Funding
This work was supported by the Fund project of Qiqihar Academy of Medical Sciences (Grant No. [QMSI 2020 M-07]), Foundation of Heilongjiang Educational Committee (Grant No. [2021-KYYWF-0381]), and Qiqihar Science and Technology Plan Joint Guidance Project (Grant No. [LSFGG-2022037]).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Zhenyan Liu, Fangyi Pei, Di Xue, Yuchao Liu, **feng Zhu, Deshui Liu and Hui Li. The first draft of the manuscript was written by Fangyi Pei and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Declarations
All authors have approved the final version of the manuscript. All authors have read and agreed to the published version of the manuscript.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Z., Pei, F., Zhu, J. et al. Production, characterization and antioxidant activity of exopolysaccharide from Sporidiobolus pararoseus PFY-Z1. World J Microbiol Biotechnol 39, 10 (2023). https://doi.org/10.1007/s11274-022-03453-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-022-03453-8