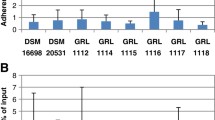
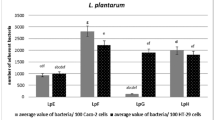
Abstract
Surface properties like hydrophobicity, aggregation ability, adhesion to mucosal surfaces and epithelial cells and transit time are key features for the characterization of probiotic strains. In this study, we used two Lactobacillus paracasei subsp. paracasei strains (BGNJ1-64 and BGSJ2-8) strains which were previously described with very strong aggregation capacity. The aggregation promoting factor (AggLb) expressed in these strains showed high level of binding to collagen and fibronectin, components of extracellular matrix. The working hypothesis was that strains able to aggregate have an advantage to resist in intestinal tract. So, we assessed whether these strains and their derivatives (without aggLb gene) are able to bind or not to intestinal components and we compared the transit time of each strains in mice. In that purpose parental strains (BGNJ1-64 and BGSJ2-8) and their aggregation negative derivatives (BGNJ1-641 and BGSJ2-83) were marked with double antibiotic resistance in order to be tracked in in vivo experiments in mice. Comparative analysis of binding ability of WT and aggregation negative strains to different human intestinal cell lines and mucin revealed no significant difference among them, excluding involvement of AggLb in interaction with surface of intestinal cells and mucin. In vivo experiments showed that surviving and transit time of marked strains in mice did not drastically depend on the presence of the AggLb aggregation factor.




Similar content being viewed by others
References
Bogovic-Matijasevic B, Narat M, Zoric M (2003) Adhesion of L. gasseri strains on Caco-2 cells. Food Technol Biotechnol 41(1):83–88
Chew SY, Cheah YK, Seow HF, Sandai D, Than L (2015) Probiotic Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14 exhibit strong antifungal effects against vulvovaginal candidiasis-causing Candida glabrata isolates. J Appl Microbiol 118(5):1180–1190. https://doi.org/10.1111/jam.12772
Dar HY, Shukla P, Mishra PK, Anupam R, Mondal RK, Tomar GB, Sharma V, Srivastava RK (2018) Lactobacillus acidophilus inhibits bone loss and increases bone heterogeneity in osteoporotic mice via modulating Treg-Th17 cell balance. Bone Rep 8:46–56. https://doi.org/10.1016/j.bonr.2018.02.001
Esmaeili SA, Mahmoudi M, Rezaieyazdi Z, Sahebari M, Tabasi N, Sahebkar A, Rastin M (2018) Generation of tolerogenic dendritic cells using Lactobacillus rhamnosus and Lactobacillus delbrueckii as tolerogenic probiotics. J Cell Biochem 119(9):7865–7872. https://doi.org/10.1002/jcb.27203
García-Cayuela T, Korany AM, Bustos I, Gómez de Cadiñanos LP, Requena T et al (2014) Adhesion abilities of dairy Lactobacillus plantarum strains showing an aggregation phenotype. Food Res Int 57:44–50. https://doi.org/10.1016/j.foodres.2014.01.010
George F, Daniel C, Thomas M, Singer E, Guilbaud A, Tessier FJ, Revol-Junelles AM, Borges F, Foligné B (2018) Occurrence and dynamism of lactic acid bacteria in distinct ecological niches: a multifaceted functional health perspective (eCollection 2018, Review). Front Microbiol 9:2899. https://doi.org/10.3389/fmicb.2018.02899
Gerritsen J, Smidt H, Rijkers GT, de Vos WM (2011) Intestinal microbiota in human health and disease: the impact of probiotics. Genes Nutr 6(3):209–240. https://doi.org/10.1007/s12263-011-0229-7
Kechaou N, Chain F, Gratadoux JJ, Blugeon S, Bertho N, Chevalier C, Le Goffic R, Courau S, Molimard P, Chatel JM, Langella P, Bermudez-Humaran LG (2013) Identification of one novel candidate probiotic Lactobacillus plantarum strain active against influenza virus infection in mice by a large-scale screening. Appl Environ Microbiol 79(5):1491–1499. https://doi.org/10.1128/AEM.03075-12
