Abstract
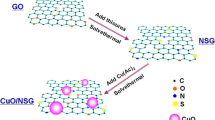
This work reports the synthesis of nitrogen-doped graphene (NG) in one step by the solvothermal method, obtaining small nitrogen graphene sheets.XPS reveals that pyridinic nitrogen groups have a higher proportion than pyrrolytic, graphitic and oxide nitrogen in NG. The NG material stands out to have a higher conductivity than graphene oxide (GO) and reduced graphene (RG). The NG material stands out to have a higher conductivity than graphene oxide and graphene nitrogen. The nitrogen-doped graphene sheets were employed in the electrochemical determination of glucose through the cathodic redissolution technique. Glucose quantification was carried out at pH 7 with a sensitivity of 1.3767 µA µM−1 in a range of 0 to 100 µM. The glucose determination with nitrogen-doped graphene was compared with reduced graphene (RG) and graphene oxide (GO) to evaluate the nitrogen effect.








Similar content being viewed by others
References
Kaushal S, Kaur M, Kaur N, Kumari V, Singh PP (2020) Heteroatom-doped graphene as sensing materials: a mini review. RSC Adv. https://doi.org/10.1039/D0RA04432F
Qiu B, **ng M, Zhang J (2018) Recent advances in three-dimensional graphene-based materials for catalysis applications. Chem Soc Rev. https://doi.org/10.1039/C7CS00904F
Duan J, Chen S, Jaroniec M, Qiao SZ (2015) Heteroatom-doped graphene-based materials for energy-relevant electrocatalytic processes. ACS Catal. https://doi.org/10.1021/acscatal.5b00991
Lee YH, Chang KH, Hu CC (2013) Differentiate the pseudocapacitance and double-layer capacitance contributions for nitrogen-doped reduced graphene oxide in acidic and alkaline electrolytes. J Power Sources. https://doi.org/10.1016/j.jpowsour.2012.11.026
Sliwak A, Grzyb B, Dıez N, Gryglewicz G (2017) Nitrogen-doped reduced graphene oxide as electrode material for high rate supercapacitors. Appl Surf Sci. https://doi.org/10.1016/j.apsusc.2016.12.060
Mohammadian A, Ebrahimi M, Maleh HK (2018) Synergic effect of 2D nitrogen doped reduced graphene nano-sheet and ionic liquid as a new approach for fabrication of anticancer drug sensor in analysis of doxorubicin and topotecan. J Mol Liq. https://doi.org/10.1016/j.molliq.2018.07.026
Cai ZX, Song XH, Chen YY, Wang YR, Chen X (2016) 3D nitrogen-doped graphene aerogel: a low-cost, facile prepared direct electrode for H2O2 sensing. Sens Actuators B. https://doi.org/10.1016/j.snb.2015.08.094
Ma CC, Shao XH, Cao DP (2014) Nitrogen-doped graphene as an excellent candidate for selective gas sensing. Science. https://doi.org/10.1007/s11426-014-5066-2
Cadore AR, Mania E, Alencar AB, Rezende NP, Oliveira S, Watanabe K, Taniguchi T, Chacham H, Campos LC, Lacerda RG (2018) Enhancing the response of NH3 graphene-sensors by using devices with different graphene-substrate distances. Sens Actuators B. https://doi.org/10.1016/j.snb.2018.03.164
Ren X, Ma H, Zhang T, Zhang Y, Yan T, Du B, Wei Q (2017) Sulfur-doped graphene-based immunological biosensing platform for multianalysis of cancer. ACS Appl Mater Interfaces. https://doi.org/10.1021/acsami.7b13416
Tian Y, Ma Y, Liu H, Zhang X, Peng W (2015) One-step and rapid synthesis of nitrogen and sulfur co-doped graphene for hydrogen peroxide and glucose sensing. J Electroanal Chem. https://doi.org/10.1016/j.jelechem.2015.01.029
Li J, Hu H, Li H, et al (2017) Recent developments in electrochemical sensors. based on nanomaterials for determining glucose and its byproduct H2O2. J Mater Sci. https://doi.org/10.1007/s10853-017-1221-4
Wang C, Hu H, Ding S, Dong M, Li L (2021) Highly sensitive electrochemical non-enzymatic glucose biosensor based on squamous NiCo2O4 nanosheets decorated nitrogen-doped reduced graphene oxide. J Mater Sci. https://doi.org/10.1007/s10853-017-1221-4
Gowthaman NSK, Amal M, John S (2017) Nitrogen-doped graphene as a robust scaffold for the homogeneous deposition of copper nanostructures: a non-enzymatic disposable glucose sensor. ACS Sustain Chem Eng. https://doi.org/10.1021/acssuschemeng.6b02390
