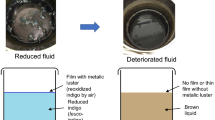
Abstract
Although the indigo reduction process is performed via natural fermentation and maintained under open-air condition, the indigo-reducing reactions continue for 6 months (on average) or longer. Identifying the mechanism underlying the maintenance of this process could lead to the development of a novel, long-lasting, unsterilized bioprocesses. To determine the mechanisms underlying the maintenance of the indigo fermentation system microbiota for more than 6 months in a reduced state in an anaerobic alkaline environment, we examined changes in the microbiota in one early-phase batch and two aged batches of indigo fermentation fluid. The microbiota in the aged fermentation fluid consisted mainly of the genera Alkalibacterium, Amphibacillus, Anaerobacillus and Polygonibacillus and the family Proteinivoraceae. The genera Alkalibacterium, Amphibacillus and Polygonibacillus are known to include indigo-reducing bacteria. Although the transition speed was slower in the aged fermentation fluid than in the early-stage fluid, the microbiota in the aged fermentation fluid maintained for more than 6 months was drastically changed within a period of 3 months. The results of this study indicate that the bacterial consortia consisted of various indigo-reducing species that replace the previous group of indigo-reducing bacteria. The notable transitional changes may be concomitant with changes in the environmental conditions, such as the nutritional conditions, observed over 3 months. This flexibility may lead to important changes in the microbiota that allow for the maintenance of a fermentation-reducing state over a long period.




Similar content being viewed by others
References
Aino K, Narihiro T, Minamida K, Kamagata Y, Yoshimune K, Yumoto I (2010) Bacterial community characterization and dynamics of indigo fermentation. FEMS Microbiol Ecol 74:174–183
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Clark RJH, Cooksey CJ, Daniels MAM, Withnall R (1993) Indigo, woad, and Tyrian purple: important vat dyes from antiquity to the present. Endeavour 17:191–199
Edger RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461
Farrow JAE, Lawson PA, Hippe H, Gauglitz U, Collins MD (1995) Phylogenetic evidence that the gram-negative nonsporulating bacterium Tissierella (Bacteroides) praeacuta is a member of the Clostridium subphylum of the gram-positive bacteria and description of Tissierella creatinini sp. nov. Int J Syst Bacteriol 45:436–440
Felsentein J (1985) Confidence limits on phylogenies: an approach using phylogenies: an approach using the bootstrap. Evol Int J org Evol 39:783–791
Fuller SJ, McMillan DGG, Renz MB, Schmidt M, Burke IT, Stewart DI (2014) Extracellular electron transport-mediated Fe(III) reduction by a community of alkaliphilic bacteria that use flavins as electron shuttle. Appl Environ Microbiol 80:128–137
Guindon S, Gascuel O. (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704
Hirota K, Aino K, Nodasaka Y, Morita N, Yumoto I (2013a) Amphibacillus indicireducens sp. nov., an alkaliphile that reduces an indigo dye. Int J Syst Evol Microbiol 63:464–469
Hirota K, Aino K, Nodasaka Y, Yumoto I (2013b) Oceanobacillus indicireducens sp. nov., a facultatively alkaliphile that reduces an indigo dye. Int J Syst Evol Microbiol 63:1437–1442
Hirota K, Aino K, Yumoto I (2013c) Amphibacillus iburiensis sp. nov., an alkaliphile that reduces an indigo dye. Int J Syst Evol Microbiol 63:4303–4308
Hirota K, Aino K, Yumoto I (2016a) Fermentibacillus polygoni gen. nov., sp. nov., an alkaliphile that reduces indigo dye. Int J Syst Evol Microbiol 66:2247–2253
Hirota K, Okamoto T, Matsuyama H, Yumoto I (2016b) Polygonibacillus indicireducensgen, nov., sp. nov., an indigo-reducing and obligate alkaliphile isolated from indigo fermentation liquor for dyeing. Int J Syst Evol Microbiol. doi:10.1099/ijsem.0.001015