Kojic M, Strahinic I, Topisirovic L (2005) Proteinase PI and lactococcin A genes are located on the largest plasmid in Lactococcus lactis subsp. lactis bv. diacetylactis S50. Can J Microbiol 51:305–314. https://doi.org/10.1139/w05-009
Kojic M, Strahinic I, Fira D, Jovcic B, Topisirovic L (2006) Plasmid content and bacteriocin production by five strains of Lactococcus lactis isolated from semi-hard homemade cheese. Can J Microbiol 52:1110–1120. https://doi.org/10.1139/w06-072
Kojic M, Jovcic B, Strahinic I, Begovic J, Lozo J, Veljovic K, Topisirovic L (2011) Cloning and expression of novel lactococcal aggregation factor from Lactococcus lactis subsp. lactis BGKP1. BMC Microbiol 11:265. https://doi.org/10.1111/jam.12772
Kravtsov EG, Yermolayev AV, Anokhina IV, Yashina NV, Chesnokova VL, Dalin MV (2008) Adhesion characteristics of Lactobacillus is a criterion of the probiotic choice. Bull Exp Biol Med 145:232–234
Lozo J, Jovcic B, Kojic M, Dalgalarrondo M, Chobert JM, Haertlé T, Topisirovic L (2007) Molecular characterization of a novel bacteriocin and an unusually large aggregation factor of Lactobacillus paracasei subsp. paracasei BGSJ2-8, a natural isolate from homemade cheese. Curr Microbiol 55:266–271. https://doi.org/10.1007/s00284-007-0159-1
Lukic J, Strahinić I, Jovčić B, Filipić B, Topisirović L, Kojić M, Begović J (2012) Different roles for lactococcal aggregation factor and mucin binding protein in adhesion to gastrointestinal mucosa. Appl Environ Microbiol 78(22):7993–8000. https://doi.org/10.1128/AEM.02141-12
Lukic J, Strahinic I, Milenkovic M, Nikolic M, Tolinacki M, Kojic M, Begovic J (2014) Aggregation factor as an inhibitor of bacterial binding to gut mucosa. Microb Ecol 68(3):633–644. https://doi.org/10.1007/s00248-014-0426-1
McNaught C, MacFie J (2001) Probiotics in clinical practice: a critical review of the evidence. Nutr Res 21:343–353
Messaoudi S, Manai M, Kergourlay G, Prévost H, Connil N, Chobert JM, Dousset X (2013) Lactobacillus salivarius: bacteriocin and probiotic activity. Food Microbiol 36(2):296–304. https://doi.org/10.1016/j.fm.2013.05.010
Miljkovic M, Strahinic I, Tolinacki M, Zivkovic M, Kojic S, Golic N, Kojic M (2015) AggLb is the largest cell-aggregation factor from Lactobacillus paracasei subsp. paracasei BGNJ1–64, functions in collagen adhesion, and pathogen exclusion in vitro. PLoS ONE 10:e0126387. https://doi.org/10.1371/journal.pone.0126387.
Miljkovic M, Bertani I, Fira D, Jovcic B, Novovic K, Venturi V, Kojic M (2016) Shortening of the Lactobacillus paracasei subsp. paracasei BGNJ1–64 AggLb protein switches its activity from auto-aggregation to biofilm formation. Front Microbiol 7:1422. https://doi.org/10.3389/fmicb.2016.01422
Miljkovic M, Marinkovic P, Novovic K, Jovcic B, Terzic-Vidojevic A, Kojic M (2018) AggLr, a novel aggregation factor in Lactococcus raffinolactis BGTRK10-1: its role in surface adhesion. Biofouling 34(6):685–698. https://doi.org/10.1080/08927014.2018.1481956
Miquel S, Beaumont M, Martín R, Langella P, Braesco V, Thomas M (2015) A proposed framework for an appropriate evaluation scheme for microorganisms as novel foods with a health claim in Europe. Microb Cell Fact 14:48. https://doi.org/10.1186/s12934-015-0229-1
Selvam RM, Vinothini G, Thaiyammal SP, Latha S, Chinnathambi A, Dhanasekaran D, Padmanabhan P, Alharbi SA, Archunan G (2016) The cell aggregating propensity of probiotic actinobacterial isolates: isolation and characterization of the aggregation inducing peptide pheromone. Biofouling 32(1):71–79. https://doi.org/10.1080/08927014.2015.1122759
