Abstract
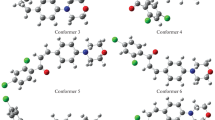
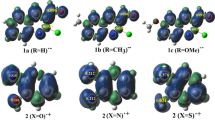
Density functional theory has been utilized for exploring the mechanism of Diels–Alder reaction between 7H-benzo[a]phenalene and maleic anhydride. 7H-Benzo[a]phenalene is an antiviral compound, and information available about its cycloaddition reactions with possible reaction path and mechanism is scarce. In order to work on the synthesis of its further potential derivatives, the mechanism of its reaction with all aspects should be well understood. Five novel intermediates involved in this reaction are reported in this work. Diels–Alder reaction of maleic anhydride has found many applications in the synthesis of wide range of useful products. One of the major concerns of this work is to evaluate the consequences of introducing electron donating and electron withdrawing substituents on the reactivity of maleic anhydride towards 7H-benzo[a]phenalene. Thermodynamic parameters, activation parameters, energies of frontier orbitals, global reactivity indices and global electron density transfer (GEDT) have been determined for all the reactions. Fukui functions are computed for each reactant in order to identify the most reactive sites. All the reactions have been found to proceed via normal electron demand having polar nature. The substituents with opposite electronic properties were expected to affect the reactivity of dienophile in an inverse manner; however, the results are not according to this assumption. Rather, both kinds of substituents increased the activation barrier of the reaction. This behavior has been explained in the light of various parameters such as the stability of reacting species and gap of frontier molecular orbitals. Experimental studies reported previously are in agreement with these results.









Similar content being viewed by others
Availability of data and materials
All the relevant data and materials are available and can be provided as per requirement.
Code availability
Not applicable.
References
Merzoud L, Guégan F, Chermette H, Morell C (2021) Understanding the intermolecular Diels-Alder cycloaddition promotion: activation strain model/energy decomposition analysis model and conceptual density functional theory viewpoints. J Comput Chem 42:1364–1372. https://doi.org/10.1002/jcc.26548
Samba WK, Tia R, Adei E (2021) Regio-, enantio-, peri-, and stereo-selectivities of the reactions of five-membered cyclodiene derivatives with itaconic anhydride toward the formation of norbornene lactones. J Phys Org Chem 34(2):e4132. https://doi.org/10.1002/poc.4132
Khan TS, Gupta S, Ahmad M, Alam MI, Haider MA (2020) Effect of substituents and promoters on the Diels-Alder cycloaddition reaction in the biorenewable synthesis of trimellitic acid. RSC Adv 10(51):30656–30670. https://doi.org/10.1039/D0RA04318D
Oluwasanmi A, Hoskins C (2021) Potential use of the Diels-Alder reaction in biomedical and nanomedicine applications. Int J Pharm 604:120727. https://doi.org/10.1016/j.ijpharm.2021.120727
Medrán NS, Dezotti F, Pellegrinet SC (2019) Remarkable reactivity of boron-substituted furans in the Diels-Alder reactions with maleic anhydride. Org Lett 21(13):5068–5072. https://doi.org/10.1021/acs.orglett.9b01662
Sultan MA, Karama U (2016) Substituent effects on regioselectivity of the Diels-Alder reactions: reactions of 10-allyl-1, 8-dichloroanthracene with 2-chloroacrylonitrile, 1-cyanovinyl acetate and phenyl vinyl sulfone. J Chem 2016:1–5. https://doi.org/10.1155/2016/3943060