Hobbie SN, Li X, Basen M, Stingl U, Brune A (2012) Humic substance-mediated Fe(III) reduction by a fermenting Bacillus strain from the alkaline gut of a humus-feeding scarab beetle larva. Syst Appl Microbiol 35:226–232
Kamagata Y (2015) Keys to cultivating uncultured microbes: elaborate enrichment strategies and resuscitation of dormant cells. Microb Environ 30:289–290
Kevbrin V, Boltyanskaya Y, Zhilina T, Kolganova T, Lavrentjeva E, Kuznetsov B (2013) Proteinivorax tanatarense gen. nov., sp. nov., an anaerobic, haloalkaliphilic, proteolytic bacterium isolated from a decaying algal bloom, and proposal of Proteinivoraceae fam. nov. Extremophiles 17:747–756
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Lucena-Padrós H, Ruiz-Barba JL (2016) Diversity and enumeration of halophilic and alkaliphilic bacteria in Spanish-style green table-olive fermentations. Food Microbiol 53:53–62
Mori K, Kamagata Y (2014) The challenges of studying the anaerobic microbial world. Microb Environ 29:335–337
Nakajima K, Hirota K, Nodasaka Y, Yumoto I (2005) Alkalibacterium iburiense sp. nov., an obligate alkaliphile that reduces an indigo dye. Int J Syst Evol Microbiol 55:1525–1530
Narihiro T, Kamagata Y (2013) Cultivating yet-to-be cultivated microbes: the challenge continues. Microb Environ 28:163–165
Ono H, Nishio S, Tsurii J, Kawamoto T, Sonomoto K, Nakayama J (2014) Monitoring of the microbiota profile in nukadoko, a naturally fermented rice bran bed for pickling vegetables. J Bioci Bioeng 118:520–525
Padden AN, Dillon VM, John P, Edmonds J, Collins MD, Alvarez N (1998) Clostridium used in medieval dyeing. Nature 396:225
Padden AN, Dillon VM, Edmonds J, Collins MD, Alvarez N, John P (1999) An indigo-reducing moderate thermophile from a woad vat, Clostridium isatidis sp. nov. Int J Syst Bacteriol 49:1025–1031
Roth E, Schwenninger SM, Eugster-Meier E, Lacroix C (2011) Facultative anaerobic halophilic and alkaliphilic bacteria isolated from a natural smear ecosystem inhibit Listeria growth in early ripening stages. Int J Food Microbiol 147:26–32
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Schmidt H (1997) Indigo—100 Jahre industrielle synthese. Chem Uns Zeit 31:121–128
Takahara Y, Tanabe O (1960) Studies on the reduction of indigo in industrial fermentation vat (VII). J Ferment Technol 38:329–331
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: Molecular evolutionary genetic analysis version 6.0. Mol Biol Evol 30:2725–2729
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucl Acid Res 22:4673–4680
Tiago I, Pires C, Mendes V, Morais PV, da Costa MS, Veríssimo A (2006) Bacillus foraminis sp. nov., isolated from a non-saline alkaline groundwater. Int J Syst Evol Microbiol 56:2571–2574
Vázquez-Baeza Y, Pirrung M, Gonzalez A, Knight R (2013) EMPeror: a tool for visualizing high-throughput microbial community data. GigaScience 2:16
Wei Q, Wang H, Chen Z, Lv Z, **e Y, Lu F (2013) Profiling of dynamic changes in the microbial community during the soy sauce fermentation process. Appl Microbiol Biotechnol 97:9111–9119
Yumoto I, Hirota K, Nodasaka Y, Yokota Y, Hoshino T, Nakajima K (2004) Alkalibacterium psychrotolerans sp. nov., a psychrotolerant obligate alkaliphile that reduces an indigo dye. Int J Syst Evol Microbiol 54:2379–2383
Yumoto I, Hirota K, Nodasaka Y, Tokiwa Y, Nakajima K (2008) Alkalibacterium indicireducens sp. nov., an obligate alkaliphile that reduces an indigo dye. Int J Syst Evol Microbiol 58:901–905
Acknowledgements
This study was supported by the Japan Society for the Promotion of Science with Grant-in-Aids for Scientific Research Nos. 23570128 and 16K07684 to I.Y. and 26630250 and 15H05331 to T.N.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that they have no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Okamoto, T., Aino, K., Narihiro, T. et al. Analysis of microbiota involved in the aged natural fermentation of indigo. World J Microbiol Biotechnol 33, 70 (2017). https://doi.org/10.1007/s11274-017-2238-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-017-2238-1