Radziwill-Bienkowska JM, Robert V, Drabot K, Chain F, Cherbuy C, Langella P, Thomas M, Bardowski JK, Mercier-Bonin M, Kowalczyk M (2017) Contribution of plasmid-encoded peptidase S8 (PrtP) to adhesion and transit in the gut of Lactococcus lactis IBB477 strain. Appl Microbiol Biotechnol 101:5709–5721. https://doi.org/10.1007/s00253-017-8334-1
Rajoka MS, Zhao H, Lu Y, Lian Z, Li N, Hussain N, Shao D, ** M, Li Q, Shi J (2018) Anticancer potential against cervix cancer (HeLa) cell line of probiotic Lactobacillus casei and Lactobacillus paracasei strains isolated from human breast milk. Food Funct 9(5):2705–2715. https://doi.org/10.1039/c8fo00547h
Tuomola EM, Salminen SJ (1998) Adhesion of some probiotic and dairy Lactobacillus strains to Caco-2 cell cultures. Int J Food Microbiol 41(1):45–51. https://doi.org/10.1016/S0168-1605(98)00033-6
Turpin W, Humblot C, Noordine M-L, Wrzosek L, Tomas J, Mayeur C, Chetbuy C, Guyot JP, Thomas M (2013) Behavior of lactobacilli isolated from fermented slurry (ben-saalga) in gnotobiotic rats. PLoS ONE 8(4):e57711. https://doi.org/10.1371/journal.pone.0057711
Van Zyl WF, Deane SM, Dicks LMT (2018) In vivo bioluminescence imaging of the spatial and temporal colonization of Lactobacillus plantarum 423 and Enterococcus mundtii ST4SA in the intestinal tract of mice. BMC Microbiol 18(1):171. https://doi.org/10.1186/s12866-018-1315-4
Veljović K, Popović N, Miljković M, Tolinački M, Terzić-Vidojević A, Kojić M (2017) Novel aggregation promoting factor AggE contributes to the probiotic properties of Enterococcus faecium BGGO9-28. Front Microbiol 8:1843. https://doi.org/10.3389/fmicb.2017.01843
Vinusha KS, Deepika K, Johnson TS, Agrawal GK, Rakwal R (2018) Proteomic studies on lactic acid bacteria: a review. Biochem Biophys Rep 14:140–148. https://doi.org/10.1016/j.bbrep.2018.04.009
Walter J (2008) Ecological role of lactobacilli in the gastrointestinal tract: implications for fundamental and biomedical research. Appl Environ Microbiol 74(16):4985–4996. https://doi.org/10.1128/AEM.00753-08
Waśko A, Polak-Berecka M, Paduch R, Jóźwiak K (2014) The effect of moonlighting proteins on the adhesion and aggregation ability of Lactobacillus helveticus. Anaerobe 30:161–168. https://doi.org/10.1016/j.anaerobe.2014.10.002
Yang DC, Blair KM, Salama NR (2016) Staying in shape: the impact of cell shape on bacterial survival in diverse environments. Microbiol Mol Biol Rev 80:187–203. https://doi.org/10.1128/MMBR.00031-15
Acknowledgements
This work was co-supported by The Ministry of Education, Science and Technological Development of the Republic of Serbia, Republic of Serbia (Grant No. 173019 and Grant for Co-financing of Postdoctoral Training of Researchers in 2017).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. Mice experiments were carried out in accordance with the European guidelines for the care and use of laboratory animals (Directive 2010/63/UE). The studies received ethical approval from the local ethics committee (COMETHEA), authorized by the Ministry of Education of Higher Education and Research for the period 2015–2018 (APAFIS # 00680.01). The animal facility was accredited by the Direction des Services Vétérinaires (reference A78-187).
Rights and permissions
About this article
Cite this article
Miljkovic, M., Thomas, M., Serror, P. et al. Binding activity to intestinal cells and transient colonization in mice of two Lactobacillus paracasei subsp. paracasei strains with high aggregation potential. World J Microbiol Biotechnol 35, 85 (2019). https://doi.org/10.1007/s11274-019-2663-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-019-2663-4