Rulíšek L, Šebek P, Havlas Z, Hrabal R, Čapek P, Svatoš A (2005) An experimental and theoretical study of stereoselectivity of furan-maleic anhydride and furan-maleimide Diels-Alder reactions. J Org Chem 70(16):6295–6302. https://doi.org/10.1021/jo050759z
Lording WJ, Fallon T, Sherburn MS, Paddon-Row MN (2020) The simplest Diels-Alder reactions are not endo-selective. Chem Sci 11(43):11915–11926. https://doi.org/10.1039/D0SC04553E
Cole CJ, Fuentes L, Snyder SA (2020) Asymmetric pyrone Diels-Alder reactions enabled by dienamine catalysis. Chem Sci 11(8):2175–2180. https://doi.org/10.1039/C9SC05738B
El Ghozlani M, Barhoumi A, Elkacmi R, Aitouna AO, Zeroual A, El Idrissi M (2020) Mechanistic study of hetero-Diels–Alder [4+ 2] cycloaddition reactions between 2-nitro-1H-pyrrole and isoprene. Chemistry Africa 3(4):901–909. https://doi.org/10.1007/s42250-020-00187-8
Gara R, Zouaghi MO, L.M.H. ALshandoudi, Y. Arfaoui, (2021) DFT investigation of solvent, substituent, and catalysis effects on the intramolecular Diels-Alder reaction. J Mol Model 27(5):1–12. https://doi.org/10.1007/s00894-021-04729-w
Yoshioka S, Aoyama H, Fujioka H, Arisawa M (2018) Cascade multiple Diels-Alder reactions of styrene derivatives with maleimide or maleic anhydride. J Org Chem 83(12):6599–6606. https://doi.org/10.1021/acs.joc.8b00890
Mojica M, Méndez F, Alonso JA (2016) The Diels-Alder cycloaddition reaction of substituted hemifullerenes with 1, 3-butadiene: effect of electron-donating and electron-withdrawing substituents. Molecules 21(2):200. https://doi.org/10.3390/molecules21020200
Mahardiani L, Shrotri A, Kobayashi H, Fukuoka A (2020) Structure and activity of activated carbon functionalized with maleic anhydride by diels-alder reaction. Catal Today 357:409–415. https://doi.org/10.1016/j.cattod.2019.08.056
Khorief Nacereddine A, Merzoud L, Morell C, Chermette H (2021) A computational investigation of the selectivity and mechanism of the Lewis acid catalyzed oxa-Diels–Alder cycloaddition of substituted diene with benzaldehyde. J Comput Chem 42(18):1296–1311. https://doi.org/10.1002/jcc.26547
Kořenková M, Kremláček V, Hejda M, Turek J, Khudaverdyan R, Erben M, Jambor R, Růžička A, Dostál L (2020) Hetero Diels–Alder reactions of masked dienes containing heavy group 15 elements. Chem Eur J 26(5):1144–1154. https://doi.org/10.1002/chem.201904953
Sahrane M, Marakchi K, Ghailane R (2021) Theoretical study of the Diels-Alder reaction of 3-bromo-1-phenylprop-2-ynone with furan and 2-methylfuran. Theoret Chem Acc 140(8):1–10. https://doi.org/10.1007/s00214-021-02812-2
Kącka-Zych A, Jasiński R (2020) Molecular mechanism of Hetero Diels-Alder reactions between (E)-1, 1, 1-trifluoro-3-nitrobut-2-enes and enamine systems in the light of Molecular Electron Density Theory. J Mol Graph Model 101:107714. https://doi.org/10.1016/j.jmgm.2020.107714
Hall H Jr, Nogues P, Rhoades J, Sentman R, Detar M (1982) (Carbomethoxy) maleic anhydride, a highly reactive new dienophile and comonomer. J Org Chem 47(8):1451–1455. https://doi.org/10.1021/jo00347a014
Sakata K, Fujimoto H (2016) Origin of the endo selectivity in the Diels-Alder reaction between cyclopentadiene and maleic anhydride. Eur J Org Chem 2016(25):4275–4278. https://doi.org/10.1002/ejoc.201600676
Jia W, Si Z, Feng Y, Zhang X, Zhao X, Sun Y, Tang X, Zeng X, Lin L (2020) Oxidation of 5-[(Formyloxy) methyl] furfural to maleic anhydride with atmospheric oxygen using α-MnO2/Cu (NO3) 2 as catalysts. ACS Sustain Chem Eng 8(21):7901–7908. https://doi.org/10.1021/acssuschemeng.0c01144