**aoyi Y, Yue G, Cong L, Bo Z, Yaru L, Tingting Z, Zhiquan Z, Ming Y (2018) A non-enzymatic glucose sensor based on the CuS nanoflakes–reduced graphene oxide nanocomposite. Anal Methods. https://doi.org/10.1039/C7AY02290E
Raza W, Ahmad K, Kim H (2022) Nitrogen-doped graphene as an efficient metal-free catalyst for ammonia and non-enzymatic glucose sensing. J Phys Chem Solids. https://doi.org/10.1016/j.jpcs.2021.110359
Rahsepar M, Foroughi F, Kim H (2018) A new enzyme-free biosensor based on nitrogen-doped graphene with high sensing performance for electrochemical detection of glucose at biological pH value. Sens Actuators B. https://doi.org/10.1016/j.snb.2018.11.078
Rivera-Lugo YY, Pérez-Muñoz KI, Trujillo-Navarrete B, Silva-Carrillo C, Reynoso-Soto EA, Calva Yañez JC, Wai Lin S, Flores-Hernández JB, Félix-Navarro RM (2020) PtPd Hybrid composite catalysts as cathodes for proton exchange membrane fuel cells. Energies https://www.mdpi.com/1996-1073/13/2/316
Hummers WS, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc. https://doi.org/10.1021/ja01539a017
Wang YH, Deng HH, Liu YH, Shi XQ, Liu AL, Peng HP, Hong GL, Chen W (2016) Partially reduced graphene oxide as highly efficient DNA nanoprobe. Biosens Bioelectron. https://doi.org/10.1016/j.bios.2016.01.052
Deng D, Pan X, Yu L, Cui Y, Jiang Y, Qi J, Li WX, Fu Q, Ma X, Xue Q, Sun G, Bao X (2011) Toward N-doped graphene via solvothermal synthesis. Chem Mater. https://doi.org/10.1021/cm102666r
Al-Gaashani R, Najjar A, Zakaria Y, Mansour S, Atieh MA (2019) XPS and structural studies of high quality graphene oxide and reduced graphene oxide prepared by different chemical oxidation methods. Ceram. https://doi.org/10.1016/j.ceramint.2019.04.165
Zhang Y, Sun Z, Wang H, Wang Y, Liang M, Xue S (2015) Nitrogen-doped graphene as a cathode material for dye-sensitized solar cells: effects of hydrothermal reaction and annealing on electrocatalytic performance. RSC Adv. https://doi.org/10.1039/C4RA13224F
Baltrusaitis J, Jayaweera PM, Grassian VH (2009) XPS study of nitrogen dioxide adsorption on metal oxide particle surfaces under different environmental conditions. Phys Chem Chem Phys. https://doi.org/10.1039/B907584D
Kim G, Lee J, Liu T, Grey CP (2021) Characterizing nitrogen sites in nitrogen-doped reduced graphene oxide: a combined solid-state 15N NMR, XPS, and DFT approach. J Phys Chem C. https://doi.org/10.1021/acs.jpcc.1c02669
Farah S, Farkas A, Madarász J, László K (2020) Comparison of thermally and chemically reduced graphene oxides by thermal analysis and Raman spectroscopy. J Thermal Anal Calorim 142(1):331–337
Peng E, Todorova N, Yarovsky I (2018) Effects of size and functionalization on the structure and properties of graphene oxide nanoflakes: an in silico investigation. ACS Omega. https://doi.org/10.1021/acsomega.8b00866
Zaib M, Athar M (2017) Electrochemical characterization of a Porphyridium cruentum-modified carbon paste electrode by cyclic voltammetry. Instrum Sci Technol. https://doi.org/10.1080/10739149.2017.1394879
Rezaei B, Damiri S (2008) Voltammetric behavior of multi-walled carbon nanotubes modified electrode-hexacyanoferrate(II) electrocatalyst system as a sensor for determination of captopril. Sens. Actuators B. doi: https://doi.org/10.1016/j.snb.2008.05.004
Kang Z, Jiao K, Yu C, Dong J, Peng R, Hu Z, Jiao S (2017) Direct electrochemistry and bioelectrocatalysis of glucose oxidase in CS/CNC film and its application in glucose biosensing and biofuel cells. RSC Adv. https://doi.org/10.1039/C6RA26636C
Acknowledgements
The authors thank Tijuana Technological Institute for providing the facilities for this research. The authors also thank TecNM for financial assistance through project 10831.21-P.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Reynoso-Soto, E.A., Félix-Navarro, R.M., Rivera-Lugo, Y.Y. et al. Electrochemical Determination of Glucose Using Nitrogen-Doped Graphene. Top Catal 65, 1235–1243 (2022). https://doi.org/10.1007/s11244-022-01658-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-022-01658-7