Huynh VN, Leitner M, Bhattacharyya A, Uhlstein L, Kreitmeier P, Sakrausky P, Rehbein J, Reiser O (2020) Diels-Alder reactions and electrophilic substitutions with atypical regioselectivity enable functionalization of terminal rings of anthracene. Commun Chem 3(1):1–9. https://doi.org/10.1038/s42004-020-00407-9
Andrews L, Keefer R (1955) A kinetic study of the Diels-Alder reaction of various anthracene and maleic anhydride derivatives. J Am Chem Soc 77(23):6284–6289. https://doi.org/10.1021/ja01628a066
Karas LJ, Campbell AT, Alabugin IV, Wu JI (2020) Antiaromaticity gain activates tropone and nonbenzenoid aromatics as normal-electron-demand Diels-Alder dienes. Org Lett 22(18):7083–7087. https://doi.org/10.1021/acs.orglett.0c02343
Atalay A, Abbasoglu R (2019) Theoretical investigation on facial and stereoselectivity in Diels-Alder cycloadditions of maleic anhydride to dissymmetric cage-annulated cyclohexa-1, 3-dienes. J Phys Org Chem 32(2):e3893. https://doi.org/10.1002/poc.3893
Çelebi-Ölçüm N, Sanyal A, Aviyente V (2009) Understanding the stereoselection induced by chiral anthracene templates in Diels−Alder cycloaddition: a DFT study. J Org Chem 74(6):2328–2336. https://doi.org/10.1021/jo802365v
Stewart CA Jr (1963) Diene structure and Diels-Alder reactivity. J Org Chem 28(12):3320–3323. https://doi.org/10.1021/jo01047a010
Welker ME (2020) Boron and silicon-substituted 1, 3-dienes and dienophiles and their use in Diels-Alder reactions. Molecules 25(16):3740. https://doi.org/10.3390/molecules25163740
Dewyer AL, Zimmerman PM (2017) Finding reaction mechanisms, intuitive or otherwise. Org Biomol Chem 15(3):501–504. https://doi.org/10.1039/C6OB02183B
Akuamoah DA, Tia R, Adei E (2020) Computational study on the mechanism of the reaction of benzenesulfonyl azides with oxabicyclic alkenes. J Mol Model 26(11):1–9. https://doi.org/10.1007/s00894-020-04582-3
Ahraminejad M, Ghiasi R, Mohtat B, Ahmadi R (2021) Computational investigation of the substituent effect in the [2+ 4] Diels-Alder cycloaddition reactions of HSi≡ Si (para-C6H4X) with benzene. J Chin Chem Soc 68:806–816. https://doi.org/10.1002/jccs.202000428
Funes-Ardoiz I, Schoenebeck F (2020) Established and emerging computational tools to study homogeneous catalysis—from quantum mechanics to machine learning. Chem 6(8):1904–1913. https://doi.org/10.1016/j.chempr.2020.07.008
Abbiche K, Acharjee N, Salah M, Hilali M, Laknifli A, Komiha N, Marakchi K (2020) Unveiling the mechanism and selectivity of [3+ 2] cycloaddition reactions of benzonitrile oxide to ethyl trans-cinnamate, ethyl crotonate and trans-2-penten-1-ol through DFT analysis. J Mol Model 26(10):1–12. https://doi.org/10.1007/s00894-020-04547-6
Michael R. Johnson, Myung-Chol Kang, Alexei G. Nemazany, Kenner C. Rice. EP1021397A1 European Patent Office. https://patents.google.com/patent/EP1021397A1/en
Bakalova SM, Santos AG (2004) A computational study of the Diels-Alder reaction of ethyl-S-lactyl acrylate and cyclopentadiene. Origins of Stereoselectivity. J Org Chem 69(24):8475–8481. https://doi.org/10.1021/jo049298s
Domingo LR, Ríos-Gutiérrez M, Pérez P (2020) A molecular electron density theory study of the participation of tetrazines in aza-Diels–Alder reactions. RSC Adv 10(26):15394–15405. https://doi.org/10.1039/D0RA01548B
Uzun S, Esen Z, Koç E, Usta NC, Ceylan M (2019) Experimental and density functional theory (MEP, FMO, NLO, Fukui functions) and antibacterial activity studies on 2-amino-4-(4-nitrophenyl)-5, 6-dihydrobenzo [h] quinoline-3-carbonitrile. J Mol Struct 1178:450–457. https://doi.org/10.1016/j.molstruc.2018.10.001
Salavati-fard T, Caratzoulas S, Doren DJ (2015) DFT Study of solvent effects in acid-catalyzed Diels-Alder cycloadditions of 2, 5-dimethylfuran and maleic anhydride. J Phys Chem A 119(38):9834–9843. https://doi.org/10.1021/acs.jpca.5b05060
Hernández-Mancera JP, Núñez-Zarur F, Vivas-Reyes R (2020) Diels-Alder reactivity of a chiral anthracene template with symmetrical and unsymmetrical dienophiles: a DFT study. ChemistryOpen 9(7):748. https://doi.org/10.1002/open.202000137
Singh MD, Ningombam A (2010) High stereoselectivity in the Diels-Alder reaction of substituted anthracenes: reactions of 1-succinimidoanthracene and 1-phthalimidoanthracene with maleic anhydride. Indian J Chem 49:789–794
Kloetzel MC (2011) The Diels‐Alder reaction with maleic anhydride. Org Synthesis 30–31
Linder M, Brinck T (2013) On the method-dependence of transition state asynchronicity in Diels-Alder reactions. Phys Chem Chem Phys 15(14):5108–5114. https://doi.org/10.1039/C3CP44319A
Hernandez Mancera JP, Nunez-Zarur F, Gutierrez-Oliva S, Toro-Labbe A, Vivas-Reyes R (2020) Diels-Alder reaction mechanisms of substituted chiral anthracene: a theoretical study based on the reaction force and reaction electronic flux. J Comput Chem 41(23):2022–2032. https://doi.org/10.1002/jcc.26360
Kron KJ, Kosich M, Cave RJ, Vosburg DA (2018) Divergent Diels-Alder reactions in the biosynthesis and synthesis of endiandric-type tetracycles: a computational study. J Org Chem 83(18):10941–10947. https://doi.org/10.1021/acs.joc.8b01594
Jasiński R (2018) β-trifluoromethylated nitroethenes in Diels-Alder reaction with cyclopentadiene: a DFT computational study. J Fluorine Chem 206:1–7. https://doi.org/10.1016/j.jfluchem.2017.12.008
Fakhar Z, Govender T, Lamichhane G, Maguire GEM, Kruger HG, Honarparvar B (2017) Computational model for the acylation step of the β-lactam ring: potential application for l, d-transpeptidase 2 in mycobacterium tuberculosis. J Mol Struct 1128:94–102. https://doi.org/10.1016/j.molstruc.2016.08.049
Domingo LR, Ríos-Gutiérrez M, Pérez P (2017) How does the global electron density transfer diminish activation energies in polar cycloaddition reactions? A molecular electron density theory study. Tetrahedron 73(13):1718–1724. https://doi.org/10.1016/j.tet.2017.02.012
Bakalova SM, Kaneti J, Markova N (2017) Computational study of exo-selective Diels-Alder cycloadditions, catalyzed by aluminum chloride. Bul Chem Commun 49:49195–49200
Yepes D, Pérez P, Jaque P, Fernández I (2017) Effect of Lewis acid bulkiness on the stereoselectivity of Diels-Alder reactions between acyclic dienes and α, β-enals. Org Chem Front 4(7):1390–1399. https://doi.org/10.1039/C7QO00154A
Bandyopadhyay P, Ray S, Seikh MM (2019) Unraveling the regioselectivity of odd electron halogen bond formation using electrophilicity index and chemical hardness parameters. Phys Chem Chem Phys 21(48):26580–26590. https://doi.org/10.1039/C9CP05374C
Jasinski R, Jasinska E, Dresler E (2017) A DFT computational study of the molecular mechanism of [3 + 2] cycloaddition reactions between nitroethene and benzonitrile N-oxides. J Mol Model 23(1):13. https://doi.org/10.1007/s00894-016-3185-8
Jasinski R (2017) One-step versus two-step mechanism of Diels-Alder reaction of 1-chloro-1-nitroethene with cyclopentadiene and furan. J Mol Graph Model 75:55–61. https://doi.org/10.1016/j.jmgm.2017.04.008
Afshari T, Mohsennia M (2019) A molecular electron density theory study of Diels-Alder reaction between Danishefsky’s diene and (2E)-3-phenyl-2-(trifluoromethyl) acrylonitrile. J Phys Org Chem 32(6):e3937. https://doi.org/10.1002/poc.3937
De Proft F, Van Alsenoy C, Peeters A, Langenaeker W, Geerlings P (2002) Atomic charges, dipole moments, and Fukui functions using the Hirshfeld partitioning of the electron density. J Comput Chem 23(12):1198–1209. https://doi.org/10.1002/jcc.10067
Ali JM, Lelisho TA (2020) Chemical fixation of CO2 with propylene oxide catalyzed by trimethylsulfonium bromide in the presence of HBr: a computational study. Theoret Chem Acc 139(10):1–10. https://doi.org/10.1007/s00214-020-02673-1
Soleymani M (2018) DFT study of double 1,3-dipolar cycloaddition of nitrilimines with allenoates. Monatsh Chem 149(12):2183–2193. https://doi.org/10.1007/s00706-018-2311-y
Hoque MJ, Ahsan A, Hossain MB (2018) Molecular docking, pharmacokinetic, and DFT calculation of naproxen and its degradants. Biomed J Sci Tech Res 9(5):7360–7365
Schmauck J, Breugst M (2017) The potential of pnicogen bonding for catalysis—a computational study. Org Biomol Chem 15(38):8037–8045. https://doi.org/10.1039/C7OB01599B
Soleymani M (2019) Theoretical study on the [4+2] cycloaddition of 1,3-dimethylindole with 2,6-dimethylquinone. Struct Chem 30(4):1173–1184. https://doi.org/10.1007/s11224-018-1259-1
Andrés J, Berski S, Silvi B (2016) Curly arrows meet electron density transfers in chemical reaction mechanisms: from Electron Localization Function (ELF) analysis to valence-shell electron-pair repulsion (VSEPR) inspired interpretation†. ChemComm 52(53):8183–8195. https://doi.org/10.1039/C5CC09816E
Lawal MM, Govender T, Maguire GEM, Kruger HG, B, (2018) Honarparvar, DFT study of the acid-catalyzed esterification reaction mechanism of methanol with carboxylic acid and its halide derivatives. Int J Quantum Chem 118(4):e25497. https://doi.org/10.1002/qua.25497
Grimmel SA, Reiher M (2019) The electrostatic potential as a descriptor for the protonation propensity in automated exploration of reaction mechanisms. Faraday Discuss 220:443–463. https://doi.org/10.1039/C9FD00061E
Patel TR, Ganguly B (2020) Revealing the origin of Π-facial and regioselectivity in the Diels-Alder reaction of unsymmetrical, cage-annulated 1,3-cyclohexadiene with ethyl propiolate dienophile: a DFT study. ChemistrySelect 5(43):13524–13529. https://doi.org/10.1002/slct.202003443
Sandhya KS, Suresh CH (2014) Designing metal hydride complexes for water splitting reactions: a molecular electrostatic potential approach. Dalton Trans 43(32):12279–12287. https://doi.org/10.1039/C4DT01343C
Nazeer U, Rasool N, Mujahid A, Mansha A, Zubair M, Kosar N, Mahmood T, Raza Shah A, Shah SAA, Zakaria ZA, Akhtar MN (2020) Selective arylation of 2-bromo-4-chlorophenyl-2-bromobutanoate via a Pd-catalyzed Suzuki cross-coupling reaction and its electronic and non-linear optical (NLO) properties via DFT studies. Molecules 25(15):3521. https://doi.org/10.3390/molecules25153521
Mubarik A, Rasool N, Hashmi MA, Mansha A, Zubair M, Shaik MR, Sharaf MAF, Awwad EM, Abdelgawad A (2021) Computational study of structural, molecular orbitals, optical and thermodynamic parameters of thiophene sulfonamide derivatives. Curr Comput-Aided Drug Des 11(2):211. https://doi.org/10.3390/cryst11020211
Odame F (2018) DFT Study of the reaction mechanism of N-(carbomylcarbamothioyl) benzamide. Acta Chimica Slovenica 65(2):328–332. https://doi.org/10.17344/acsi.2017.4001
Zou Y, Yang S, Sanders JN, Li W, Yu P, Wang H, Tang Z, Liu W, Houk KN (2020) Computational investigation of the mechanism of Diels-Alderase PyrI4. J Am Chem Soc 142(47):20232–20239. https://doi.org/10.1021/jacs.0c10813
Sultan MA, Karama U, Almansour AI, Soliman SM (2016) Theoretical study on regioselectivity of the Diels-Alder reaction between 1,8-dichloroanthracene and acrolein. Molecules 21(10):1277. https://doi.org/10.3390/molecules21101277
Borisevich SS, Kovalskaya AV, Tsypysheva IP, Khursan SL (2014) Thermodynamically controlled Diels-Alder reaction of 12-N-methylcytisine: a DFT study. J Theor Comput Chem 13(06):1450048. https://doi.org/10.1142/S0219633614500485
Benallou A (2019) Solvent effect on Hetero-Diels-Alder reaction of isoselenazole with symmetrical acetylenic dienophiles: a MEDT study. Comput Theor Chem 1154:17–25. https://doi.org/10.1016/j.comptc.2019.03.007
Hassan RJ, Abdallah HH (2020) Theoretical study of Diels-Alder reaction of but-3-en-2-one with hexa-1,2,4-triene: a density functional theory study. Aro-the Sci J Koya University 8(1):74. https://doi.org/10.14500/aro.10632
Yadav P, Tandon H, Malik B, Chakraborty T (2020) A new approach to compute atomic electrophilicity index in terms of Gordy’s electronegativity. J Chem Res 45(1–2):201–206. https://doi.org/10.1177/1747519820937272
Tandon H, Chakraborty T, Suhag V (2020) A scale of atomic electronegativity in terms of atomic nucleophilicity index. Found Chem 22(2):335–346. https://doi.org/10.1007/s10698-020-09358-4
Li W, Li C, Lyu Y (2019) A computational mechanistic insight into H2 activation and CO2 reduction over β-diketiminato-ligated group 13 metal complexes. J Catal 373:1–12. https://doi.org/10.1016/j.jcat.2019.03.029
Demircioğlu Z, Kaştaş ÇA, Büyükgüngör O (2015) Theoretical analysis (NBO, NPA, Mulliken Population Method) and molecular orbital studies (hardness, chemical potential, electrophilicity and Fukui function analysis) of (E)-2-((4-hydroxy-2-methylphenylimino)methyl)-3-methoxyphenol. J Mol Struct 1091:183–195. https://doi.org/10.1016/j.molstruc.2015.02.076
Zhang M, Wei D, Wang Y, Li S, Liu J, Zhu Y, Tang M (2014) DFT study on the reaction mechanisms and stereoselectivities of NHC-catalyzed [2 + 2] cycloaddition between arylalkylketenes and electron-deficient benzaldehydes. Org Biomol Chem 12(33):6374–6383. https://doi.org/10.1039/C4OB00606B
Kour M, Gupta R, Bansal RK (2017) Experimental and theoretical investigation of the reaction of secondary amines with maleic anhydride. Aust J Chem 70(12):1247. https://doi.org/10.1071/CH17206
Pilepić V, Uršić S (2001) Nucleophilic reactivity of the nitroso group. Fukui function DFT calculations for nitrosobenzene and 2-methyl-2-nitrosopropane. J Mol Struct Theochem 538(1–3):41–49. https://doi.org/10.1016/S0166-1280(00)00642-4
Liu S, Hao Hu, Pedersen LG (2010) Steric, quantum, and electrostatic effects on SN2 reaction barriers in gas phase. J Phys Chem A 114(18):5913–5918. https://doi.org/10.1021/jp101329f
Siadati SA, Nami N (2016) Investigation of the possibility of functionalization of C20 fullerene by benzene via Diels-Alder reaction. Physica E 84:55–59. https://doi.org/10.1016/j.physe.2016.05.041
Funding
A.M. is grateful to Higher Education Commission (HEC) Pakistan for providing financial support for this study (NRPU/5613).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by UR, AM, MZ, SA and ZAR. The first draft of the manuscript was written by AFZ, AM, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rehman, U., Mansha, A., Zahid, M. et al. Quantum mechanical modeling unveils the effect of substitutions on the activation barriers of the Diels–Alder reactions of an antiviral compound 7H-benzo[a]phenalene. Struct Chem 33, 1907–1920 (2022). https://doi.org/10.1007/s11224-022-01948-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-022-01948-6